-
Name
Selenic(VI) acid
- EINECS 231-979-4
- CAS No. 7783-08-6
- Article Data138
- CAS DataBase
- Density 1.407g/mLat 25°C
- Solubility
- Melting Point 50 C
- Formula H2O4 Se
- Boiling Point 260 C
- Molecular Weight 144.973
- Flash Point >230°F
- Transport Information UN 3264
- Appearance colourless or white crystals
- Safety Selenium compounds are poisons. A corrosive irritant to skin, eyes, and mucous membranes. When heated to decomposition it emits toxic fumes of Se. See also SELENIUM COMPOUNDS.
- Risk Codes R23/24/25;R34
-
Molecular Structure
-
Hazard Symbols

- Synonyms 7783-08-6
- PSA 74.60000
- LogP -0.97200
Synthetic route

| Conditions | Yield |
|---|---|
| In water byproducts: BaSO4; heating of BaSeO4, H2SO4 and H2O in a crucible for about 2.5 h;; | 85% |
| In water byproducts: BaSO4; heating;; | |
| In water |

| Conditions | Yield |
|---|---|
| In nitromethane dry air atm.; -20-30°C, 2 d - 6 weeks; | A 51% B n/a C n/a |


| Conditions | Yield |
|---|---|
| In water | |
| In water |

-
-
7783-08-6
selenic acid

| Conditions | Yield |
|---|---|
| With bromine In water oxidation of Ag2SeO3 with Br2;; | >99 |
| With bromine In water byproducts: AgBr; addn. of Br2 to a suspension of Ag2SeO3 in H2O under vigorous stirring; passing of a stream of air through the soln. after 2 h;; filtration; passing of air through the filtrate; addn. of HBr; addn. of aq. Br2; filtration; successive concn. of the filtrate in air and in vac.;; | |
| With bromine In water byproducts: AgBr; |

| Conditions | Yield |
|---|---|
| With bromine In water byproducts: AgBr; treatment of a suspension of Ag2SeO3 in H2O with Br2;; | |
| With Br2 In water byproducts: AgBr; treatment of a suspension of Ag2SeO3 in H2O with Br2;; |
-
-
7782-49-2
selenium

-
-
7732-18-5
water

-
-
10028-15-6
ozone

-
A
-
7783-00-8
selenious acid

-
B
-
7783-08-6
selenic acid

| Conditions | Yield |
|---|---|
| In water | A 0% B n/a |
| In water | A 0% B n/a |


| Conditions | Yield |
|---|---|
| In water reaction of ozone and Se in presence of H2O;; | A 0% B n/a |
| In water |


| Conditions | Yield |
|---|---|
| With lead(II) nitrate In neat (no solvent) byproducts: PbS; melting of clausthalite and KNO3; dissolving of the melt in HNO3; boiling; addn. of Pb(NO3)2; decompn. of the formed PbSeO4 with H2S;; | |
| With Pb(NO3)2 In neat (no solvent) byproducts: PbS; melting of clausthalite and KNO3; dissolving of the melt in HNO3; boiling; addn. of Pb(NO3)2; decompn. of the formed PbSeO4 with H2S;; | |
| With Pb(NO3)2 |

-
-
7782-49-2
selenium

-
-
7732-18-5
water

-
-
7782-50-5
chlorine

-
A
-
7783-00-8
selenious acid

-
B
-
7783-08-6
selenic acid

| Conditions | Yield |
|---|---|
| In water | |
| In water |


-
-
7783-08-6
selenic acid

| Conditions | Yield |
|---|---|
| In not given Electrolysis; |

| Conditions | Yield |
|---|---|
| In water byproducts: CuS; | |
| In water byproducts: CuSO4, CuS; | |
| In water |

| Conditions | Yield |
|---|---|
| In not given | |
| In not given |

| Conditions | Yield |
|---|---|
| With nitric acid In water byproducts: Cl2; dissolving of a suspension of Se in H2O by addn. of HNO3; concg.; addn. of HClO3; evapn.; concn. on passing of air through the soln. under reduced pressure; concn. at 150°C under reduced pressure;; diln. with H2O; filtration; concn. in vac.;; | 90-95 |
| In not given | |
| With HNO3 In water byproducts: Cl2; dissolving of a suspension of Se in H2O by addn. of HNO3; concg.; addn. of HClO3; evapn.; concn. on passing of air through the soln. under reduced pressure; concn. at 150°C under reduced pressure;; diln. with H2O; filtration; concn. in vac.;; | 90-95 |
| In not given |

| Conditions | Yield |
|---|---|
| In not given at elevated temp.;; | |
| In not given at elevated temp.;; |
-
-
7446-08-4
selenium(IV) oxide

