-
Name
(-)-MYRTENAL
- EINECS
- CAS No. 564-94-3
- Article Data97
- CAS DataBase
- Density 1.061g/cm3
- Solubility
- Melting Point 53-54 °C
- Formula C10H14 O
- Boiling Point 215.7°Cat760mmHg
- Molecular Weight 150.221
- Flash Point 78.9°C
- Transport Information
- Appearance
- Safety Poison by intravenous route. Moderately toxic by ingestion. Slightly toxic by skin contact. When heated to decomposition it emits acrid smoke and irritating vapors.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 2-Norpinene-2-carboxaldehyde,6,6-dimethyl- (6CI,7CI,8CI); (?à)-Myrtenal; 6,6-Dimethyl-2-norpinene-2-carboxaldehyde;6,6-Dimethylbicyclo[3.1.1]-hept-2-ene-2-carboxaldehyde; Myrtenal; NSC 54384
- PSA 17.07000
- LogP 2.17770
Synthetic route

| Conditions | Yield |
|---|---|
| With bromobenzene; sodium hydride; triphenylphosphine; palladium diacetate In tetrahydrofuran at 65℃; for 1h; | 100% |
| With bromobenzene; sodium hydride; palladium diacetate; triphenylphosphine In tetrahydrofuran at 65℃; for 1h; | 95% |
| With calcium hydride; oxygen; 2,2,6,6-tetramethyl-piperidine-N-oxyl; copper dichloride In acetonitrile at 25℃; for 3h; | 93% |

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With carbon tetrabromide In water; acetonitrile at 45℃; for 2h; sonication; | 99% |
-
-
80-56-8, 177698-18-9
Pinene

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With pentafluorobenzeneseleninic acid In benzene for 2h; Heating; | 85% |
| With selenium(IV) oxide In 1,4-dioxane; ethanol at 60℃; | 70% |
| With perfluorooctylselenic acid; iodosylbenzene In various solvent(s) Heating; | 62% |
| With selenium(IV) oxide | |
| With selenium(IV) oxide In 1,4-dioxane; ethanol at 65 - 95℃; |
-
-
80-56-8, 177698-18-9
Pinene

-
A
-
515-00-4
myrtenol

-
B
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With 2-(N-oxido)pyridineseleninic anhydride In dichloromethane at 20℃; for 18h; | A 57% B 28% |
-
-
515-00-4
myrtenol

-
-
108-95-2
phenol

-
A
-
514-99-8
7,7-dimethyl-2-(hydroxymethyl)bicyclo<3.1.1>heptane


-
D
-
1344744-38-2
4-(6,6-dimethylbicyclo[3.1.1]hept-2-en-2-ylmethyl)-9-isopropyl-8,9,10,10a-tetrahydro-6H-benzo[c]chromene

-
E
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: myrtenol; phenol With aluminum isopropoxide at 160℃; Stage #2: With hydrogenchloride In water | A n/a B 1.5% C 12% D n/a E 6% |

-
-
75-09-2
dichloromethane

-
-
81971-91-7
peroxyde de t-butyle et de myrtenyle

-
B
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With sodium carbonate at 110℃; for 12h; | A 10% B 6% |

-
-
177698-19-0
Beta-pinene

-
A
-
24903-95-5
nopinone

-
B
-
16812-40-1
pinocarvone

-
C
-
515-00-4
myrtenol

-
D
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; titanium(IV) oxide In acetonitrile for 6h; Ambient temperature; Irradiation; Yield given. Yields of byproduct given. Title compound not separated from byproducts; |

-
-
177698-19-0
Beta-pinene

-
A
-
16812-40-1
pinocarvone

-
B
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; sodium acetate; sodium chloride; palladium dichloride 1.) AcOH, reflux; 2.) CH3CN, irradiation λ=366 nm; Yield given. Multistep reaction. Yields of byproduct given; |
-
-
177698-19-0
Beta-pinene

