Dayang Chem (Hangzhou) Co.,Ltd.
Dayangchem's R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantities
Cas:7783-55-3
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:7783-55-3
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:7783-55-3
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stock Package:1kg Application:pharmaceutical intermediates
Henan Tianfu Chemical Co., Ltd.
High quality lower price Storage:Room Temperature Package:As per buyer's Application:For organic synthesis Transportation:Shipmed by Air /sea
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Cas:7783-55-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHunan Longxianng Runhui Trading Co.,Ltd
TrifluorophosphaneAppearance:ask Storage:Keep in dry and cool condition Package:foil aluminium bag/vacuum packing Application:intermediates Transportation:By express (Door to door) such as FEDEX, DHL, EMS for small amount. By air(airport to airport)
Qingdao Sigma Chemical Co., Ltd.
Qingdao Sigma Chemical Ltd is is a global chemical industry manufacturers and suppliers of pharmaceuticals and intermediates, peptide,Nootropis etc API, food and feed additives, herbal extracts, agrochemicals and fine chemicals etc. Our Labo
Cas:7783-55-3
Min.Order:1 Gram
FOB Price: $1.0
Type:Trading Company
inquiryHangzhou Share Chemical Co., Ltd
At Share Chemical Company, we scrupulously abide by our policy of “Excellent Quality at a Reasonable Price”. We strive to satisfy all of our customers by providing the finest quality products supported by the finest in customer servi
HuBei Ipure Biotech CO.,ltd
Strict quality control system and PHD We has passed through the ISO9001:2016 Quality System Certificate, we have strict quality control system. we have about 300 employees, including 90 full-time PHD 15 PHD and masters above, 153 undergraduates, w
Cas:7783-55-3
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Trading Company
inquiryHebei mingxing biotech co.,ltd
Known for its best quality and competitve price, this chemicals we offered is widely appreciated by our customers. Prompt reaction, good quality and best service make us reliable and outstanding in this industry.Appearance:yellow to white solid or po
Nanjing Raymon Biotech Co., Ltd.
Phosphorous trifluoride Storage:keep in dry and cool condition Package:25kg or according to cutomer's demand Application:Chemical research/pharma intermediate Transportation:By Sea,by Air,By courier like DHL or Fedx. Port:Shanghai/Shenzhen
Hebei Minshang Biotechnology Co., Ltd
Trifluorophosphane Application:intermediates
Skyrun Industrial Co.,Ltd
Adequate stocks available,prompt shipment,Strictly control on quality,Timely after-sales Application:API
Cas:7783-55-3
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHebei Ruishun Trade Co.,Ltd
Supply top quality products with a reasonable price Application:api
Chemical Co.Ltd
Phosphorus(III) fluoride, 99%Appearance:Off white to slight yellow solid Storage:Stored in shaded, cool and dry places Package:1L 5L 10L 25L bottle Application:pharma intermediate Transportation:Handle with cares to avoid damaging the packages. Prote
Synthetic route
-
-
1279123-63-5
potassium hydrogenfluoride

-
-
7789-60-8
phosphorus tribromide

-
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In acetonitrile byproducts: KBr, HBr; PBr3 in MeCN was slowly added to a boiling suspn. of KHF2 in MeCN;; the gaseous prod. was condensed in trap cooled by liq. N2 and distd. at -100°C; elem. anal.; IR; | 90% |
-
-
1279123-63-5
potassium hydrogenfluoride

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In acetonitrile byproducts: KCl, HCl; PCl3 in MeCN was slowly added to a boiling suspn. of KHF2 in MeCN;; the gaseous prod. was condensed in trap cooled by liq. N2 and distd. at -100°C; elem. anal.; IR; | 87.7% |
-
-
14791-57-2
silyl bromide

-
-
34326-59-5
bis(difluorophosphino)amine

-
-
75-50-3
trimethylamine

-
A
-
11128-24-8
fluorosilane

-
B
-
71579-72-1
bis(difluorophosphine)silylamine

-
C
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: (NHMe3)Br; absence of air and moisture (vac. line); condensing NMe3 onto mixt. of NH(PF2)2 and SiBrH3, warming to room temp. for 15 s; fractional distn.; | A n/a B 87% C n/a |


