-
Name
4,4'-Iminodianiline
- EINECS 208-673-4
- CAS No. 537-65-5
- Article Data30
- CAS DataBase
- Density 1.245g/cm3
- Solubility
- Melting Point 158°C
- Formula C12H13 N3
- Boiling Point 410.7°Cat760mmHg
- Molecular Weight 199.255
- Flash Point 239.2°C
- Transport Information
- Appearance
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 1,4-Benzenediamine,N-(4-aminophenyl)- (9CI); Diphenylamine, 4,4'-diamino- (6CI,7CI,8CI);4,4'-Diaminodiphenylamine; 4,4'-Iminodianiline; Benzenamine, 4,4'-iminobis-;Bis(4-aminophenyl)amine; Bis(p-aminophenyl)amine; C.I. 76120;Di(p-aminophenyl)amine; Diazol Black C; Indamine;N-(4-Aminophenyl)-1,4-benzenediamine; N-(4-Aminophenyl)-p-phenylenediamine; NSC33417; p,p'-Diaminodiphenylamine; p,p'-Iminodianiline
- PSA 147.05000
- LogP 4.25800
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen; nickel In ethanol at 60℃; under 30003 Torr; for 2h; Reagent/catalyst; Temperature; Autoclave; | 93.2% |
| With tin(II)chloride dihydrate; ethanol at 70℃; for 1h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With acetic acid; zinc | |
| With hydrogenchloride; tin | |
| With sodium sulfide at 140 - 150℃; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin(ll) chloride |

-
-
537-65-5
4,4'-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| With iron |
-
-
106038-75-9
N-acetyl-4,4'-dinitrodiphenylamine

-
-
537-65-5
4,4'-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| With iron |
-
-
142-04-1
aniline hydrochloride

-
-
540-24-9
p-phenylenediamine hydrochloride

-
-
537-65-5
4,4'-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| With potassium dichromate und anschliessenden Reduktion des Oxydationsprodukts (Indamin) mit Zink und Salzsaeure in der Waerme; |
-
-
142-04-1
aniline hydrochloride

-
-
60-09-3
aniline yellow

-
-
62-53-3
aniline

-
-
537-65-5
4,4'-diaminodiphenylamine

-
-
540-24-9
p-phenylenediamine hydrochloride

-
-
106-50-3
1,4-phenylenediamine

-
-
537-65-5
4,4'-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| With water at 200℃; |

| Conditions | Yield |
|---|---|
| With hydrochloride of tin dichloride |

| Conditions | Yield |
|---|---|
| at 200℃; |


| Conditions | Yield |
|---|---|
| anschl. mit Zink-Staub und HCl; | |
| und Behandeln mit Zinkstaub und Salzsaeure; |


| Conditions | Yield |
|---|---|
| dann mit verd. Schwefelsaeure; |


| Conditions | Yield |
|---|---|
| With hydrogenchloride; tin | |
| With phosphorus; hydrogen iodide |
-
-
7647-01-0
hydrogenchloride

-
-
4128-71-6
N-[4-(phenylazo)phenyl]-acetamide

-
A
-
537-65-5
4,4'-diaminodiphenylamine

-
B
-
108272-17-9
N-acetyl-4,4'-diaminodiphenylamine

-
C
-
62-53-3
aniline

-
D
-
106-50-3
1,4-phenylenediamine


-
-
537-65-5
4,4'-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| With sulfuric acid; sulfur dioxide; zinc |

-
-
537-65-5
4,4'-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| With sulfuric acid |
-
-
7647-01-0
hydrogenchloride

-
-
64-17-5
ethanol

-
-
3313-75-5
Bis-(4-nitro-phenyl)-oxy-aminyl

-
-
537-65-5
4,4'-diaminodiphenylamine

-
-
7647-01-0
hydrogenchloride

-
-
108272-17-9
N-acetyl-4,4'-diaminodiphenylamine

-
-
537-65-5
4,4'-diaminodiphenylamine
-
-
7664-93-9
sulfuric acid

-
-
17867-12-8
bis-(4-benzylidenamino-phenyl)-amine

-
A
-
537-65-5
4,4'-diaminodiphenylamine

-
B
-
100-52-7
benzaldehyde

-
-
60-29-7
diethyl ether

-
-
861362-54-1
N,N-bis-(4-nitro-phenyl)-o-trityl-hydroxylamine

-
A
-
76-84-6
triphenylmethyl alcohol

-
B
-
537-65-5
4,4'-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| Hydrogenation; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: alcohol; glacial acetic acid; isopentyl nitrite; nitric acid / Man laesst 24 Stdn. stehen und behandelt das erhaltene, von der Mutterlauge getrennte, mit Alkohol und Aether gewaschene Reaktionsprodukt mit einem Gemisch gleicher Teile Alkohol und rauchender Salzsaeure 2: acetic acid; zinc View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: nitrogen dioxide; benzene; diethyl ether 2: tin (II)-chloride; aqueous alcoholic hydrochloric acid View Scheme |