-
A
-
58434-29-0
myrtenyl hydroperoxide

-
-
105815-51-8
7,7-Dimethyl-3-oxatricyclo<4.1.1.02,4>octane-2-methanol

-
C
-
515-00-4
myrtenol

-
D
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; 5,15,10,20-tetraphenylporphyrin; Ti(O-t-Bu) In chloroform-d1 at 0℃; for 28h; Product distribution; Irradiation; other catalysts; | A 43 % Spectr. B 23 % Spectr. C 16 % Spectr. D 18 % Spectr. |
| With oxygen; 5,15,10,20-tetraphenylporphyrin; Ti(O-t-Bu) In chloroform-d1 at 0℃; for 28h; Irradiation; | A 43 % Spectr. B 23 % Spectr. C 16 % Spectr. D 18 % Spectr. |

-
-
80-56-8, 177698-18-9
Pinene

-
A
-
16812-40-1
pinocarvone

-
-
33081-46-8
6,6-Dimethylspirobicyclo<3.1.1>heptan-3-ol-2,2'-oxirane

-
C
-
28664-04-2
3-hydroxy-6,6-dimethylbicyclo[3.1.1]heptan-2-one

-
D
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; 5,15,10,20-tetraphenylporphyrin; titanium(IV) isopropylate In dichloromethane at 0℃; for 20h; Irradiation; | A 5 % Spectr. B 81 % Spectr. C 9 % Spectr. D 3 % Spectr. |

-
-
80-56-8, 177698-18-9
Pinene

-
A
-
16812-40-1
pinocarvone

-
-
33081-46-8
6,6-Dimethylspirobicyclo<3.1.1>heptan-3-ol-2,2'-oxirane

-
C
-
28664-04-2
3-hydroxy-6,6-dimethylbicyclo[3.1.1]heptan-2-one

-
-
32162-27-9
(1α,2β,4β,6α)-2,2,7-trimethyl-3-oxatricyclo[4.1.1.02,4]octane

-
E
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; thiamine diphosphate; bis(acetylacetonate)oxovanadium In dichloromethane at 0℃; Product distribution; Irradiation; other catalysts; | A 8 % Spectr. B 46 % Spectr. C 28 % Spectr. D 10 % Spectr. E 8 % Spectr. |

-
-
58434-29-0
myrtenyl hydroperoxide

-
A
-
16812-40-1
pinocarvone


-
C
-
515-00-4
myrtenol

-
D
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| In hexane at 40℃; for 3528h; Product distribution; isomerization reaction; |

-
-
22321-84-2, 51131-99-8, 51547-39-8, 75992-91-5, 129263-38-3
trans-pinocarveyl hydroperoxide

-
A
-
16812-40-1
pinocarvone


-
C
-
58434-29-0
myrtenyl hydroperoxide

-
D
-
515-00-4
myrtenol

-
E
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With copper acetylacetonate Product distribution; Ambient temperature; other metal compounds; other hydroperoxide; |

-
-
515-00-4
myrtenol

-
A
-
19250-17-0
myrtenic acid

-
B
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With chlorine dioxide In tetrachloromethane at 50℃; for 0.75h; | A 4 % Chromat. B 41 % Chromat. |

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| d-myrtenal; |

-
-
80-56-8, 177698-18-9
Pinene

-
A
-
4501-58-0, 23727-15-3, 91200-86-1, 91819-58-8
campholenyc aldehyde

-
B
-
1686-14-2
α-pinene epoxide

-
C
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With 9,10-Dicyanoanthracene; oxygen In various solvent(s) Product distribution; Further Variations:; Reagents; Solvents; Oxidation; Irradiation; |

-
-
80-56-8, 177698-18-9
Pinene

-
A
-
4501-58-0, 23727-15-3, 91200-86-1, 91819-58-8
campholenyc aldehyde


-
C
-
1686-14-2
α-pinene epoxide

-
D
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With 9,10-Dicyanoanthracene; oxygen In acetonitrile Product distribution; Further Variations:; Solvents; Oxidation; Irradiation; |