-
-
13455-01-1
phosphorous triiodide

-
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In pyridine | 69% |
-
-
14335-40-1
chlorodifluorophosphine

-
-
7664-41-7
ammonia

-
A
-
25757-74-8
aminodifluorophosphine

-
-
60448-09-1
diaminodifluorophosphorane

-
C
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In neat (no solvent) expanding excess of NH3 into PClF2, standing for 3 min; removal of volatiles, fractional low-temp. distn. (vac., trap at 227 K);mass spectroscopy; | A n/a B 42% C n/a |


| Conditions | Yield |
|---|---|
| In melt passing PCl3 vapour into a melt of alkali fluorides at 500°C;; | 20% |

| Conditions | Yield |
|---|---|
| 500°C, closed reactor; | 5% |
| In not given heating red P with Hf under pressure;; |

-
-
13465-66-2, 271781-78-3
selenium(IV) fluoride

-
A
-
7446-08-4
selenium(IV) oxide

-
B
-
13478-20-1
trifluorophosphoric acid

-
C
-
1087413-67-9
nitrosylsulfuric acid fluoride

-
D
-
7783-55-3
trifluorophosphane


-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
A
-
15597-63-4
phosphorus fluoride dichloride

-
B
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In neat (no solvent) refluxing with dry NH4F;; | |
| In neat (no solvent) heating PCl3 with dry NH4F with a reflux condenser;; | |
| In neat (no solvent) refluxing with dry NH4F;; | |
| In neat (no solvent) heating PCl3 with dry NH4F with a reflux condenser;; |


-
-
7647-14-5
sodium chloride

-
A
-
13478-20-1
trifluorophosphoric acid

-
B
-
13769-76-1
phosphoryl dichloride fluoride

-
C
-
13769-75-0
phosphorus oxydifluorochloride

-
D
-
7783-55-3
trifluorophosphane

-
E
-
10025-87-3, 12599-09-6, 63736-95-8
trichlorophosphate

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating with CaF2 in presence of NaCl;; | |
| In neat (no solvent) heating with CaF2 in presence of NaCl;; |


| Conditions | Yield |
|---|---|
| at red heat; | 0% |

-
-
13454-99-4
dichloro-trifluoro-phosphorane

-
A
-
7783-55-3
trifluorophosphane

-
B
-
7719-12-2, 52843-90-0
phosphorus trichloride

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating PF3Cl2 with P at 120 °C;; | |
| In neat (no solvent) decompn. with P at 110 °C;; |


-
-
13454-99-4
phosphorus dichloride trifluoride

-
A
-
7783-55-3
trifluorophosphane

-
B
-
7719-12-2, 52843-90-0
phosphorus trichloride


| Conditions | Yield |
|---|---|
| In neat (no solvent) at 180°C;; |

-
-
1087413-67-9
nitrosylsulfuric acid fluoride

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
A
-
16921-91-8
nitrosyl hexafluorophosphate

-
B
-
7446-09-5
sulfur dioxide

-
C
-
10102-43-9
nitrogen(II) oxide

-
D
-
2696-92-6
nitrosylchloride

-
E
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In not given react. with FSO2NO; formation of NO(PF6), NO, NOCl, SO2 and PF3;; | |
| In not given react. with FSO2NO; formation of NO(PF6), NO, NOCl, SO2 and PF3;; |

-
-
7446-08-4
selenium(IV) oxide

-
-
7664-39-3
hydrogen fluoride

-
A
-
13478-20-1
trifluorophosphoric acid

-
B
-
7783-55-3
trifluorophosphane


| Conditions | Yield |
|---|---|
| High Pressure; 200°C, conversion increases in presence of alkali or alkaline earth fluride; | A n/a B 23-27 |

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
7783-56-4
antimony(III) fluoride

-
-
7647-19-0, 874483-74-6
phosphorus pentafluoride

-
B
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: phosphorus fluoride chloride; react. of PCl5 with SbF3;; | |
| In neat (no solvent) vigorous react. of solid PCl5 with SbF3;; | |
| In neat (no solvent) byproducts: phosphorus fluoride chloride; react. of PCl5 with SbF3;; |

