-
Name
METHYL PYRAZINE-2-CARBOXYLATE
- EINECS 228-198-6
- CAS No. 6164-79-0
- Article Data66
- CAS DataBase
- Density 1.213 g/cm3
- Solubility
- Melting Point 57-61 °C(lit.)
- Formula C6H6N2O2
- Boiling Point 220.4 °C at 760 mmHg
- Molecular Weight 138.126
- Flash Point 87.1 °C
- Transport Information
- Appearance white powder
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Pyrazinecarboxylicacid, methyl ester (7CI,8CI,9CI);Pyrazinoic acid methyl ester (6CI);2-Methoxycarbonylpyrazine;Methyl 2-pyrazinecarboxylate;Methylpyrazinecarboxylate;Methyl pyrazinoate;NSC 32337;Pyrazine-2-carboxylic acid,methyl ester;
- PSA 52.08000
- LogP 0.26320
Synthetic route
-
-
186581-53-3, 908094-01-9
diazomethane

-
-
98-97-5
2-pyrazylcarboxylic acid

-
-
6164-79-0
methyl pyrazine-2-carboxylate

| Conditions | Yield |
|---|---|
| In diethyl ether; dichloromethane at 20℃; for 0.25h; | 100% |
-
-
14508-49-7
2-chloropyrazin

-
-
67-56-1
methanol

-
-
201230-82-2
carbon monoxide

-
-
6164-79-0
methyl pyrazine-2-carboxylate

| Conditions | Yield |
|---|---|
| With dichloro[2,2'-bis(diphenylphosphino)-1,1'-binaphthyl]palladium(II); triethylamine at 100℃; under 2585.74 Torr; for 4h; | 99% |
| With 1,3-bis-(diphenylphosphino)propane; palladium diacetate; triethylamine In acetonitrile at 180℃; under 25502.6 Torr; Flow reactor; | 61% |
| With triethylamine; palladium bis(dibenzylideneacetone)palladium(0); triphenylphosphine at 120℃; under 29420.3 Torr; for 16h; other chloropyrazines; | 93 % Chromat. |
| With ethyl azide; triphenylphosphine; bis(dibenzylideneacetone)-palladium(0) at 120℃; under 29420.3 Torr; for 16h; | 93 % Chromat. |


| Conditions | Yield |
|---|---|
| With sulfuric acid for 2h; Reflux; | 98% |
| With sulfuric acid for 48h; Heating; | 95% |
| With sulfuric acid at 80 - 85℃; for 5h; | 94.8% |

| Conditions | Yield |
|---|---|
| With sodium persulfate; sulfuric acid; silver nitrate In dichloromethane; water for 1.5h; Heating; | 93% |

| Conditions | Yield |
|---|---|
| With Eosin Y; di-tert-butyl 1,4-dihydro-2,6-dimethyl-3,5-pyridine-dicarboxylate In acetonitrile at 20℃; for 5h; Inert atmosphere; Irradiation; chemoselective reaction; | 88% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-pyrazylcarboxylic acid With sulfuric acid In methanol at 20℃; for 5h; Reflux; Stage #2: With sodium hydrogencarbonate In methanol pH=8.5; | 63.63% |
| Multi-step reaction with 2 steps 1: 74 percent / SOCl2 / benzene / 2 h / Heating 2: pyridine / CH2Cl2 / a) 0 deg C, 1 h, b) RT, overnight View Scheme | |
| With thionyl chloride; sodium bicarbonate In methanol; di-isopropyl ether; water | |
| With sulfuric acid; sodium hydrogencarbonate In methanol; toluene |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 72h; | 8% |
-
-
73763-86-7
3-[(methyloxy)carbonyl]-2-pyrazinecarboxylic acid

-
-
6164-79-0
methyl pyrazine-2-carboxylate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane a) 0 deg C, 1 h, b) RT, overnight; |
-
-
13925-00-3
2-ethylpyrazine