-
-
177698-19-0
Beta-pinene

-
A
-
16812-40-1
pinocarvone

-
-
1674-08-4
3-hydroxy-β-pinene

-
C
-
515-00-4
myrtenol

-
D
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; Co-Fe3O4 at 60℃; under 760.051 Torr; for 7h; Product distribution; Further Variations:; Catalysts; |

-
-
177698-19-0
Beta-pinene


-
B
-
515-00-4
myrtenol

-
C
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With 2,6-dimethylpyridine; oxygen; rose bengal; acetic anhydride In methanol at 20℃; for 4h; UV-irradiation; |

-
-
177698-19-0
Beta-pinene

-
A
-
515-00-4
myrtenol

-
B
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; rose bengal In N,N-dimethyl-formamide at 20℃; for 4h; UV-irradiation; |
-
-
177698-19-0
Beta-pinene

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; rose bengal; acetic anhydride In methanol at 20℃; for 4h; UV-irradiation; |
-
-
80-56-8, 177698-18-9
Pinene

-
A
-
80-57-9
verbenone


-
C
-
515-00-4
myrtenol

-
D
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen; rose bengal In N,N-dimethyl-formamide at 20℃; for 4h; UV-irradiation; | |
| With dihydrogen peroxide In acetonitrile at 60℃; for 24h; Temperature; | A 41 %Chromat. B 13 %Chromat. C 17 %Chromat. D 11 %Chromat. |

-
-
80-56-8, 177698-18-9
Pinene

-
A
-
80-57-9
verbenone


-
C
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With 2,6-dimethylpyridine; oxygen; rose bengal; acetic anhydride In methanol at 20℃; for 4h; UV-irradiation; |

-
-
177698-19-0
Beta-pinene

-
A
-
16812-40-1
pinocarvone

-
C
-
515-00-4
myrtenol

-
D
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen at 60℃; under 760.051 Torr; for 6h; Neat (no solvent); |

-
-
80-56-8, 177698-18-9
Pinene

-
A
-
80-57-9
verbenone


-
C
-
16812-40-1
pinocarvone

-
E
-
1686-14-2
α-pinene epoxide

-
F
-
515-00-4
myrtenol

-
G
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen at 60℃; under 760.051 Torr; for 6h; Neat (no solvent); |

-
-
80-56-8, 177698-18-9
Pinene

-
A
-
80-57-9
verbenone


-
C
-
16812-40-1
pinocarvone

-
E
-
515-00-4
myrtenol

-
F
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With oxygen at 60℃; under 3800.26 Torr; for 3.5h; Neat (no solvent); Autoclave; |

-
-
80-56-8, 177698-18-9
Pinene

-
A
-
16812-40-1
pinocarvone


-
C
-
22321-84-2
pinocarveyl hydroperoxide

-
D
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With 6,7-bis[3-(Nε-tert-butyloxycarbonyllysine methyl ester)]-1,3,5,8-tetramethyl-2,4-divinylporphyrin immobilized in silica gel In chloroform at 20℃; for 30h; Quantum yield; Time; Irradiation; |

-
-
80-56-8, 177698-18-9
Pinene

-
A
-
16812-40-1
pinocarvone

-
C
-
22321-84-2
pinocarveyl hydroperoxide

-
D
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With chlorophyll extract of fresh spinach leaves immobilized in silica gel In chloroform at 20℃; Reagent/catalyst; Irradiation; |

-
-
130827-43-9
(1S,2S)-1-phenyl-N2-methyl-1,2-propandiamine

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
-
130827-39-3, 130857-87-3, 130857-92-0
(1R,2R,3R,4S,5S)-2-(6,6-Dimethyl-bicyclo[3.1.1]hept-2-en-2-yl)-1,5-dimethyl-4-phenyl-imidazolidine

| Conditions | Yield |
|---|---|
| In chloroform for 24h; Ambient temperature; | 100% |
-
-
130857-96-4
1R,2S-N,N-dimethyl-2-phenyl-3-methyl-1,3-diaminoethane

