-
Name
SUCCINALDEHYDE
- EINECS 211-333-8
- CAS No. 638-37-9
- Article Data124
- CAS DataBase
- Density 0.965 g/cm3
- Solubility
- Melting Point
- Formula C4H6O2
- Boiling Point 154.1 °C at 760 mmHg
- Molecular Weight 86.0904
- Flash Point 50.4 °C
- Transport Information
- Appearance
- Safety 36/37
- Risk Codes 40
-
Molecular Structure
- Hazard Symbols Xn
- Synonyms Succinaldehyde(6CI,8CI);1,4-Butanedial;1,4-Butanedione;4-Oxobutyraldehyde;NSC 11057;Butanedial;Succinic aldehyde;Succinic dialdehyde;β-Formylpropionaldehyde;
- PSA 34.14000
- LogP 0.16440
Synthetic route
-
-
5259-72-3, 10060-40-9, 111-78-4
1,5-dicyclooctadiene

-
-
638-37-9
butanedial

| Conditions | Yield |
|---|---|
| Stage #1: cyclo-octa-1,5-diene With ozone In ethanol; dichloromethane at -78℃; Stage #2: With triphenylphosphine In ethanol; dichloromethane at -78 - 20℃; | 98% |
| Stage #1: cyclo-octa-1,5-diene With oxygen; ozone In dichloromethane at -78℃; Stage #2: With triphenylphosphine In dichloromethane at -78 - 20℃; | 98% |
| Stage #1: cyclo-octa-1,5-diene With ozone In dichloromethane at -78℃; Stage #2: With triphenylphosphine In dichloromethane at -78 - 20℃; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With thexylbromoborane dimethyl sulfide complex In carbon disulfide; dichloromethane at -20 - 20℃; for 1h; | 93% |

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; [2,2]bipyridinyl; tetrakis(acetonitrile)copper(I) trifluoromethanesulfonate; 9-azabicyclo<3.3.1>nonane-N-oxyl In acetonitrile at 22℃; for 2h; | A 92% B 8% |
-
-
110-82-7
cyclohexane

-
-
85684-59-9
percarbonate de O,O-t-butyle et O-vinyle

-
A
-
625-34-3
3-oxobutyraldehyde

-
B
-
638-37-9
butanedial

-
C
-
92-51-3
cyclohexylcyclohexane

-
D
-
5664-21-1
cyclohexylacetaldehyde

-
E
-
67-64-1
acetone

-
F
-
75-65-0
tert-butyl alcohol

| Conditions | Yield |
|---|---|
| at 140℃; for 3.5h; Product distribution; Mechanism; sealed tube; | A 7% B 4% C n/a D 25% E 5% F 90% |


| Conditions | Yield |
|---|---|
| With sodium hypochlorite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical In dichloromethane; water at 0 - 10℃; for 1h; | 88.6% |
| With pivaloyl chloride; dimethyl sulfoxide; triethylamine In dichloromethane at -78 - 20℃; for 1h; | 86% |
| With oxalyl dichloride; dimethyl sulfoxide; triethylamine In dichloromethane at -10 - 0℃; for 2.5h; | |
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; [bis(acetoxy)iodo]benzene In dichloromethane at 35℃; for 0.075h; Flow reactor; chemoselective reaction; | 91 %Chromat. |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; water at 20℃; for 0.25h; | 87% |
| With hydrogenchloride In water at 90℃; for 1h; | 77% |
| With hydrogenchloride; water; sodium carbonate for 10h; | 70% |

| Conditions | Yield |
|---|---|
| With acetic acid In water for 1h; | 85% |

-
-
638-37-9
butanedial

| Conditions | Yield |
|---|---|
| With acetic acid In water for 1h; | 82% |

-
-
638-37-9
butanedial

| Conditions | Yield |
|---|---|
| With acetic acid In water for 1h; | 82% |
-
-
98-82-8
Isopropylbenzene

-
-
85684-59-9
percarbonate de O,O-t-butyle et O-vinyle

-
A
-
625-34-3
3-oxobutyraldehyde

-
B
-
638-37-9
butanedial

-
C
-
1889-67-4
dicumene

-
D
-
75-07-0
acetaldehyde

-
E
-
67-64-1
acetone

-
F
-
75-65-0
tert-butyl alcohol

| Conditions | Yield |
|---|---|
| at 140℃; for 3.5h; Product distribution; Mechanism; sealed tube; | A 15% B 10% C n/a D n/a E 25% F 69% |

