-
Name
N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine
- EINECS 212-344-0
- CAS No. 793-24-8
- Article Data19
- CAS DataBase
- Density 1.039 g/cm3
- Solubility <0.1 g/100 mL at 17 °C in water
- Melting Point 45-46 °C
- Formula C18H24N2
- Boiling Point 412.3 °C at 760 mmHg
- Molecular Weight 268.402
- Flash Point 245.8 °C
- Transport Information
- Appearance Purple flakes
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 1,4-Benzenediamine,N-(1,3-dimethylbutyl)-N'-phenyl- (9CI);p-Phenylenediamine,N-(1,3-dimethylbutyl)-N'-phenyl- (7CI,8CI);4-(1,3-Dimethylbutylamino)diphenylamine;6PPD;Antage 6C;Antozite 67;CD 13;CI;DBDA;DMBPD;Diafen FDMB;Flexzone 7F;Flexzone 7P;Forte 6C;N-(1,3-Dimethylbutyl)-N'-phenyl-1,4-benzenediamine;N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine;N-Phenyl-N'-(1,3-dimethylbutyl)-1,4-phenylenediamine;Nocrac 6C;Ozonone 6C;Permanax 120;Santoflex 13F;UOP 562;UOP 588;Vulkanox 4020;Vulkanox 4020LG;Wingstay 300;
- PSA 24.06000
- LogP 5.42270
Synthetic route
-
-
108-10-1
4-methyl-2-pentanone

-
-
101-54-2
N-phenylphenylene-1,4-diamine

-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

| Conditions | Yield |
|---|---|
| With hydrogen at 99.84℃; under 18751.9 Torr; for 4h; Catalytic behavior; Reagent/catalyst; Time; Autoclave; | 100% |
| With hydrogen; pyrographite at 20 - 150℃; under 22052.2 Torr; for 0.1h; Autoclave; | 99.4% |
| With platinum on activated charcoal; isopropyl alcohol at 50 - 150℃; under 36775.4 Torr; Hydrogenation; |
-
-
63302-40-9
C18H22N2

-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen In 5,5-dimethyl-1,3-cyclohexadiene at 100℃; under 7500.75 Torr; Solvent; Temperature; | 92.6% |
-
-
156-10-5
p-nitrosodiphenylamine

-
-
108-10-1
4-methyl-2-pentanone

-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

| Conditions | Yield |
|---|---|
| With platinum on activated charcoal; isopropyl alcohol at 50 - 150℃; under 25742.8 Torr; Hydrogenation; |
-
-
836-30-6
4-ntrophenyl(phenyl)amine

-
-
108-10-1
4-methyl-2-pentanone

-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

| Conditions | Yield |
|---|---|
| With platinum on activated charcoal; isopropyl alcohol at 50 - 150℃; under 25742.8 Torr; Hydrogenation; |

-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

| Conditions | Yield |
|---|---|
| With formic acid; palladium on activated charcoal at 85℃; Yield given; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 5percent Pd/C 2: 5percent palladium-on-carbon / 85 °C 3: HCOOH / 5percent Pd/C / 85 °C View Scheme | |
| Multi-step reaction with 3 steps 1: formic acid / 5percent Pd/C / 85 °C 2: 5percent palladium-on-carbon / 85 °C 3: HCOOH / 5percent Pd/C / 85 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 5percent palladium-on-carbon / 85 °C 2: HCOOH / 5percent Pd/C / 85 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 5percent palladium-on-carbon / 85 °C 2: HCOOH / 5percent Pd/C / 85 °C View Scheme |
-
-
15761-50-9, 34017-11-3, 54548-48-0, 108-09-8
2-amino-4-methylpentane

-
-
2406-04-4
4-(phenylimino)cyclohexa-2,5-dienone

-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

| Conditions | Yield |
|---|---|
| In methanol |
-
-
108-10-1
4-methyl-2-pentanone

-
-
101-54-2
N-phenylphenylene-1,4-diamine

-
A
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

-
B
-
63302-40-9
C18H22N2

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen at 200℃; under 30003 Torr; Reagent/catalyst; | |
| With hydrogen at 160℃; under 22502.3 Torr; for 4h; Autoclave; |