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
-
130827-41-7, 130857-89-5, 130857-95-3
(1S,2S,3R,4S,5R)-2-(6,6-Dimethyl-bicyclo[3.1.1]hept-2-en-2-yl)-1,3,4-trimethyl-5-phenyl-imidazolidine

| Conditions | Yield |
|---|---|
| In chloroform for 24h; Ambient temperature; | 100% |
-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
-
4764-14-1
6,6-dimethylbicyclo<3.3.1>heptane-2-carboxaldehyde

| Conditions | Yield |
|---|---|
| With hydrogen; SC-1 Ni2B In methanol at 25℃; under 760 Torr; for 24h; | 96% |
| With hydrido(triphenylphosphine)copper(I) hexamer; phenylsilane In toluene for 5h; Ambient temperature; | 80% |
-
-
67-64-1
acetone

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
-
56933-99-4
4-(6,6-dimethylbicyclo[3,1,1]hept-2-en-2-yl)-2-butanone

| Conditions | Yield |
|---|---|
| With Pd-MgO; hydrogen at 75℃; under 3750.38 Torr; for 3h; | 95% |
-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
-
149-73-5
trimethyl orthoformate

-
-
1263208-49-6
2,2-dimethoxymethyl-6,6-dimethyl-bicyclo[3.1.1]hept-2-ene

| Conditions | Yield |
|---|---|
| With 1-ethyl-3-methylimidazolium hydrogensulfate In methanol at 5 - 20℃; for 2h; | 93% |

| Conditions | Yield |
|---|---|
| With ethylenediamine diacetic acid In neat (no solvent) at 20℃; for 0.5h; Knoevenagel Condensation; Green chemistry; | 93% |
-
-
676-58-4
methylmagnesium chloride

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
-
33829-99-1
1-(6,6-dimethyl-bicyclo[3.1.1]hept-2-en-2-yl)-ethanol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 12h; Ambient temperature; | 92% |

| Conditions | Yield |
|---|---|
| With pyrrolidine In methanol for 5h; Inert atmosphere; Schlenk technique; | 90% |
-
-
52509-14-5
(1,3-dioxolan-2-yl-methyl)triphenylphosphonium bromide

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
-
124931-24-4
(E)-3-(6,6-dimethyl-bicyclo[3.1.1]hept-2-en-2-yl)prop-2-enal

| Conditions | Yield |
|---|---|
| Stage #1: (1,3-dioxolan-2-yl-methyl)triphenylphosphonium bromide With potassium tert-butylate In tetrahydrofuran at 0℃; for 0.5h; Sealed tube; Inert atmosphere; Stage #2: 6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde In tetrahydrofuran at 20℃; for 6h; Sealed tube; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| With lithium pyrrolidinoborohydride at 25℃; | 89% |
| With LiPyrrBH3 In tetrahydrofuran at 25℃; for 3h; | 89% |
| With sodium triphenyl boronhydride In tetrahydrofuran at 0℃; for 1h; | 100 % Chromat. |
| With formic acid; iron(II) tetrafluoroborate hexahydrate; tris(2-diphenylphosphinoethyl)phosphine In tetrahydrofuran at 60℃; for 2h; Schlenk technique; Inert atmosphere; | 99 %Chromat. |
-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
-
22339-11-3
α-pinenyl bromide

| Conditions | Yield |
|---|---|
| Stage #1: 6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde With sodium tetrahydroborate In tetrahydrofuran at 20℃; for 8h; Stage #2: With carbon tetrabromide; triphenylphosphine In tetrahydrofuran; dichloromethane at 0℃; for 0.5h; | 84% |
-
-
1123-49-5
3,5-dimethyl-4-nitroisoxazole