-
-
537-65-5
4,4'-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: diethyl ether 2: nitrogen dioxide; benzene; diethyl ether 3: tin (II)-chloride; aqueous alcoholic hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: diethyl ether 2: nitrogen dioxide; benzene; diethyl ether 3: tin (II)-chloride; aqueous alcoholic hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium acetate / ethyl methyl ether / 7 h / Inert atmosphere; Reflux 2: tin(II)chloride dihydrate; ethanol / 1 h / 70 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: sodium hydroxide / dimethyl sulfoxide / 12 h / 80 °C 2: hydrogenchloride; tin / water / 18 h / Inert atmosphere; Reflux View Scheme | |
| Multi-step reaction with 2 steps 1.1: sodium hydroxide / dimethyl sulfoxide / 12 h / 80 °C 2.1: hydrogenchloride; tin / water / 18 h / Reflux; Inert atmosphere 2.2: pH 12 / Inert atmosphere View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium acetate / ethyl methyl ether / 7 h / Inert atmosphere; Reflux 2: tin(II)chloride dihydrate; ethanol / 1 h / 70 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: sodium acetate / ethyl methyl ether / 7 h / Inert atmosphere; Reflux 2: tin(II)chloride dihydrate; ethanol / 1 h / 70 °C / Inert atmosphere View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium acetate / ethyl methyl ether / 7 h / Inert atmosphere; Reflux 2: tin(II)chloride dihydrate; ethanol / 1 h / 70 °C / Inert atmosphere View Scheme |

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 30℃; under 11250900 Torr; for 72h; | 100% |
-
-
537-65-5
4,4'-diaminodiphenylamine

-
-
350-46-9
4-Fluoronitrobenzene

-
-
258269-00-0
N,N’’-bis(4’’-nitrophenyl)-4,4’-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 40℃; under 11250900 Torr; for 72h; | 98.6% |
| With triethylamine In dimethyl sulfoxide | 88% |
| With triethylamine In dimethyl sulfoxide at 90℃; for 72h; Inert atmosphere; | 87% |

| Conditions | Yield |
|---|---|
| With triethylamine In N,N-dimethyl-formamide at 40℃; under 11250900 Torr; for 72h; | 97.5% |

-
-
537-65-5
4,4'-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| Stage #1: 2-chloro-4,6-bis-((2,2,6,6-tetramethyl-piperidin-4-yl)butylamino)-1,3,5-triazine; 4,4'-diaminodiphenylamine In toluene at 85℃; for 5.5h; Stage #2: With sodium hydroxide In toluene at 115℃; for 7h; Solvent; Temperature; Time; | 95.2% |
-
-
537-65-5
4,4'-diaminodiphenylamine

-
-
555-16-8
4-nitrobenzaldehdye

-
-
52167-06-3
4-[(E)-(4-nitrophenyl)methyleneamino]-N-[4-[(E)-(4-nitrophenyl)methyleneamino]phenyl]aniline

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In ethanol at 20℃; for 14h; | 95% |
| With toluene-4-sulfonic acid In ethanol at 20℃; | 95% |
| With ethanol |

| Conditions | Yield |
|---|---|
| With di(tertbutyl)phenylphosphine; potassium tert-butylate; bis(dibenzylideneacetone)-palladium(0) In toluene at 90℃; for 2.5h; Inert atmosphere; | 95% |
| With di(tertbutyl)phenylphosphine; bis(dibenzylideneacetone)-palladium(0); sodium t-butanolate In toluene at 90℃; for 2.5h; Inert atmosphere; | 95% |
-
-
463-71-8
thiophosgene

-
-
537-65-5
4,4'-diaminodiphenylamine

-
-
1048037-23-5
bis(4-isothiocyanatophenyl)amine

| Conditions | Yield |
|---|---|
| In ethanol; water | 86% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In ethanol at 20℃; | 84% |

| Conditions | Yield |
|---|---|
| With di(tertbutyl)phenylphosphine; palladium diacetate; sodium t-butanolate In toluene at 90℃; for 6h; Inert atmosphere; | 83% |
| With di(tertbutyl)phenylphosphine; acetic acid; sodium t-butanolate In toluene at 90℃; for 6h; Inert atmosphere; | 83% |
| With di(tertbutyl)phenylphosphine; palladium diacetate In toluene at 90℃; for 6h; Inert atmosphere; |
-
-
537-65-5
4,4'-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; bis(dibenzylideneacetone)-palladium(0); sodium t-butanolate In toluene at 50℃; for 26h; Inert atmosphere; | 83% |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 20℃; | 83% |

| Conditions | Yield |
|---|---|
| With di(tertbutyl)phenylphosphine; palladium diacetate; sodium t-butanolate In toluene at 100℃; for 3.5h; Inert atmosphere; | 81% |