-
-
108-10-1
4-methyl-2-pentanone

-
-
101-54-2
N-phenylphenylene-1,4-diamine

-
A
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

-
B
-
62-53-3
aniline

| Conditions | Yield |
|---|---|
| With 10% Pd/C; hydrogen at 160℃; under 22502.3 Torr; for 4h; Autoclave; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: hydrogen 2: pyrographite; ; hydrogen / 0.1 h / 20 - 150 °C / 22052.2 Torr / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: sulfuric acid; nitric acid / chloroform; benzene / 5 h / 50 °C / Industrial scale 2: tetramethyl ammoniumhydroxide; platinum on carbon; hydrogen / water / 80 °C / 11251.1 Torr 3: pyrographite; ; hydrogen / 0.1 h / 20 - 150 °C / 22052.2 Torr / Autoclave View Scheme | |
| Multi-step reaction with 4 steps 1: sulfuric acid; nitric acid / chloroform; benzene / 5 h / 50 °C / Industrial scale 2: hydrogen / 200 °C / Industrial scale 3: tetramethyl ammoniumhydroxide; platinum on carbon; hydrogen / water / 80 °C / 11251.1 Torr 4: pyrographite; ; hydrogen / 0.1 h / 20 - 150 °C / 22052.2 Torr / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: ammonia / 450 °C / 760.05 Torr / Industrial scale 2: tetramethyl ammoniumhydroxide; platinum on carbon; hydrogen / water / 80 °C / 11251.1 Torr 3: pyrographite; ; hydrogen / 0.1 h / 20 - 150 °C / 22052.2 Torr / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: hydrogen / 200 °C / Industrial scale 2: tetramethyl ammoniumhydroxide; platinum on carbon; hydrogen / water / 80 °C / 11251.1 Torr 3: pyrographite; ; hydrogen / 0.1 h / 20 - 150 °C / 22052.2 Torr / Autoclave View Scheme | |
| Multi-step reaction with 3 steps 1: tetramethyl ammoniumhydroxide / water / 2.1 h / 75 °C / 55 Torr / Dean-Stark 2: hydrogen; 5%-palladium/activated carbon / 0.5 h / 80 °C / 11251.1 Torr / Autoclave 3: 3 % platinum on carbon; hydrogen / 0.5 h / 155 °C / 26252.6 Torr / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: tetramethyl ammoniumhydroxide / water / 2.1 h / 75 °C / 55 Torr / Dean-Stark 2: hydrogen; 5%-palladium/activated carbon / 0.5 h / 80 °C / 11251.1 Torr / Autoclave 3: 3 % platinum on carbon; hydrogen / 0.5 h / 155 °C / 26252.6 Torr / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran at 20℃; for 48h; Cooling with ice; | 86% |

| Conditions | Yield |
|---|---|
| Stage #1: N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine With magnesium sulfate; silver(l) oxide In toluene at 25℃; for 15h; Stage #2: prop-2-ene-1-thiol In ethanol at 25℃; | 65% |
-
-
74-85-1
ethene

-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

-
A
-
944727-84-8
C22H32N2

-
C
-
944727-85-9
C24H36N2

| Conditions | Yield |
|---|---|
| diethylaluminium chloride In hexane for 12 - 16h; Product distribution / selectivity; | A 47.2% B n/a C n/a |


| Conditions | Yield |
|---|---|
| Stage #1: Adipic acid With thionyl chloride; N,N-dimethyl-formamide In tetrahydrofuran; chloroform for 8h; Reflux; Stage #2: N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine With pyridine In tetrahydrofuran at 20℃; Cooling with ice; | 39% |
-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: Ag2O; MgSO4 / toluene / 15 h / 25 °C 1.2: 65 percent / ethanol / 25 °C 2.1: 75 percent / 150 - 155 °C View Scheme |
-
-
463-71-8
thiophosgene

-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

-
-
584-08-7
potassium carbonate

-
-
118745-60-1
N-(4-anilinophenyl)-N-(1,3-dimethyl-butyl)-thiocarbamate-O-methyl ester

| Conditions | Yield |
|---|---|
| In methanol; chloroform |

-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

-
-
74-88-4
methyl iodide

-
-
118745-59-8
N-(4-anilinophenyl)-N-(1,3-dimethyl-butyl)-dithiocarbamate methyl ester

| Conditions | Yield |
|---|---|
| With carbon disulfide; sodium hydroxide In tetrahydrofuran |

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; aluminum (III) chloride at 160 - 170℃; for 27h; Friedel Crafts Alkylation; |
-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

-
-
20170-32-5
4-hydroxy-3,5-di-tert-butylphenylpropionic acid

| Conditions | Yield |
|---|---|
| With boric acid In toluene for 15h; Dean-Stark; Inert atmosphere; Reflux; |
-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

-
-
6386-38-5
methyl 3-(4-hydroxy-3,5-di-tert-butyl)phenylpropanoate

| Conditions | Yield |
|---|---|
| With boric acid In toluene for 15h; Dean-Stark; Inert atmosphere; Reflux; Molecular sieve; |
-
-
108-77-0
1,3,5-trichloro-2,4,6-triazine

-
-
36294-23-2
4-(3-hydroxypropyl)-2,6-di-tert-butylphenol

-
-
793-24-8
N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine

| Conditions | Yield |
|---|---|
| Stage #1: 1,3,5-trichloro-2,4,6-triazine; 4-(3-hydroxypropyl)-2,6-di-tert-butylphenol With triethylamine In tetrahydrofuran at 0℃; for 4h; Inert atmosphere; Stage #2: N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine In tetrahydrofuran at 65℃; for 15h; Inert atmosphere; |

