-
Name
Tetrabutyl titanate
- EINECS 227-006-8
- CAS No. 5593-70-4
- Density 0.996 g/cm3
- Solubility rapidly hydrolized in water
- Melting Point -55 °C
- Formula C16H36O4Ti
- Boiling Point 310-314 °C
- Molecular Weight 340.339
- Flash Point 78 °C
- Transport Information UN 1993 3/PG 3
- Appearance colourless to light yellow viscous liquid
- Safety 16-26-39-24/25
- Risk Codes 10-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 1-Butanol,titanium(4+) salt (9CI);Butyl titanate(IV) ((BuO)4Ti);Orgatix T 25;Orgatix TA 25;TBT;TBT 100;Tetra-n-butoxytitanium;Tetra-n-butyl orthotitanate;Tetra-n-butyl titanate;Tetrabutoxytitanium;Tetrabutyl orthotitanate;Tetrakis(butanolato)titanium;Titanium butoxide;Titaniumn-butoxide;Titanium tetra-n-butoxide;Titaniumtetrabutylate;Titanium(IV) butoxide;Titanium, tetrabutoxy-;Tyzor TNBT;n-Butanol titaniumsalt;n-Butyl titanate;Terabutyl titanate;
- PSA 36.92000
- LogP 5.06960
Synthetic route

| Conditions | Yield |
|---|---|
| With NH4NO3-NH3 In toluene soln. of TiCl4 added to alcohol with cooling and stirring, soln. added to NH4NO3-NH3 with stirring and cooling at 0-15°C; organic layer separated, solvent removed, product distilled under vacuum; | 86% |
| With formamide In butan-1-ol byproducts: NH4Cl; soln. of TiCl4 added to alcohol with cooling and stirring, HCONH2 added, mixture boiled with stirring for 5-6 h, cooled; NH4Cl separated by filtration or HCONH2 added with stirring, product distilled under vacuum; | 66% |
| With formamide In n-heptane byproducts: NH4Cl; soln. of TiCl4 added to alcohol with cooling and stirring, HCONH2 added, mixture boiled with stirring for 5-6 h, cooled; NH4Cl separated by filtration or HCONH2 added with stirring, product distilled under vacuum; | 60% |

| Conditions | Yield |
|---|---|
| With ammonia | |
| With sodium; benzene | |
| With ammonia |

| Conditions | Yield |
|---|---|
| unter Kuehlung und nachfolgendes Erhitzen des Reaktionsgemisches zum Sieden; |

| Conditions | Yield |
|---|---|
| Electrochem. Process; anodic oxidn. of Ti in BuOH (dry argon); |

| Conditions | Yield |
|---|---|
| In butan-1-ol Kinetics; mixing 0.2 M butoxide and 3.0 M water (pptn.), stay for 1440 min; detd. by photocolorimetry, thermal analysis; | 97% |
| In butan-1-ol Kinetics; mixing 0.4 M butoxide and 0.4 M water (pptn.), stay for 4 h, addn. 1 volume part of 2.8 M water, stay for 4320 min; detd. by photocolorimetry, thermal analysis; | 97.1% |
| In butan-1-ol Kinetics; mixing 0.2 M butoxide and 3.0 M water (pptn.), stay for 240 min; detd. by photocolorimetry, thermal analysis; | 95.5% |
-
-
864862-42-0
4-(2,3-dihydro-1H-inden-5-yloxy)phthalonitrile

-
-
5593-70-4
titanium (IV) butoxide

| Conditions | Yield |
|---|---|
| With urea In octanol at 150℃; for 6h; | 94.1% |

| Conditions | Yield |
|---|---|
| at 270℃; | 92% |

| Conditions | Yield |
|---|---|
| at 260℃; | 91% |

| Conditions | Yield |
|---|---|
| at 240℃; | 88% |

| Conditions | Yield |
|---|---|
| at 245℃; | 87% |

| Conditions | Yield |
|---|---|
| at 120℃; | 87% |

| Conditions | Yield |
|---|---|
| at 140℃; | 87% |
-
-
5593-70-4
titanium (IV) butoxide

-
-
57-13-6
urea

-
-
91-15-6
phthalonitrile

-
-
26201-32-1
oxo(phthalocyaninato)titanium(IV)

| Conditions | Yield |
|---|---|
| In further solvent(s) byproducts: NH3, 1-octyl carbamate; N2 atmosphere; stirring alkoxide, urea and DCNB (150°C., 6 h, in 1-octanol); addn. of MeOH, refluxing (30 min, crystn.), filtration, washing (toluene, MeOH, H2O), drying (100°C, 1 Torr, 3 - 6 h); elem. anal.; | 86.2% |
| In benzyl alcohol stirring alkoxide, urea and DCNB (140 - 150°C.); | 77% |
| In pentan-1-ol stirring alkoxide, urea and DCNB (140 - 150°C.); | 77% |


| Conditions | Yield |
|---|---|
| at 240℃; | 82% |

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In hexane; toluene for 24h; Ambient temperature; | 78% |
-
-
55-21-0
benzamide