-
-
537-65-5
4,4'-diaminodiphenylamine

| Conditions | Yield |
|---|---|
| With di(tertbutyl)phenylphosphine; bis(dibenzylideneacetone)-palladium(0); sodium t-butanolate In toluene at 90℃; for 3h; Inert atmosphere; | 80% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In ethanol at 20℃; | 80% |
-
-
2033-24-1
cycl-isopropylidene malonate

-
-
537-65-5
4,4'-diaminodiphenylamine

-
-
149-73-5
trimethyl orthoformate

| Conditions | Yield |
|---|---|
| Stage #1: cycl-isopropylidene malonate; trimethyl orthoformate for 1h; Reflux; Stage #2: 4,4'-diaminodiphenylamine for 2h; Reflux; | 79% |

-
-
537-65-5
4,4'-diaminodiphenylamine

-
-
173300-83-9
2-thioxo-imidazolidine-1,3-dicarboxylic acid di-tert-butyl ester

-
-
292634-05-0
4,4'-bis[1,3-di(tert-butoxycarbonyl)-2-imidazolidinylimino]diphenylamine

| Conditions | Yield |
|---|---|
| With triethylamine; mercury dichloride In N,N-dimethyl-formamide at 0 - 20℃; Substitution; | 76% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In ethanol at 20℃; | 75% |

| Conditions | Yield |
|---|---|
| at 200℃; for 4h; | 68.5% |
-
-
537-65-5
4,4'-diaminodiphenylamine

-
-
145013-05-4
tert-butyl N-({[(tert-butoxy)carbonyl]amino}methanethioyl)carbamate

| Conditions | Yield |
|---|---|
| With triethylamine; mercury dichloride In N,N-dimethyl-formamide at 0 - 20℃; Substitution; | 66% |

| Conditions | Yield |
|---|---|
| With triethylamine In dimethyl sulfoxide at 90℃; for 72h; Inert atmosphere; | 65.8% |
| With triethylamine In dimethyl sulfoxide at 90℃; for 24h; Inert atmosphere; | 0.28 g |
N-(4-Aminophenyl)-1,4-benzenediamine Chemical Properties
IUPAC Name: 1-N-(4-aminophenyl)benzene-1,4-diamine
Empirical Formula: C12H13N3
Molecular Weight: 199.2517g/mol
EINECS: 208-673-4
Structure of 1,4-Benzenediamine,N1-(4-aminophenyl)- (CAS NO.537-65-5):
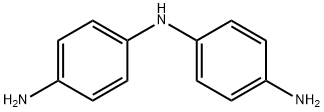
Index of Refraction: 1.733
Molar Refractivity: 64.09 cm3
Molar Volume: 160 cm3
Polarizability: 25.41×10-24cm3
Surface Tension: 63.7 dyne/cm
Density: 1.245 g/cm3
Flash Point: 239.2 °C
Enthalpy of Vaporization: 66.31 kJ/mol
Boiling Point: 410.7 °C at 760 mmHg
Vapour Pressure: 5.89E-07 mmHg at 25°C
Canonical SMILES: C1=CC(=CC=C1N)NC2=CC=C(C=C2)N
InChI: InChI=1S/C12H13N3/c13-9-1-5-11(6-2-9)15-12-7-3-10(14)4-8-12/h1-8,15H,13-14H2
InChIKey: QZHXKQKKEBXYRG-UHFFFAOYSA-N
N-(4-Aminophenyl)-1,4-benzenediamine Specification
1,4-Benzenediamine,N1-(4-aminophenyl)- , its cas register number is 537-65-5. It also can be called 4,4'-Diaminodiphenylamine ; 4,4'-Iminodianiline ; AI3-12116 ; Aniline, 4,4'-iminodi- ; Benzenamine, 4,4'-iminobis- ; Bis(p-aminophenyl)amine ; C.I. 76120 ; CI 76120 ; Di(p-aminophenyl)amine ; Diazol Black C ; Indamine ; NSC 33417 ; p,p'-Diaminodiphenylamine ; p-Phenylenediamine, N-(p-aminophenyl)- .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View