-
-
98-82-8
Isopropylbenzene

-
-
85684-59-9
percarbonate de O,O-t-butyle et O-vinyle

-
A
-
625-34-3
3-oxobutyraldehyde

-
B
-
638-37-9
butanedial

-
C
-
67-64-1
acetone

-
D
-
75-65-0
tert-butyl alcohol

| Conditions | Yield |
|---|---|
| at 140℃; for 3.5h; sealed tube; | A 15% B 10% C 25% D 69% |


| Conditions | Yield |
|---|---|
| Stage #1: cis,trans-2,5-dimethoxytetrahydrofuran In water at 75℃; for 4h; Stage #2: methanol In water at 115℃; | 69% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In water Reflux; Inert atmosphere; | 56% |
| With hydrogenchloride | |
| With hydrogenchloride |
-
-
280-53-5
2,3-dioxabicyclo<2.2.2>octane

-
A
-
74-85-1
ethene

-
B
-
638-37-9
butanedial

-
C
-
637-88-7
1,4-Cyclohexanedione

| Conditions | Yield |
|---|---|
| at 450℃; under 0.001 Torr; Product distribution; Mechanism; | A n/a B 56% C 42% |


| Conditions | Yield |
|---|---|
| at 450℃; | A 56% B 42% |
-
-
280-53-5
2,3-dioxabicyclo<2.2.2>octane

-
A
-
74-85-1
ethene

-
B
-
638-37-9
butanedial

-
C
-
931-71-5
cis-1,4-cyclohexanediol

-
D
-
13482-22-9
4-hydroxycyclohexanone

| Conditions | Yield |
|---|---|
| With tris(triphenylphosphine)ruthenium(II) chloride In dichloromethane at 50℃; for 2h; Product distribution; ruthenium(II) catalyzed reaction of 1,4-endoperoxide; | A 43% B 19% C 17% D 11% |
| With tris(triphenylphosphine)ruthenium(II) chloride In dichloromethane at 50℃; for 2h; Title compound not separated from byproducts; | A 43 % Chromat. B 19 % Spectr. C 17 % Chromat. D 11 % Chromat. |


| Conditions | Yield |
|---|---|
| With hydrogenchloride In tetrahydrofuran Ambient temperature; | A 38% B n/a |

| Conditions | Yield |
|---|---|
| With ozone In dichloromethane at -78℃; | A 8% B 25% C 15% D 30% |

-
-
280-53-5
2,3-dioxabicyclo<2.2.2>octane

-
A
-
638-37-9
butanedial

-
B
-
931-71-5
cis-1,4-cyclohexanediol

-
C
-
13482-22-9
4-hydroxycyclohexanone

-
D
-
637-88-7
1,4-Cyclohexanedione

| Conditions | Yield |
|---|---|
| With cobalt(II) 5,10,15,20-tetraphenylporphyrin In chloroform at 60℃; for 11h; Further byproducts given; | A 21% B 28% C 17% D 5% |


| Conditions | Yield |
|---|---|
| With ozone In dichloromethane at -78℃; | A 8% B 27% C 25% D 14% |


| Conditions | Yield |
|---|---|
| With oxygen; silver at 350℃; | |
| With oxygen; copper at 350℃; |

| Conditions | Yield |
|---|---|
| With hydrogen; copper at 250℃; |

| Conditions | Yield |
|---|---|
| With cis-nitrous acid | |
| With water unter Kuehlung mit den nitrosen Gasen, die man aus Arsenik und Salpetersaeure; | |
| With water ueber mehrere Stufen; |

| Conditions | Yield |
|---|---|
| With mineral acid |
-
-
13544-77-9
2,5-dihydroxyadipic acid

-
-
638-37-9
butanedial

| Conditions | Yield |
|---|---|
| With water; dihydrogen peroxide; iron(II) sulfate |
-
-
52190-10-0
N,N'-dimethyl-N,N'-diphenylsuccinamide

-
-
638-37-9
butanedial

| Conditions | Yield |
|---|---|
| With tetrahydrofuran; lithium aluminium tetrahydride |
-
-
103397-76-8
1,2-bis-(1,3-diphenyl-imidazolidin-2-yl)-ethane

-
-
638-37-9
butanedial

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With sodium; toluene anschliessend Behandeln mit wss.Oxalsaeure; |
-
-
37612-79-6
3,5,3',5'-tetramethyl-1H,1'H-1,1'-succinyl-bis-pyrazole