-
-
7697-37-2
nitric acid

-
A
-
7783-00-8
selenious acid

-
B
-
7783-08-6
selenic acid

| Conditions | Yield |
|---|---|
| In not given | A n/a B 0% |
| In not given |


| Conditions | Yield |
|---|---|
| In nitric acid formation of small amounts of selenic acid, addition of HCl, avoids formation of selenic acid;; |

| Conditions | Yield |
|---|---|
| In not given | A n/a B 0% |
| In not given |


-
-
7783-08-6
selenic acid

| Conditions | Yield |
|---|---|
| In water Electrolysis; cathode: Pt vessel, anode: Pt spiral, cathodic current density: 0.2 A/dm*dm, ambient temp.;; |

| Conditions | Yield |
|---|---|
| In water incomplete reaction;; |

| Conditions | Yield |
|---|---|
| In water byproducts: PbS; introduction of H2S into an aq. soln. of PbSeO4;; filtration; boiling of the filtrate; concn.;; | |
| In water byproducts: PbS; | |
| In water introduction of H2S into a suspension of PbSeO4 in H2O;; |

| Conditions | Yield |
|---|---|
| In melt oxidation with melting KNO3;; |

| Conditions | Yield |
|---|---|
| With lead(II) nitrate In not given melting of SeO2 and KNO3; dissolving of the melt in aq. HNO3; boiling; addition of Pb(NO3)2; decomposition of the formed PbSeO4 with H2S;; | |
| With Pb(NO3)2 In not given melting of SeO2 and KNO3; dissolving of the melt in aq. HNO3; boiling; addition of Pb(NO3)2; decomposition of the formed PbSeO4 with H2S;; | |
| With Pb(NO3)2 |


| Conditions | Yield |
|---|---|
| In melt oxidation with melting KNO3;; |

| Conditions | Yield |
|---|---|
| With hydrogen peroxide In water 50°C in O2 atm.; dehydrated with P2O5-dried oxygen stream and a partial vac.; |

| Conditions | Yield |
|---|---|
| With mercury(I) chloride In hydrogenchloride | |
| In hydrogenchloride |

| Conditions | Yield |
|---|---|
| In water repeated evapn. of SeO2 and an excess of 30% H2O2 in an open vessel;; | |
| In water boiling of SeO2 and 30% H2O2 under reflux for some hours;; | |
| In water repeated evapn. of SeO2 and an excess of 30% H2O2 in an open vessel;; | |
| With O2 In water; dihydrogen peroxide storage of an aq. soln. of SeO2 and H2O2 for 24 hours; boiling for 12 hours under reflux and passing of O2 through the solution;; concn. under reduced pressure;; | |
| In water boiling of SeO2 and 30% H2O2 under reflux for some hours;; |

| Conditions | Yield |
|---|---|
| In melt oxidation with melting NaNO3;; |

| Conditions | Yield |
|---|---|
| In water Electrolysis; use of Pb-anode;; |


| Conditions | Yield |
|---|---|
| In nitric acid Electrolysis; free of SeO2;; | |
| With bromine In water equilibrium reaction;; not isolated;; | |
| With bromine In water equilibrium reaction;; |

| Conditions | Yield |
|---|---|
| In water Electrolysis; various conditions;; | |
| In water Electrolysis; use of a diaphragm, mixture of 6% H2SeO4 and 10% Na2SeO4 as catholyte, aq. H2SeO3 as anolythe;; |

| Conditions | Yield |
|---|---|
| at 10 °C, with excess of H2SeO4; washing with water and acetone; | 100% |
| with dild. soln. of H2SeO4, mixing, at 10 °C; | 100% |
| at 10 °C, with excess of H2SeO4; washing with water and acetone; | 100% |
| with dild. soln. of H2SeO4, mixing, at 10 °C; | 100% |