-
-
14913-58-7
antimony tetrachloride fluoride

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
A
-
18460-56-5
{PCl4}(1+)*{SbCl6}(1-)={PCl4}{SbCl6}

-
B
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In not given react. with SbCl4F;; | |
| In not given react. with SbCl4F;; |

-
-
7783-70-2
antimony pentafluoride

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| byproducts: HCl; at room temp.; | |
| at low temp.; | 0% |
| In not given byproducts: HCl; vigorous react. of PCl3 with SbF5 at ambient temp.;; | |
| byproducts: HCl; at room temp.; | |
| at low temp.; | 0% |
-
-
7783-77-9
molybdenum(VI) fluoride

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
A
-
13819-84-6, 194303-41-8
molybdenum(V) fluoride

-
-
7647-19-0, 874483-74-6
phosphorus pentafluoride

-
C
-
7782-50-5
chlorine

-
D
-
7783-55-3
trifluorophosphane

-
E
-
13320-71-3
molybdenum(IV) chloride

| Conditions | Yield |
|---|---|
| In not given react. with an excess of MoF6 forming PF3, MoCl5 and Cl2; react. of MoCl5 forming MoCl4 and Cl2; further react. of MoF6 with PF3 forming PF5 and MoF5;; | |
| In not given react. with an excess of MoF6 forming PF3, MoCl5 and Cl2; react. of MoCl5 forming MoCl4 and Cl2; further react. of MoF6 with PF3 forming PF5 and MoF5;; |

-
-
7783-77-9
molybdenum(VI) fluoride

-
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
A
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
B
-
10241-05-1
molybdenum(V) chloride

-
C
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In not given react. of an excess of PCl3 with MoF6 forming PF3, MoCl5, Cl2; further react. of an excess of PCl3 with Cl2 forming PCl5;; | |
| In not given react. of an excess of PCl3 with MoF6 forming PF3, MoCl5, Cl2; further react. of an excess of PCl3 with Cl2 forming PCl5;; |


| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: O2, P; decompn. of PF3 by SiO2;; | |
| In neat (no solvent) byproducts: O2, P; decompn. of PF3 by SiO2;; |

-
A
-
13478-20-1
trifluorophosphoric acid

-
B
-
124-38-9
carbon dioxide

-
C
-
7783-61-1
silicon tetrafluoride

-
D
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In neat (no solvent) heating at 700 °C;; | |
| In neat (no solvent) heating at 700 °C;; |

-
-
14335-40-1
chlorodifluorophosphine

-
A
-
14984-74-8
difluorophosphine

-
B
-
7803-51-2
phosphan

-
C
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran under inert atmosphere, equimolar amt. of PF2Cl was reacted with L-Selectride in THF at temperature from -100 to -90°C; product contained other unidentified PF2-containing species; | A 0% B n/a C n/a |


| Conditions | Yield |
|---|---|
| by heating, mol. ratio 3:1; | |
| by heating, mol. ratio 3:1; |

-
-
14335-40-1
chlorodifluorophosphine

-
-
67276-04-4
sodium tri-sec-butylborohydride

-
A
-
14984-74-8
difluorophosphine

-
B
-
7803-51-2
phosphan

-
C
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran under inert atmosphere, equimolar amt. of PF2Cl was reacted with N-Selectride in THF at temperature from -100 to -90°C; product contained other unidentified PF2-containing species; | A 0% B n/a C n/a |

-
-
14335-40-1
chlorodifluorophosphine

-
A
-
14984-74-8
difluorophosphine

-
B
-
7803-51-2
phosphan

-
C
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In tetrahydrofuran under inert atmosphere, equimolar amt. of PF2Cl was reacted with K-Selectride in THF at temperature from -100 to -90°C; product contained other unidentified PF2-containing species; | A 0% B n/a C n/a |


| Conditions | Yield |
|---|---|
| with excess of P PF3 developing; |

-
-
12185-10-3
phosphorus

-
-
13465-66-2, 271781-78-3
selenium(IV) fluoride

-
A
-
7782-49-2
selenium

-
B
-
7783-55-3
trifluorophosphane

-
-
7783-55-3
trifluorophosphane

-
-
13454-99-4
phosphorus dichloride trifluoride

| Conditions | Yield |
|---|---|
| With chlorine In neat (no solvent) react. at ambient temp. in the gas phase; light sensitive;; | 99% |
| With Cl2 In neat (no solvent) react. at ambient temp. in the gas phase; light sensitive;; | 99% |
| With chlorine In neat (no solvent) heating; volume contraction;; | |
| With Cl2 In neat (no solvent) heating; volume contraction;; |