-
-
638-37-9
butanedial

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride |

| Conditions | Yield |
|---|---|
| Stage #1: 2-bromoprop-1-ene With magnesium In tetrahydrofuran at 40℃; for 2h; Inert atmosphere; Stage #2: butanedial In tetrahydrofuran at 20℃; for 2h; Inert atmosphere; | 100% |
| (i) Mg, (ii) /BRN= 1735656/; Multistep reaction; |

| Conditions | Yield |
|---|---|
| With ionic liquid-supported SiO2*CF3SO3(1-) In ethanol at 20℃; for 0.166667h; Paal-Knorr Furan Synthesis; Green chemistry; | 98% |
| With calcium(II) nitrate tetrahydrate In neat (no solvent) Reflux; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide; iron pentacarbonyl; carbon monoxide In ethanol; water for 5h; Ambient temperature; | 96% |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
638-37-9
butanedial

-
-
667870-41-9
(7aR,10R,12S)-10-(1H-benzotriazol-1-yl)-7a,8,9,10-tetrahydro-12-phenyl-12H-naphtho[1,2-e]pyrrolo[2,1-b][1,3]oxazine

| Conditions | Yield |
|---|---|
| In dichloromethane at -5 - 0℃; for 5h; | 93% |
| In dichloromethane; water at 0℃; for 4h; | 93% |


| Conditions | Yield |
|---|---|
| With L-proline In 1,2-dichloro-ethane at 20℃; Inert atmosphere; | 93% |
-
-
638-37-9
butanedial

-
-
112402-14-9
5-Bromo-2,2-dimethyl-1-(tetrahydropyranyloxy)-pentane

-
-
738606-57-0
1,14-bis(tetrahydropyran-2-yloxy)-2,2,13,13-tetramethyltetradecan-6,9-diol

| Conditions | Yield |
|---|---|
| Stage #1: 5-Bromo-2,2-dimethyl-1-(tetrahydropyranyloxy)-pentane With magnesium In tetrahydrofuran for 2h; Heating; Stage #2: butanedial In tetrahydrofuran at 20℃; | 92% |
| Stage #1: 5-Bromo-2,2-dimethyl-1-(tetrahydropyranyloxy)-pentane With magnesium In tetrahydrofuran for 2h; Heating / reflux; Stage #2: butanedial In tetrahydrofuran at 0 - 20℃; Stage #3: With hydrogenchloride In tetrahydrofuran; water pH=1 - 2; | 92% |

| Conditions | Yield |
|---|---|
| With indium In water at 60℃; under 1241.19 - 2585.81 Torr; for 0.00416667h; Microwave irradiation; Green chemistry; chemoselective reaction; | 92% |

| Conditions | Yield |
|---|---|
| With tin; acetic acid In methanol | 91% |

| Conditions | Yield |
|---|---|
| With ytterbium(III) triflate In ethanol at 20℃; for 3h; Paal-Knorr Furan Synthesis; | 91% |

-
-
638-37-9
butanedial

-
-
86302-43-4
(tert-Butoxycarbonylmethylene)triphenylphosphorane

-
-
679436-07-8
(2E,6E)-octa-2,6-diendioic acid di-tert-butyl diester

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; Wittig reaction; Inert atmosphere; | 90% |
-
-
638-37-9
butanedial

-
-
21204-67-1
methyl (triphenylphosphoranylidene)acetate

-
A
-
32347-20-9
(2Z,6Z)-octa-2,6-dienedioic acid dimethyl ester

-
B
-
26505-44-2
dimethyl (E,E)-octa-2,6-diendioate

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; Wittig reaction; Inert atmosphere; | A n/a B 90% |


| Conditions | Yield |
|---|---|
| With ytterbium(III) triflate In ethanol at 20℃; for 5h; Paal-Knorr Furan Synthesis; | 90% |


| Conditions | Yield |
|---|---|
| With ytterbium(III) triflate In ethanol at 20℃; for 2h; Paal-Knorr Furan Synthesis; | 90% |

-
-
638-37-9
butanedial

-
-
2570-98-1
4-aminobenzonitrile hydrochloride

-
-
934236-05-2
1-(2-chloro-5-pyridylmethyl)-2-nitromethyleneimidazolidine

| Conditions | Yield |
|---|---|
| In acetonitrile at 0℃; for 1.5h; | 89.4% |


| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol at -5 - 45℃; for 14h; Temperature; Reagent/catalyst; Inert atmosphere; | 89.3% |
-
-
638-37-9
butanedial