-
-
5593-70-4
titanium (IV) butoxide

-
-
91-15-6
phthalonitrile

-
-
26201-32-1
oxo(phthalocyaninato)titanium(IV)

| Conditions | Yield |
|---|---|
| In further solvent(s) stirring alkoxide, benzamide and DCNB (140 - 150°C., in 1-octanol); | 78% |

-
-
7664-41-7
ammonia

-
-
5593-70-4
titanium (IV) butoxide

-
-
91-15-6
phthalonitrile

-
-
26201-32-1
oxo(phthalocyaninato)titanium(IV)

| Conditions | Yield |
|---|---|
| In further solvent(s) addn. of DCBN and solvent (1-octanol) to mixt. of alkoxide and liquid NH3, heating (155°C); | 77% |
| In further solvent(s) NH3 atmosphere (bubbling); stirring alkoxide and DCNB (150°C., in1-octanol); | 75% |


| Conditions | Yield |
|---|---|
| In butan-1-ol Kinetics; mixing 0.1 M butoxide and 1.5 M water (pptn.), stay for 11400 min; detd. by photocolorimetry, thermal analysis; | 73.8% |
| In butan-1-ol Kinetics; mixing 0.1 M butoxide and 1.5 M water (pptn.), stay for 1440 min; detd. by photocolorimetry, thermal analysis; elem. anal.; | 53.7% |
| In butan-1-ol Kinetics; mixing 0.1 M butoxide and 1.5 M water (pptn.), stay for 360 min; detd. by photocolorimetry, thermal analysis; elem. anal.; | 29.1% |
-
-
60-35-5
acetamide

-
-
5593-70-4
titanium (IV) butoxide

-
-
91-15-6
phthalonitrile

-
-
26201-32-1
oxo(phthalocyaninato)titanium(IV)

| Conditions | Yield |
|---|---|
| In further solvent(s) stirring alkoxide, acetamide and DCNB (140 - 150°C., in 1-octanol); | 71% |

-
-
201230-82-2
carbon monoxide

-
-
85-46-1
1-Naphthalenesulfonyl chloride

-
-
5593-70-4
titanium (IV) butoxide

-
A
-
3007-95-2
naphthalene-1-carboxylic acid butyl ester

-
B
-
39178-11-5
1,2-di(naphthalen-1-yl)disulfane

| Conditions | Yield |
|---|---|
| tetrakis(triphenylphosphine) palladium(0) In acetonitrile at 150 - 160℃; under 7600 Torr; | A 43% B 14% |


| Conditions | Yield |
|---|---|
| In tert-butyl alcohol at 80℃; for 96h; | 41% |
-
-
5593-70-4
titanium (IV) butoxide

-
-
121-44-8
triethylamine

-
-
213119-80-3
1,3,5-tris(2,3-dihydroxybenzamido)benzene

| Conditions | Yield |
|---|---|
| In methanol addn. of soln. of butoxide to soln. of ligand and triethylamine, stirring (ambient temp., 12 h, pptn.); centrifugation, suspn. in DMF, refluxing (N2 atmosphere, 12 h), filtration, evapn. to dryness (vac.); | 37% |


| Conditions | Yield |
|---|---|
| In butan-1-ol for 17h; Heating; | 29% |
-
-
563-80-4
3-methyl-butan-2-one

-
-
463-51-4
Ketene

-
-
5593-70-4
titanium (IV) butoxide

-
-
54074-72-5
3-Hydroxy-3,4-dimethyl-pentanoic acid butyl ester

| Conditions | Yield |
|---|---|
| (i), (ii) aq. H2SO4; Multistep reaction; |


| Conditions | Yield |
|---|---|
| at 100℃; under 1 - 2 Torr; |
-
-
556-67-2
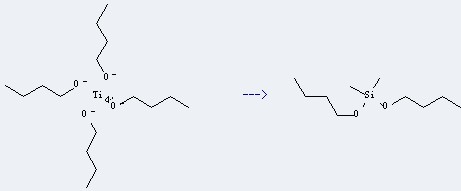
octamethylcyclotetrasiloxane

-
-
5593-70-4
titanium (IV) butoxide

-
-
1591-02-2
dimethyldibutoxysilane

| Conditions | Yield |
|---|---|
| at 180℃; |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
5593-70-4
titanium (IV) butoxide

-
-
18043-98-6
1,3-dibutoxy-1,1,3,3-tetramethyldisiloxane

| Conditions | Yield |
|---|---|
| at 180℃; |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
5593-70-4
titanium (IV) butoxide

-
-
17866-36-3
hexamethyl-1,5-di-n-butoxy-trisiloxane

| Conditions | Yield |
|---|---|
| at 180℃; |
-
-
556-67-2
octamethylcyclotetrasiloxane

-
-
5593-70-4
titanium (IV) butoxide

-
-
18547-28-9
1,7-Dibutyloxy-octamethyl-tetrasiloxan

| Conditions | Yield |
|---|---|
| at 180℃; |
-
-
1451-99-6
octaethylcyclotetrasiloxane