-
-
7783-55-3
trifluorophosphane

-
-
70620-83-6
η5-indenylbis(trifluorophosphine)rhodium(I)

| Conditions | Yield |
|---|---|
| In neat (no solvent) byproducts: CH2CH2; N2 atmosphere; heating in evacuated ampoule from -196°C to room temp.; fractionation; | 99% |

-
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In pentane PF3 bubbled through soln. of Rh complex in pentane for 2-4 min; concd., chromd. (SiO2, hexane/CH2Cl2 5/3); elem. anal.; | 97% |

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 0.5h; Inert atmosphere; Schlenk technique; | 96% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) 3.0mmol (OC)BH3, 2.5mmol, F2PX, -78°C, 2-3d;; very pure product;; | A 95% B n/a |
| In neat (no solvent) 3.0mmol (OC)BH3, 2.5mmol, F2PX, -78°C, 2-3d;; very pure product;; | A 95% B n/a |


| Conditions | Yield |
|---|---|
| In diethyl ether PF3 passed over soln. of ((C3H5)3WCl)2 in ether (under Ar, -50°C); | 95% |
-
-
311819-28-0, 25087-75-6, 55354-87-5
RuH(OAc)(PPh3)3

-
-
7783-55-3
trifluorophosphane

-
-
67840-34-0
tris(trifluorophosphine)bis(triphenylphosphine)ruthenium(0)

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: CH3COOH; a mixt. of Ru-complex, ligand and CH2Cl2 was sealed off in an ampoule and warmed from -196°C to room temp., the mixt. was shaken overnight; evapd., washed with hexane, crystd. from CH2Cl2/hexane; elem. anal.; | 93.6% |
-
-
7440-44-0
pyrographite

-
-
7783-55-3
trifluorophosphane

-
A
-
116-14-3
polytetrafluoroethylene

-
B
-
75-73-0
carbon tetrafluoride

| Conditions | Yield |
|---|---|
| In neat (no solvent) react. of C with PF3 in a Ni-tube at 1100°C;; | A 90% B 9% |


-
-
7783-55-3
trifluorophosphane

-
-
115805-07-7
IrH2(P(C6H5)3)2(PF3)(1+)

| Conditions | Yield |
|---|---|
| In neat (no solvent) 300 mm of PF3, 16 h, room temp.; | 90% |
-
-
1449195-56-5
dimethyl(7-phenylnorbornadiene)platinum(II)

-
-
7783-55-3
trifluorophosphane

-
-
1262969-68-5, 1263030-29-0
cis-dimethylbis(trifluorophosphane)platinum(II)

| Conditions | Yield |
|---|---|
| In Dimethyl ether at -196 - 20℃; for 1h; Glovebox; | 89% |

| Conditions | Yield |
|---|---|
| In hexane Irradiation (UV/VIS); 90 min; recrystn. from hexane at -20°C, elem. anal.; | 86% |
-
-
93842-30-9
chlorobis[[(diphenylphosphino)ethyl]dimethylsilyl]iridium(III)

-
-
7783-55-3
trifluorophosphane

-
-
93842-42-3
[Ir((C6H5)2PCH2CH2Si(CH3)2)2(Cl)(PF3)]

| Conditions | Yield |
|---|---|
| In tetrahydrofuran PF3 was bubbled through a soln. of complex; solvent was removed, hexane was added; elem.anal.; | 81% |

| Conditions | Yield |
|---|---|
| In benzene N2-atmosphere; addn. of phosphine to AuCl suspn.; pptn. on pentane addn., filtration, washing (pentane), drying (vac.); | 80% |
| In toluene (N2; Ar); PF3 condensed into suspension of AuCl in toluene; cooled at -65 °C for 2 days; filtrated; pentane added; crystals precipitated at 6°C; |

| Conditions | Yield |
|---|---|
| In tetradecane at 150℃; under 7.60051 - 760.051 Torr; for 6.5h; Inert atmosphere; | 78% |