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With piperidine In ethanol at 65℃; for 2h; | 81% |

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
-
884536-72-5
(RR,RS)-5-cyano-2-(6',6'-dimethylbicyclo[3.1.1]hept-2'-en-2'-yl)-6-phenylamino-2,3-dihydro-1,3-thiazin-4(1H)-one

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In ethanol for 2h; Heating; | 80% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol at 85℃; | 80% |

| Conditions | Yield |
|---|---|
| With sodium acetate on alumina for 0.0333333h; microwave irradiation; | 76% |
-
-
75-05-8
acetonitrile

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
-
324538-16-1
C12H17NO

| Conditions | Yield |
|---|---|
| With [2,6-(iPr2PO)2C6H3]NiCH2CN at 20℃; for 24h; Inert atmosphere; | 73% |
-
-
130827-42-8
(1R,2S)-1-phenyl-N2-methyl-1,2-propandiamine

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
A
-
130827-39-3, 130857-87-3, 130857-92-0
(1R,2R,3S,4R,5S)-2-(6,6-Dimethyl-bicyclo[3.1.1]hept-2-en-2-yl)-1,5-dimethyl-4-phenyl-imidazolidine

-
B
-
130827-39-3, 130857-87-3, 130857-92-0
(1S,2S,3S,4R,5S)-2-(6,6-Dimethyl-bicyclo[3.1.1]hept-2-en-2-yl)-1,5-dimethyl-4-phenyl-imidazolidine

| Conditions | Yield |
|---|---|
| In chloroform for 24h; Ambient temperature; | A 71% B 12% |

-
-
1875-48-5
N-aminophthalamide

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
A
-
1445-69-8
2,3-dihydrophthalazine-1,4-dione

-
B
-
121597-24-8
myrtenal phthaloylhydrazone

| Conditions | Yield |
|---|---|
| In ethanol for 2.5h; Heating; | A n/a B 67% |

-
-
64-18-6
formic acid

-
-
931-53-3
Cyclohexyl isocyanide

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| In dichloromethane at 0 - 20℃; for 48h; Passerini Condensation; Inert atmosphere; | 66% |

-
-
64-18-6
formic acid

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| In dichloromethane at 0 - 20℃; for 48h; Passerini Condensation; Inert atmosphere; | 64% |


-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: N-Boc-(tri-n-butylstannyl)dimethylamine With n-butyllithium In tetrahydrofuran at -78℃; for 0.283333h; Metallation; Stage #2: In tetrahydrofuran at -78 - -53℃; for 0.416667h; Transmetallation; Stage #3: 6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde With chloro-trimethyl-silane In tetrahydrofuran at -78 - 20℃; Substitution; | 62% |
-
-
130857-97-5
(1S,2S)-1-phenyl-N1,N2-dimethyl-1,2-propandiamine

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
A
-
130827-41-7, 130857-89-5, 130857-95-3
(1R,2R,3R,4S,5S)-2-(6,6-Dimethyl-bicyclo[3.1.1]hept-2-en-2-yl)-1,3,4-trimethyl-5-phenyl-imidazolidine

-
B
-
130827-41-7, 130857-89-5, 130857-95-3
(1R,2S,3R,4S,5S)-2-(6,6-Dimethyl-bicyclo[3.1.1]hept-2-en-2-yl)-1,3,4-trimethyl-5-phenyl-imidazolidine

| Conditions | Yield |
|---|---|
| In chloroform for 24h; Ambient temperature; | A 60% B 40% |

-
-
109-80-8
1.3-propanedithiol

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

-
-
69460-06-6
2-(6,6-dimethyl-bicyclo[3.1.1]hept-2-en-2-yl)-[1,3]dithiane

| Conditions | Yield |
|---|---|
| With TAFF In benzene for 3h; Heating; | 60% |
-
-
934-06-5
(S)-5-oxopyrrolidine-2-carbohydrazide

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In water at 20℃; for 1h; | 60% |
-
-
64-18-6
formic acid