-
-
5593-70-4
titanium (IV) butoxide

-
-
18127-33-8
Diaethyl-dibutyloxy-silan

| Conditions | Yield |
|---|---|
| at 180℃; |
-
-
2554-06-5
2,4,6,8-tetramethyl-2,4,6,8-tetravinyl-cyclotetrasiloxane

-
-
5593-70-4
titanium (IV) butoxide

-
-
18002-58-9
Dibutyloxy-methyl-vinyl-silan

| Conditions | Yield |
|---|---|
| at 180℃; |
Tetrabutyl titanate Consensus Reports
Tetrabutyl titanate Standards and Recommendations
Tetrabutyl titanate Specification
The IUPAC name of Butyl titanate is butan-1-ol; titanium. With the CAS registry number 5593-70-4, it is also named as Tetrabutoxy titanium. The product's categories are Organic-metal Salt; Catalysts for Organic Synthesis; Classes of Metal Compounds; Homogeneous Catalysts; Synthetic Organic Chemistry; Ti (Titanium) Compounds; Titanium Alkoxides, etc. (Homogeneous Catalysts); Transition Metal Compounds, and the other registry numbers are 104183-13-3; 106206-82-0; 114486-31-6; 118547-86-7; 124760-72-1; 1336-30-7; 153313-64-5; 156138-67-9; 15821-58-6; 159179-54-1; 176679-99-5; 184529-19-9; 188622-64-2; 210407-19-5; 216859-05-1; 250741-36-7; 26198-43-6; 597533-81-8; 59978-00-6; 75659-54-0; 757-47-1. Besides, it is colourless to light yellow viscous liquid, which should be stored in sealed, cool, dark and dry place. In addition, this chemical is stable, but moisture sensitive. It is also incompatible with water, moisture, strong oxidizing agents, strong acids.
The other characteristics of this product can be summarized as: (1)H-Bond Donor: 4; (2)H-Bond Acceptor: 4; (3)Rotatable Bond Count: 8; (4)Exact Mass: 344.240607; (5)MonoIsotopic Mass: 344.240607; (6)Topological Polar Surface Area: 80.9; (7)Heavy Atom Count: 21; (8)Complexity: 82.3; (9)Density: 1.00 g/mL at 20 °C(lit.); (10)Melting point: -55 °C; (11)Boiling point: 310-314 °C; (12)Refractive index: 1.486; (13)Flash point: 78 °C; (14)EINECS: 227-006-8.
Preparation of Butyl titanate: this chemical can be prepared by the reaction of Titanium tetrachloride and Butanol. The Chemical equation is as follows:
![]()
Uses of Butyl titanate: this chemical is mainly used as catalyst in ester exchange and condensation reaction. It also can be used as modifier of intensity polyester paint, additive of high-temperature coatings, medical adhesives, cross-linking agent and so on. And it is used as modified adhesives for producing metal and rubber, metal and plastic. Additionally, it also can be used for producing Dibutoxy-dimethyl-silane.
This reaction will occur at temperature of 260 °C. The yield is 91 %.
When you are using this chemical, please be cautious about it as the following: it is flammable. Please keep away from sources of ignition. It is also irritating to eyes, respiratory system and skin. You should wear eye / face protection to avoid contact with skin and eyes. Moreover, in case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
People can use the following data to convert to the molecule structure.
(1)SMILES:[Ti+2].[Ti+2].CCCC[O-].[O-]CCCC.[O-]CCCC.[O-]CCCC
(2)InChI:InChI=1/4C4H9O.2Ti/c4*1-2-3-4-5;;/h4*2-4H2,1H3;;/q4*-1;2*+2
(3)InChIKey:DLNIVGYQXIWPEV-UHFFFAOYAM
(4)Std. InChI:InChI=1S/4C4H9O.2Ti/c4*1-2-3-4-5;;/h4*2-4H2,1H3;;/q4*-1;2*+2
(5)Std. InChIKey:DLNIVGYQXIWPEV-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intravenous | 180mg/kg (180mg/kg) | U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. Vol. NX#01650, | |
| rat | LD50 | oral | 3122mg/kg (3122mg/kg) | "Prehled Prumyslove Toxikologie; Organicke Latky," Marhold, J., Prague, Czechoslovakia, Avicenum, 1986Vol. -, Pg. 1263, 1986. |
Related Products
- Tetrabutyl ammonium chloride hydrate
- TETRABUTYL AMMONIUM, SALT WITH 4-NITROPYROCATECO
- Tetrabutyl dichlorostannoxane
- Tetrabutyl orthosilicate
- Tetrabutyl titanate
- Tetrabutylammonium
- Tetrabutylammonium 4-toluenesulfonate
- Tetrabutylammonium acetate
- Tetrabutylammonium azide
- Tetrabutylammonium bis-trifluoromethane
- 559-37-5
- 55937-50-3
- 55940-03-9
- 55944-70-2
- 5594-61-6
- 55947-46-1
- 55948-94-2
- 55950-07-7
- 55954-23-9
- 55954-25-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View