-
-
6164-79-0
methyl pyrazine-2-carboxylate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: potassium permanganate / H2O / 20 °C 2: HCl View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 210 °C / 3 - 4 Torr 2: HCl View Scheme |

| Conditions | Yield |
|---|---|
| In methanol |
-
-
67-56-1
methanol

-
-
1254073-95-4
4-[(3-chloro-2-methylphenyl)carbonyl]-2-piperazinone

-
-
6164-79-0
methyl pyrazine-2-carboxylate

| Conditions | Yield |
|---|---|
| With thionyl chloride at 20℃; Reflux; |

-
-
6164-79-0
methyl pyrazine-2-carboxylate

| Conditions | Yield |
|---|---|
| With potassium carbonate In dichloromethane for 5h; | 37 g |

| Conditions | Yield |
|---|---|
| With hydrazine In ethanol for 2h; Heating / reflux; | 100% |
| With hydrazine In ethanol for 2h; Heating / reflux; | 100% |
| With hydrazine In ethanol for 2h; Reflux; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: methyl pyrazine-2-carboxylate With lithium aluminium tetrahydride In tetrahydrofuran at -78 - 72℃; for 0.333333h; Stage #2: With hydrogenchloride; water | 100% |
| With lithium aluminium tetrahydride In tetrahydrofuran at -83℃; for 2h; | 68% |
| With lithium aluminium tetrahydride In tetrahydrofuran at -78℃; for 1.83333h; Inert atmosphere; Schlenk technique; | 53.4% |
-
-
6164-79-0
methyl pyrazine-2-carboxylate

-
-
554-12-1
propanoic acid methyl ester

-
-
324737-10-2
methyl 2-methyl-3-(pyrazin-2-yl)-3-oxopropionate

| Conditions | Yield |
|---|---|
| Stage #1: methyl pyrazine-2-carboxylate With sodium hydride In toluene at 15 - 25℃; for 0.5h; Industrial scale; Stage #2: propanoic acid methyl ester In methanol; toluene at 15 - 40℃; Temperature; Solvent; Reagent/catalyst; Industrial scale; | 100% |
| Stage #1: propanoic acid methyl ester With potassium tert-butylate In tetrahydrofuran at 0℃; for 0.5h; Stage #2: methyl pyrazine-2-carboxylate In tetrahydrofuran at 0 - 25℃; for 3.5h; Stage #3: With ammonium chloride In tetrahydrofuran for 0.5h; | 89% |
| With sodium hydride In N,N-dimethyl-formamide; mineral oil at 20℃; for 5h; Inert atmosphere; | 79% |
| With NaH; ammonium chloride In toluene |

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran; mineral oil for 20h; Reflux; | 96% |

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; sodium methylate In methanol at 25℃; for 3h; Inert atmosphere; | 95% |
| Stage #1: methyl pyrazine-2-carboxylate With sodium tetrahydroborate In water for 0.5h; Stage #2: With water; potassium carbonate In ethanol for 0.5h; | 80% |
| With sodium tetrahydroborate; calcium chloride In ethanol at 25℃; for 5h; | 75% |

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide at 20℃; for 12h; | 94% |
-
-
6164-79-0
methyl pyrazine-2-carboxylate

-
-
73183-34-3
bis(pinacol)diborane

-
-
1393720-91-6
methyl 1,4-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1,4-dihydropyrazine-2-carboxylate

| Conditions | Yield |
|---|---|
| In pentane N2; ligand and B compd. (1.1:1 molar ratio) stirred at room temp. for 2 h; | 93% |
| In pentane at 20℃; for 2h; Inert atmosphere; | 93% |
-
-
6164-79-0
methyl pyrazine-2-carboxylate

-
-
60-34-4
methylhydrazine

-
-
80364-57-4
pyrazine-2-carboxylic acid N'-methylhydrazide

| Conditions | Yield |
|---|---|
| In ethanol Heating; | 90% |
| In ethanol Heating; | 81.1% |
| In ethanol for 1.5h; Heating; | 68% |
-
-
6164-79-0
methyl pyrazine-2-carboxylate

-
-
100-46-9
benzylamine

-
-
347368-07-4
pyrazine-2-carboxylic acid benzylamide

| Conditions | Yield |
|---|---|
| With niobium(V) oxide In neat (no solvent) at 140℃; for 30h; Molecular sieve; Inert atmosphere; | 90% |
| for 2h; Heating; | 70% |
| With potassium phosphate; 2,2,2-trifluoroethanol In tetrahydrofuran at 90℃; for 22h; Sealed tube; | 64% |

| Conditions | Yield |
|---|---|
| With sodium t-butanolate In neat (no solvent) at 20℃; for 1h; Inert atmosphere; Schlenk technique; Green chemistry; | 90% |
-
-
6164-79-0
methyl pyrazine-2-carboxylate