| Conditions | Yield |
|---|---|
| compd. is heated with selenic acid in a sealed tube at 553 K (1 wk); | 99.9% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Pd powder (30 mg) and H2SeO4 (1 ml) sealed in glass ampoule; heated (350°C, 72 h); cooled (3°C/h); | 99% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Pd powder (15 mg) and H2SeO4 (1 ml) sealed in glass ampoule; heated (350°C, 72 h); cooled (3°C/h); | 99% |
| In further solvent(s) on dissolving Pd sponge in a hot mixture of H2SeO4 and HNO3 and slow cooling down of concd. soln.;; on evaporization of soln. of Pd in H2SeO4, selenate is not obtained without impurity of H2SeO4 or H2SeO3;; | |
| In further solvent(s) on dissolving Pd sponge in a hot mixture of H2SeO4 and HNO3 and slow cooling down of concd. soln.;; on evaporization of soln. of Pd in H2SeO4, selenate is not obtained without impurity of H2SeO4 or H2SeO3;; |

| Conditions | Yield |
|---|---|
| In neat (no solvent) Pd powder (8 mg) and H2SeO4 (1 ml) and SeO3 (50 mg) sealed in glass ampoule; heated to 250°C; cooled (3°C/h); | 99% |


| Conditions | Yield |
|---|---|
| at 20℃; | 97% |

| Conditions | Yield |
|---|---|
| In water mixt. of butylamine, H2SeO4 and U compd. in H2O was stirred until complete homogenization; poured into watch glass and evapd. for 6-7 d; | 90% |


| Conditions | Yield |
|---|---|
| In water refluxing MoO3 and pyrazole with excess H2SeO4; recrystn. (Me2CO diffusion into aq. soln.); elem. anal.; | 89% |


-
-
7783-08-6
selenic acid

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: Cl2; H2SeO4 and Cr-complex were warmed at 70°C for 6 h, soln. was allowed to cool to room temp., MeOH was added; collected, washed with MeOH; elem. anal.; | 89% |

| Conditions | Yield |
|---|---|
| In water High Pressure; AgNO3, MoO3, H2SeO4 and H2O placed in autoclave, sealed, heated at 200°C for 3 d, cooled to room temp. at 9°C/h; ppt. washed with H2O and MeOH, dried; | 88.8% |


| Conditions | Yield |
|---|---|
| With sulfuric acid; potassium iodide In sulfuric acid byproducts: I2; sepn. of the formed I2 by distn.;; | 87.8% |
| With sulfuric acid; potassium iodide In sulfuric acid byproducts: I2, H2O; storage at room temperature for 4 weeks;; | 38.4% |
| With sodium hypophosphite In water heating for a longer period of time;; | >99 |

| Conditions | Yield |
|---|---|
| With Si(OEt)4; HNO3 In water High Pressure; Nd2O3 (1 equiv.) (acidified with HNO3), KOH (2 equiv.) and H2SeO4 (3 equiv.) were mixed in H2O; soln. was stirred for 30 min; Si(OEt)4 was addedgradually; stirred at 353 K; sealed in autoclave; heated at 483 K for 3 d; elem. anal.; | 85% |


-
-
7732-18-5
water

-
-
7783-08-6
selenic acid

-
-
603-35-0
triphenylphosphine

-
-
332401-94-2
[Ag(P(C6H5)3)2(HSeO4)(H2O)]*[Ag(P(C6H5)3)2(HSeO4)]*H2O

| Conditions | Yield |
|---|---|
| In ethanol mixt. of Ag salt and ligand dissolved in warm EtOH, cooled, H2SeO4/H2O added, heated with stirring; crystd. on slow cooling to room temp., washed (ice-cold aq. EtOH); elem.anal.; | 84% |


| Conditions | Yield |
|---|---|
| With KNO3 In water High Pressure; under hydrothermal conditions; appropriate amt. Pd(NO3)2, KNO3, concd. H2SeO4 and H2O placed in autoclave; sealed; heated at 200°C for 4 d; cooled to room temp. (9°C/h); crystals washed with H2O and MeOH; dried; detd. by EDX analysis; | 82% |

| Conditions | Yield |
|---|---|
| With KOH; Si(OEt)4; HNO3 In water High Pressure; La2O3 (1 equiv.) (acidified with HNO3), KOH (2 equiv.) and H2SeO4 (3 equiv.) were mixed in H2O; soln. was stirred for 30 min; Si(OEt)4 was addedgradually; stirred at 353 K; sealed in autoclave; heated at 473 K for 3 d; elem. anal.; | 80% |

| Conditions | Yield |
|---|---|
| In water High Pressure; a few pieces of Au and concd. selenic acid placed in steel autoclave; heated at 250°C for 10 h; cooled slowly (5°C/h); crystals washed with water; dried in air; | 80% |

| Conditions | Yield |
|---|---|
| In water High Pressure; under hydrothermal conditions; U compd., H2SeO4 and piperazine (molar ratio 1:23:2.7) loaded in autoclave; water added; sealed; heated at 130°C for 2 d; cooled to 23°C (9°C/h); mother liquor decanted; crystals washed with MeOH; dried; elem. anal.; | 80% |


| Conditions | Yield |
|---|---|
| In water at 200℃; for 120h; Autoclave; | 80% |


| Conditions | Yield |
|---|---|
| at 20℃; under 760.051 Torr; | 77% |


| Conditions | Yield |
|---|---|
| With potassium nitrate In water at 220℃; for 96h; Glovebox; Autoclave; | 73.6% |