-
-
39546-47-9
p-chlorobenzylisocyanide

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| In dichloromethane at 0 - 20℃; for 48h; Passerini Condensation; Inert atmosphere; | 57% |

-
-
51054-41-2
6,7-dimethoxy-1-cyanomethylene-1,2,3,4-tetrahydroisoquinoline

-
-
564-94-3
6,6-dimethylbicyclo[3.1.1]hept-2-ene-2-carbaldehyde

| Conditions | Yield |
|---|---|
| With cerium(III) chloride In acetonitrile at 20℃; for 6h; | 52% |
Myrtenal Chemical Properties
Molecular Structure of Myrtenal (CAS NO.564-94-3):
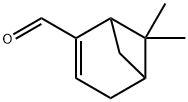
EINECS: 209-274-8
IUPAC Name: 7,7-Dimethylbicyclo[3.1.1]hept-3-ene-4-carbaldehyde
Molecular Formula: C10H14O
Molecular Weight: 150.217560 g/mol
XLogP3-AA: 2.1
H-Bond Donor: 0
H-Bond Acceptor: 1
Canonical SMILES: CC1(C2CC=C(C1C2)C=O)C
InChI: InChI=1S/C10H14O/c1-10(2)8-4-3-7(6-11)9(10)5-8/h3,6,8-9H,4-5H2,1-2H3
InChIKey: KMRMUZKLFIEVAO-UHFFFAOYSA-N
Index of Refraction: 1.561
Molar Refractivity: 45.86 cm3
Molar Volume: 141.4 cm3
Surface Tension: 40.2 dyne/cm
Density: 1.061 g/cm3
Flash Point: 78.9 °C
Enthalpy of Vaporization: 45.21 kJ/mol
Boiling Point: 215.7 °C at 760 mmHg
Vapour Pressure: 0.145 mmHg at 25 °C
Water Solubility: 215.9 mg/L at 25 °C
Refractive Index: n20/D 1.504
Storage Temp.: 2-8 °C
Myrtenal Toxicity Data With Reference
| 1. | orl-rat LD50:2300 mg/kg | FCTOD7 Food and Chemical Toxicology. 26 (1988),329. | ||
| 2. | ivn-mus LD50:170 mg/kg | FCTOD7 Food and Chemical Toxicology. 26 (1988),329. | ||
| 3. | skn-rbt LD50:>5 g/kg | FCTOD7 Food and Chemical Toxicology. 26 (1988),329. |
Myrtenal Consensus Reports
Reported in EPA TSCA Inventory.
Myrtenal Safety Profile
Safety Information of Myrtenal (CAS NO.564-94-3):
Safety Statements: 23-24/25
S23:Do not breathe vapour
S24/25:Avoid contact with skin and eyes
WGK Germany: 2
RTECS: DT5180000
F: 10-23
Poison by intravenous route. Moderately toxic by ingestion. Slightly toxic by skin contact. When heated to decomposition it emits acrid smoke and irritating vapors.
Myrtenal Specification
Myrtenal with CAS Registry Number of 564-94-3 is clear colourless to yellow liquid, also known as 2-Formyl-6,6-dimethylbicyclo(3.1.1)hept-2-ene ; 2-Norpinene-2-carboxaldehyde, 6,6-dimethyl- ; 6,6-Dimethyl-2-norpinene-2-carboxaldehyde ; 6,6-Dimethylbicyclo(3.1.1)hept-2-ene-2-carboxaldehyde ; Benihinal ; Bicyclo(3.1.1)hept-2-ene-2-carboxaldehyde, 6,6-dimethyl- ; FEMA No. 3395 ; NSC 54384 ; 2-Norpinene-2-carboxaldehyde, 6,6-dimethyl- (8CI) ; Pin-2-ene-1-carbaldehyde . Myrtenal is a kind of spice and can be used for synthesis of more valuable spices such as basil Draba .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View