-
-
7783-55-3
trifluorophosphane

-
-
823786-32-9
trans-[iron(II) hydride (PF3) bis[1,2-bis(diphenylphosphanyl)ethane]] tetrafluoroborate

| Conditions | Yield |
|---|---|
| In dichloromethane (H2), Fe complex in CH2Cl2 treated with PF3 gas for 10 min, stirred at room temp. for 2 h; concd., pptd.(Et2O), dried (vac.), recrystd.(CH2Cl2/Et2O) for several d; | 75% |

-
-
7783-55-3
trifluorophosphane

-
-
823786-36-3
trans-[ruthenium(II) hydride (PF3) bis[1,2-bis(diphenylphosphanyl)ethane]] tetrafluoroborate

| Conditions | Yield |
|---|---|
| In dichloromethane (H2), Ru complex in CH2Cl2 treated with PF3 gas for 10 min, stirred at room temp. for 2 h; concd., pptd.(Et2O), dried (vac.), recrystd.(CH2Cl2/Et2O) for several d; | 73% |

-
-
7783-55-3
trifluorophosphane

| Conditions | Yield |
|---|---|
| In benzene Ar, a slow stream PF3 passed through the soln. at room temp. for 20 min,stirred for 20 min at room temp.; solvent removed (vac.), recrysttd. (acetone, -78°C), crysts. separated, dried (vac.); elem. anal.; | 72% |
-
-
17742-04-0
trifluoromethylsulfenyl fluoride

-
-
7783-55-3
trifluorophosphane

-
-
66604-83-9
trifluoromethylsulfanyltetrafluorophosphorane

| Conditions | Yield |
|---|---|
| -30°C, in vac. apparatus of nickel or stainless steel; | 70% |
| -30°C, in vac. apparatus of nickel or stainless steel; | 70% |

| Conditions | Yield |
|---|---|
| In thionyl chloride PF3 condensed onto Au2Cl6 in SOCl2, stirred overnight at room temp. without light or treated 2.5 h with ultrasonics; soln. cooled to -20. - -80°C, filtered, crystals washed with SOCl2; | 70% |

| Conditions | Yield |
|---|---|
| With C4H8O In tetrahydrofuran Irradiation (UV/VIS); (N2); soln. of Cr complex was irradiated for 1 h, then PF3 was bubbled through the soln. for 10 min; solvent was removed in vac., residue chromd. (silicagel, hexane); identified by MAS; | 70% |

-
-
7783-55-3
trifluorophosphane

-
-
823786-34-1
trans-[ruthenium(II) hydride (PF3) bis[1,2-bis(diphenylphosphanyl)methane]] tetrafluoroborate

| Conditions | Yield |
|---|---|
| In dichloromethane (H2), Ru complex in CH2Cl2 treated with PF3 gas for 10 min, stirred at room temp. for 2 h; concd., pptd.(Et2O), dried (vac.), recrystd.(CH2Cl2/Et2O) for several d; | 69% |

-
-
7783-55-3
trifluorophosphane

-
-
342573-38-0
trans-chlorobis(triisopropylstibine)(trifluorophosphine)rhodium(I)

| Conditions | Yield |
|---|---|
| In benzene Ar, a slow stream of PF3 passed through the soln. for 20 min at room temp.; solvent removed (vac.); elem. anal.; | 68% |

-
-
7783-55-3
trifluorophosphane

-
-
342573-37-9
trans-chlorobis(triisopropylphosphine)(trifluorophosphine)rhodium(I)

| Conditions | Yield |
|---|---|
| In benzene Ar, a slow stream of PF3 passed through the soln. for 20 min at room temp.; solvent removed (vac.), recrystd. (acetone, -60°C), solid separate, dried (vac.); elem. anal.; | 68% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View

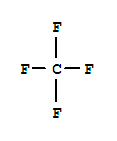






 T
T