-
-
591-51-5
phenyllithium

-
A
-
2085-90-7
1,4-diphenyl-1,4-butanediol

-
B
-
36866-66-7
5-phenyloxolan-2-ol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether; cyclohexane at -40℃; Temperature; | A 7% B 88% |


| Conditions | Yield |
|---|---|
| With ytterbium(III) triflate In ethanol at 20℃; for 4h; Paal-Knorr Furan Synthesis; | 88% |


| Conditions | Yield |
|---|---|
| With ytterbium(III) triflate In ethanol at 20℃; for 7h; Paal-Knorr Furan Synthesis; | 88% |


| Conditions | Yield |
|---|---|
| With ytterbium(III) triflate In ethanol at 20℃; for 4h; Paal-Knorr Furan Synthesis; | 88% |


| Conditions | Yield |
|---|---|
| With potassium hydroxide; iron pentacarbonyl; carbon monoxide In ethanol; water for 12h; Ambient temperature; | 87% |
-
-
638-37-9
butanedial

-
-
1099-45-2
ethyl (triphenylphosphoranylidene)acetate

-
-
71778-56-8
ethyl (2E)-6-oxohex-2-enoate

| Conditions | Yield |
|---|---|
| In dichloromethane for 24h; | 87% |
| In dichloromethane for 48h; | 58% |
| In dichloromethane at 0℃; for 48h; | 58% |
| In dichloromethane at 20℃; for 48h; | 6.3 g |

| Conditions | Yield |
|---|---|
| With N-methylcyclohexylamine; water In N,N-dimethyl-formamide at 140℃; for 5h; | 86% |
Succindialdehyde Specification
The Succindialdehyde, with the CAS registry number 638-37-9, is also known as β-Formylpropionaldehyde. Its EINECS number is 211-333-8. This chemical's molecular formula is C4H6O2 and molecular weight is 86.09. What's more, its IUPAC name is butanedial. It is highly reactive. Usually, it is handled as the hydrates or methanol-derived acetal. It is used as a crosslinking agent but is less widely used than the related dialdehyde glutardialdehyde.
Physical properties of Succindialdehyde are: (1)ACD/LogP: -0.22; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.22; (4)ACD/BCF (pH 5.5): 1; (5)ACD/KOC (pH 5.5): 18.05; (6)#H bond acceptors: 2; (7)#H bond donors: 0; (8)#Freely Rotating Bonds: 3; (9)Polar Surface Area: 34.14 Å2; (10)Index of Refraction: 1.386; (11)Molar Refractivity: 20.95 cm3; (12)Molar Volume: 89.1 cm3; (13)Polarizability: 8.3×10-24cm3; (14)Surface Tension: 30.1 dyne/cm; (15)Density: 0.965 g/cm3; (16)Flash Point: 50.4 °C; (17)Enthalpy of Vaporization: 39.09 kJ/mol; (18)Boiling Point: 154.1 °C at 760 mmHg; (19)Vapour Pressure: 3.22 mmHg at 25°C.
Preparation of Succindialdehyde: this chemical can be prepared by succinic acid at the temperature of -20 - 20 °C. This reaction will need reagent thexylbromoborane-dimethylsulfide and solvents CS2, CH2Cl2 with the reaction time of 1 hour. The yield is about 93%.

Uses of Succindialdehyde: it can be used to produce 3-benzyl-2-methylimino-thiazolidin-5-one at the ambient temperature. It will need reagents ironpentacarbonyl, 1 M potassium hydroxide, carbon monoxide and solvents ethanol, H2O with the reaction time of 20 hours. The yield is about 85%.
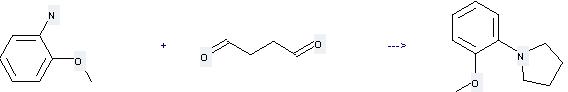
You can still convert the following datas into molecular structure:
(1)Canonical SMILES: C(CC=O)C=O
(2)InChI: InChI=1S/C4H6O2/c5-3-1-2-4-6/h3-4H,1-2H2
(3)InChIKey: PCSMJKASWLYICJ-UHFFFAOYSA-N
Related Products
- Succindialdehyde
- 638-38-0
- 63839-24-7
- 6384-08-3
- 638-41-5
- 63-84-3
- 63843-42-5
- 63843-49-2
- 63843-53-8
- 63843-89-0
- 63844-43-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View