N-(1,3-DIMETHYLBUTYL)-N'-PHENYL-P-PHENYLENEDIAMINE Chemical Properties
IUPAC Name: 1-N-(4-methylpentan-2-yl)-4-N-Phenylbenzene-1,4-diamine
Synonyms:1,4-Benzenediamine, N~1~-(1,3-dimethylbutyl)-N~4~-phenyl- ; 1,4-Benzenediamine, N~1~-(1,3-dimethylbutyl)-N~4~-phenyl- ; N-(1,3-dimethylbutyl)-N'-Phenylbenzene-1,4-diamine ; N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine ; N-(4-Methyl-2-pentanyl)-N'-phenyl-1,4-benzenediamine
Product Categories: Industrial/Fine Chemicals;Antidegradants
Molecular Structure : 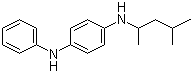
Molecular Formula: C18H24N2
Molecular Weight : 268.40
CAS NO: 793-24-8
EINECS: 212-344-0
Mol File: 793-24-8.mol
Melting point: 45-46 ºC
Boiling point: 260 ºC
Flash point: 204 ºC
Density: 1.039 g/cm3
Appearance:Purple flakes
Index of Refraction: 1.599
Surface Tension: 40.9 dyne/cm
Enthalpy of Vaporization: 66.49 kJ/mol
Vapour Pressure: 5.22E-07 mmHg at 25°C
Water solubility <0.1 g/100 mL at 17 ºC
Appearance: N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine (CAS NO. 793-24-8) is gray-black solid
N-(1,3-DIMETHYLBUTYL)-N'-PHENYL-P-PHENYLENEDIAMINE Uses
1.N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine (CAS NO. 793-24-8) is used for anti-ozone agents and antioxidants for natural rubber and synthetic rubber.
2.N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine (CAS NO. 793-24-8) is used for fine effectiveness on the protection of ozone and flex fatigue cracking .
N-(1,3-DIMETHYLBUTYL)-N'-PHENYL-P-PHENYLENEDIAMINE Production
Using the 4 - amino-2-aniline and methyl isobutyl ketone, catalytic,with hydrogenation condensation, Then, N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine (CAS NO. 793-24-8) is derived.
N-(1,3-DIMETHYLBUTYL)-N'-PHENYL-P-PHENYLENEDIAMINE Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rabbit | LD50 | skin | > 7940mg/kg (7940mg/kg) | BEHAVIORAL: FOOD INTAKE (ANIMAL) BEHAVIORAL: CHANGES IN MOTOR ACTIVITY (SPECIFIC ASSAY) | Acute Toxicity Data. Journal of the American College of Toxicology, Part B. Vol. 1, Pg. 67, 1990. |
| rat | LD50 | oral | 3580mg/kg (3580mg/kg) | SENSE ORGANS AND SPECIAL SENSES: OTHER: EYE BEHAVIORAL: FOOD INTAKE (ANIMAL) GASTROINTESTINAL: "HYPERMOTILITY, DIARRHEA" | Acute Toxicity Data. Journal of the American College of Toxicology, Part B. Vol. 1, Pg. 67, 1990. |
N-(1,3-DIMETHYLBUTYL)-N'-PHENYL-P-PHENYLENEDIAMINE Safety Profile
RTECS ST0900000
N-(1,3-DIMETHYLBUTYL)-N'-PHENYL-P-PHENYLENEDIAMINE Specification
1.Air & Water Reactions :N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine may be sensitive to prolonged exposure to air. Insoluble in water.
2.Reactivity Profile :You should protect N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine (CAS NO. 793-24-8) from exposure to strong oxidizing agents such as chlorine or hydrogen peroxide. N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine will ignite if heated strongly.
3.Fire Hazard: N-(1,3-Dimethylbutyl)-N'-phenyl-p-phenylenediamine is combustible.
Related Products
- N-(1-(o-Chlorophenoxy)-2-propyl)-2-(diethylamino)-N-ethylacetamide hydrochloride
- N-(1-(o-Chlorophenoxy)-2-propyl)-2-(diethylamino)-N-methylacetamide hydrochloride
- N-(1-(Phenylacetyl)-L-prolyl)glycine ethyl ester
- N-(1,2-Dimethylpropyl)-2-pyridinamine
- N-(1,3-DIMETHYLBUTYL)-N'-PHENYL-P-PHENYLENEDIAMINE
- N-(1,3-Dimethylbutyl)-N'-phenyl-p-quinone diimine
- N-(1,4-Dimethylpentyl)-n'-phenyl-1,4-benzenediamine
- N-(1-adamantyl)-1-pentylindole-3-carboxamide
- N-(1-Adamantyl)acetamide
- N-(1-Adamantylmethyl)-2-mercaptoacetamidine hydrochloride
- 79326-88-8
- 79326-93-5
- 79-33-4
- 79338-14-0
- 79-34-5
- 79347-94-7
- 79348-71-3
- 79349-23-8
- 79349-53-4
- 79349-67-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View