Selenic acid Chemical Properties
Selenic acid is a stronger oxidiser than sulfuric acid, capable of liberating chlorine from chloride ions, being reduced to selenous acid in the process:
2 H2SeO4 → 2 H2SeO3 + O2
IUPAC Name: Selenic acid
The MF of Selenic acid (CAS NO.7783-08-6) is H2O4Se.
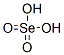
The MW of Selenic acid (CAS NO.7783-08-6) is 144.97.
Synonyms of Selenic acid (CAS NO.7783-08-6): Selenicacid, 40% solution
Product Categories: Acids;Electronic Chemicals;Micro/Nanoelectronics
Stability: Stable, but decomposes on heating. Non-flammable. Incompatible with metals, combustible materials. Hygroscopic
Apperance: colourless or white crystals
Melting Point: 58 °C
Boiling Point: 260 °C
Density: 1.407 g/mL at 25 °C
Refractive index: n20/D 1.5174
Flash Point: >230 °C
Merck: 14,8429
EINECS: 231-979-4
Selenic acid Uses
Selenic acid (CAS NO.7783-08-6) is used as analytical reagent,also for the preparation of selenium salts.
Selenic acid Production
Due to the instability of selenium trioxide, it is impractical to synthesize selenic acid by dissolving selenium trioxide in water, unlike sulfuric acid, which can be synthesized by dissolving sulfur trioxide in water. Instead, it must be prepared by oxidising selenium compounds in lower oxidation states.
One method of preparing selenic acid is by the oxidation of selenium dioxide with hydrogen peroxide:
Selenic acid may also be prepared by the oxidation of selenous acid (H2SeO3) with halogens, such as chlorine or bromine, or with potassium permanganate. However, using chlorine or bromine as the oxidising agent also produces hydrochloric or hydrobromic acid as a side-product, which needs to be removed from the solution since they can reduce the selenic acid to selenous acid.
Another method of preparing selenic acid is by the oxidation of elemental selenium in water suspension by chlorine:
Se + 4 H2O + 3 Cl2 → H2SeO4 + 6 HCl
Selenic acid Consensus Reports
Selenium and its compounds are on the Community Right-To-Know List. EPA Genetic Toxicology Program. Reported in EPA TSCA Inventory.
Selenic acid Safety Profile
Selenium compounds are poisons. A corrosive irritant to skin, eyes, and mucous membranes. When heated to decomposition it emits toxic fumes of Se. See also SELENIUM COMPOUNDS.Safety information of Selenic acid (CAS NO.7783-08-6):
Hazard Codes  T,
T, N
N
Risk Statements
23/24/25 Toxic by inhalation, in contact with skin and if swallowed
34 Causes burns
50/53 Very Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment
33 Danger of cumulative effects
Safety Statements
23 Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer)
26 In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37/39 Wear suitable protective clothing, gloves and eye/face protection
45 In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
61 Avoid release to the environment. Refer to special instructions safety data sheet
60 This material and/or its container must be disposed of as hazardous waste
28 After contact with skin, wash immediately with plenty of ... (to be specified by the manufacturer)
20/21 When using, do not eat, drink or smoke
RIDADR UN 3264 8/PG 1
RTECS VS6575000
HazardClass 8
PackingGroup I
Selenic acid Standards and Recommendations
OSHA PEL: TWA 0.2 mg(Se)/m3
ACGIH TLV: TWA 0.2 mg(Se)/m3
DFG MAK: 0.1 mg(Se)/m3
DOT Classification: 8; Label: Corrosive
Selenic acid Specification
Selenic acid is the chemical compound with the formula H2SeO4. It is an oxoacid of selenium, and its structure is more accurately described as (HO)2SeO2.
As predicted by VSEPR theory, the selenium center is tetrahedral, with a Se–O bond length of 161 pm. In the solid state, it crystallizes in an orthorhombic structure.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View