-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
31469-25-7
1,1-bis(trimethylsiloxy)-2-methylprop-1-ene

| Conditions | Yield |
|---|---|
| Stage #1: methyl pyrazine-2-carboxylate; trifluoromethylsulfonic anhydride In dichloromethane at -30℃; Inert atmosphere; Stage #2: 1,1-bis(trimethylsiloxy)-2-methylprop-1-ene In dichloromethane at -30 - 20℃; for 12h; Inert atmosphere; | 89% |

-
-
6164-79-0
methyl pyrazine-2-carboxylate

| Conditions | Yield |
|---|---|
| With hydroxylamine hydrochloride; sodium hydroxide In methanol for 24h; Inert atmosphere; Schlenk technique; Cooling with ice; | 89% |
-
-
6164-79-0
methyl pyrazine-2-carboxylate

-
-
171504-79-3
methyl 1,4,5,6-tetrahydropyrazine-2-carboxylate

| Conditions | Yield |
|---|---|
| palladium-carbon In methanol | 86% |
| With hydrogen; palladium on activated charcoal |

| Conditions | Yield |
|---|---|
| With hydroxylamine In methanol at 20℃; for 72h; | 85% |
| With hydroxylamine |
-
-
6164-79-0
methyl pyrazine-2-carboxylate

-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
40348-04-7
(Bis-trimethylsilanyloxy-methylene)-cyclohexane

| Conditions | Yield |
|---|---|
| Stage #1: methyl pyrazine-2-carboxylate; trifluoromethylsulfonic anhydride In dichloromethane at -30℃; Inert atmosphere; Stage #2: (Bis-trimethylsilanyloxy-methylene)-cyclohexane In dichloromethane at -30 - 20℃; for 12h; Inert atmosphere; | 84% |

-
-
6164-79-0
methyl pyrazine-2-carboxylate

-
-
20010-99-5
2-aminomethylpyrazine

| Conditions | Yield |
|---|---|
| In methanol for 168h; Reflux; | 83% |

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; diethyl ether; hexane -80 deg C to rt; | 80% |

| Conditions | Yield |
|---|---|
| In chloroform at 5℃; for 24h; Inert atmosphere; | 78% |
-
-
6164-79-0
methyl pyrazine-2-carboxylate

-
-
120068-79-3
5-amino-3-cyano-1-(2,6-dichloro-4-trifluoromethylphenyl)pyrazole

-
-
315208-01-6
N-[3-cyano-1-(2,6-dichloro-4-trifluoromethylphenyl)pyrazole-5-yl]pyrazine-2-carboxamide

| Conditions | Yield |
|---|---|
| With hydrogenchloride; CH3ONa In methanol; water; ethyl acetate; acetonitrile | 77% |

| Conditions | Yield |
|---|---|
| With sodium methylate In N,N-dimethyl-formamide for 2.5h; Heating; | 76% |

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide; mineral oil at 20℃; for 5h; Inert atmosphere; | 75% |
-
-
3731-51-9
(2-aminomethylpyridine)

-
-
6164-79-0
methyl pyrazine-2-carboxylate

-
-
549493-17-6
pyrazine-2-carboxylic acid (pyridine-2-ylmethyl)-amide

| Conditions | Yield |
|---|---|
| In methanol for 120h; Reflux; | 73% |
-
-
6164-79-0
methyl pyrazine-2-carboxylate

-
-
23611-75-8
6-chloro-pyrazine-2-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide In N,N-dimethyl-formamide at 80℃; | 72% |
| Multi-step reaction with 2 steps 1: 3-chloro-benzenecarboperoxoic acid / 1,2-dichloro-ethane / 16 h / 60 °C 2: thionyl chloride / 8 h / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1: 3-chloro-benzenecarboperoxoic acid / 1,2-dichloro-ethane / 60 °C 2: thionyl chloride / 85 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 3-chloro-benzenecarboperoxoic acid / 1,2-dichloro-ethane / 60 °C 2: thionyl chloride / 85 °C View Scheme | |
| Stage #1: methyl pyrazine-2-carboxylate With 3-chloro-benzenecarboperoxoic acid In 1,2-dichloro-ethane at 60℃; for 16h; Stage #2: With thionyl chloride at 75℃; for 8h; |

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In 1,2-dichloro-ethane at 60℃; for 16h; | 64% |
| With 3-chloro-benzenecarboperoxoic acid In 1,2-dichloro-ethane at 60℃; for 16h; | 64% |
| With 3-chloro-benzenecarboperoxoic acid In 1,2-dichloro-ethane at 60℃; for 16h; | 64% |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 20℃; for 48h; | 22% |
Methyl pyrazine-2-carboxylate Specification
The IUPAC name of 2-Pyrazinecarboxylic acid, methyl ester is methyl pyrazine-2-carboxylate. With the CAS registry number 6164-79-0 and EINECS 228-198-6, it is also named as Pyrazinoic acid methyl ester. The product's categories are Heterocycles; Pharmacetical; Piperazine Series; Esters; Pyrazines, Pyrimidines & Pyridazines; Pyrazine; Building Blocks; Heterocyclic Building Blocks. It is powder which is stable under normal temperature and pressure. Additionally, this chemical should be sealed in the container and stored in the cool and dry place.
The other characteristics of this product can be summarized as: (1)ACD/LogP: -0.32; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.32; (4)ACD/LogD (pH 7.4): -0.32; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 15.89; (8)ACD/KOC (pH 7.4): 15.89; (9)#H bond acceptors: 4; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 52.08 Å2; (13)Index of Refraction: 1.513; (14)Molar Refractivity: 34.2 cm3; (15)Molar Volume: 113.7 cm3; (16)Polarizability: 13.56×10-24 cm3; (17)Surface Tension: 48.6 dyne/cm; (18)Enthalpy of Vaporization: 45.68 kJ/mol; (19)Vapour Pressure: 0.113 mmHg at 25°C; (20)Rotatable Bond Count: 2; (21)Exact Mass: 138.042927; (22)MonoIsotopic Mass: 138.042927; (23)Topological Polar Surface Area: 52.1; (24)Heavy Atom Count: 10; (25)Complexity: 127.
Preparation of 2-Pyrazinecarboxylic acid, methyl ester: It can be obtained by pyrazine-2-carboxylic acid and methanol. This reaction needs reagent conc. H2SO4 by heating. The reaction time is 48 hours. The yield is 95%.
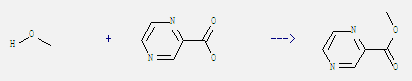
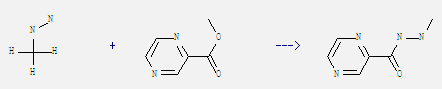
Uses of 2-Pyrazinecarboxylic acid, methyl ester: It can react with methylhydrazine to get pyrazine-2-carboxylic acid-(N'-methyl-hydrazide). This reaction needs solvent ethanol by heating. The yield is 81.1%.
When you are using this chemical, please be cautious about it as the following:
It is irritating to eyes, respiratory system and skin. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If you want to contact this product, you must wear suitable protective clothing.
People can use the following data to convert to the molecule structure.
1. SMILES:O=C(OC)c1nccnc1
2. InChI:InChI=1/C6H6N2O2/c1-10-6(9)5-4-7-2-3-8-5/h2-4H,1H3
3. InChIKey:TWIIRMSFZNYMQE-UHFFFAOYAU
Related Products
- Methyl (+)-(3R)-7-[4-(4-fluorophenyl)-6-isopropyl-2-(N-methyl-N-methanesulfonylamino)pyrimidin-5-yl]-3-hydroxy-5-oxo-(6E)-heptenoate
- Methyl (2-amino-5-methyl-1,3-thiazol-4-yl)acetate
- Methyl (2E)-3-(4-methylphenyl)propenoate
- Methyl (2E)-3-cyclohexylprop-2-enoate
- Methyl (2R)-2-[(tert-butoxycarbonyl)amino]-3-iodopropanoate
- Methyl (2R)-2-[4-(2,4-dichlorophenoxy)phenoxy]propanoate
- Methyl (2R)-2-amino-2-cyclohexylethanoate hydrochloride
- Methyl (2R)-2-bromo-2-(2-chlorophenyl)acetate
- Methyl (2R,3S)-3-(4-methoxyphenyl)-2-oxiranecarboxylate
- Methyl (2R,3S)-3-(benzoylamino)-2-hydroxy-3-phenylpropanoate
- 6164-80-3
- 6164-87-0
- 616-55-7
- 6165-68-0
- 6165-69-1
- 6165-75-9
- 6165-76-0
- 61657-67-8
- 61658-41-1
- 616-62-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View