Henan Allgreen Chemical Co.,Ltd
he company has advanced technology, as well as a large number of excellent R & D team, to provide customers from the grams to one hundred kilograms and tons of high-quality products, competitive prices and quality se T rvice Appearance:Whit
Ality Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:927-74-2
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryAnhui Dexinjia Biopharm Co., Ltd
As a leading and professional manufacturer of APIs and API intermediates in China,Anhui Dexinjia Biopharm Co., Ltd founded in 2006, Except for the R&D center, our company has built a close cooperation relation with Chinese Academy of Sciences, T
Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem’s R&D center offer custom synthesis according to the contract research and development services for t
Cas:927-74-2
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Hebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufactu
Cas:927-74-2
Min.Order:1 Kilogram
FOB Price: $1.0 / 10.0
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
CAS No.927-74-2 Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scie
Cas:927-74-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:927-74-2
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryLIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the R&
Cas:927-74-2
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHebei Sankai Chemical Technology Co., Ltd
1. Product advantages ♦ High purity, all above 98.5%, no impurities after dissolution ♦ We will test each batch to ensure quality ♦ OEM and private brand services designed for free ♦ Various cap colors available ♦ W
Cas:927-74-2
Min.Order:1 Kilogram
FOB Price: $290.0 / 499.0
Type:Trading Company
inquiryHenan Sinotech Import&Export Corporation
Product Name: 3-Butyn-1-ol Synonyms: 1-Butyn-4-ol;3-butyn-1-0l;3-Butynol;3-Butynyl alcohol;4-Hydroxy-1-butyne;HCequivCCH2CH2OH;3-BUTYNE-1-OL;3-BUTYN-1-OL CAS: 927-74-2 MF
Shanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:927-74-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryShanghai Minstar Chemical Co., Ltd
Product Name: 3-Butyn-1-ol CAS: 927-74-2 MF: C4H6O MW: 70.09 EINECS: 213-161-9 Mol File: 927-74-2.mol 3-Butyn-1-ol Structure 3-Butyn-1-ol Chemical Properties Melting point -63.6 °C (lit.) Boiling point 128.9 °C (li
Qingdao Beluga Import and Export Co., LTD
3-Butyn-1-ol CAS:927-74-2 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates,
Cas:927-74-2
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Changchun Artel lmport and Export trade company
Superiority We can customize and synthesize products that other suppliers may not be able to provide. Advantage cof, mof ligand manufacturer Product Detail bes
Cas:927-74-2
Min.Order:100 Kilogram
Negotiable
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Wuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
SHANGHAI T&W PHARMACEUTICAL CO., LTD.
A substitute for perfluorooctanoic acid, mainly used as a surfactant, dispersant, additive, etc Appearance:White solid or Colorless liquid Purity:99.3 % We will ship the goods in a timely manner as required We can provide relevant documents acc
Hangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Shaanxi Cuicheng Biomedical Technology Co., Ltd.
Why Choose Us: 1. Factory direct sales, so we can provide the competitive price and high quality product base on 8 years of production and R&D experience. 2. It is available in stock for quick shipment.Products could be packaged according to cu
Chemlyte Solutions
Chemlyte Solutions believe that customers and suppliers deserve much more than what traditional distributors can offer. To grow in today s fast-paced and increasingly competitive market it is essential to be able to quickly adapt to market forces eff
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:927-74-2
Min.Order:10 Gram
Negotiable
Type:Lab/Research institutions
inquirySHANGHAI SYSTEAM BIOCHEM CO., LTD
We are one of a few suppliers that can offer custom synthesis service of this product We are specialized in custom synthesis, chemical/pharmaceutical/ pesticides outsourcing and contract research. We are committed to prov
Cas:927-74-2
Min.Order:100 Gram
FOB Price: $100.0 / 2000.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:colourless liquid Storage:stored in a cool, dry and ventilated place to provent sun and rain Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical Interme
TaiChem Taizhou Limited
Established in May 2015, TaiChem Ltd. is initially invested by a British research and development company and started by PhDs back from aboard. The company is registered in China Medical City (CMC), Taizhou, Jiangsu Province, and the production site
Siwei Development Group Ltd.
Product name: 3-Butyn-1-ol CAS No.:927-74-2 Molecule Formula:C4H6O Molecule Weight:70.09 Purity: 97.0% Package: 200kg/drum Description:Colorless or light yellow liquid Manufacture Standards:Enterprise Standard TESTING ITEMS
Cas:927-74-2
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquirySynthetic route
-
-
40365-61-5
2-(but-3-yn-1-yloxy)tetrahydropyran

-
A
-
6581-66-4
2-methoxytetrahydropyran

-
B
-
927-74-2
1-butyn-4-ol

| Conditions | Yield |
|---|---|
| With methanol; DOWEX 50 W-X2 cation exchange resin at 40℃; for 1h; | A n/a B 98% |

-
-
927-74-2
1-butyn-4-ol

| Conditions | Yield |
|---|---|
| With zinc diacetate In methanol; dichloromethane at 20℃; for 1.5h; | 97% |
-
-
75014-48-1
4-<(triphenylmethyl)oxy>-1-butyne

-
-
927-74-2
1-butyn-4-ol

| Conditions | Yield |
|---|---|
| With indium(III) chloride; water In acetonitrile for 2h; Heating; | 93% |
-
-
40365-61-5
2-(but-3-yn-1-yloxy)tetrahydropyran

-
-
927-74-2
1-butyn-4-ol

| Conditions | Yield |
|---|---|
| With silica-supported NaHSO4 In methanol at 20℃; for 0.25h; | 91% |
| With zinc(II) trifluoromethanesulfonate In methanol at 20℃; for 0.583333h; | 87% |
| With pyridinium p-toluenesulfonate In ethanol at 25℃; for 3h; |
-
-
78592-82-2
4-(tert-butyldimethylsilyloxy)-1-butyne

-
-
927-74-2
1-butyn-4-ol

| Conditions | Yield |
|---|---|
| With sodium hydrogen sulfate; silica gel In dichloromethane at 20℃; for 0.5h; | 91% |
| With pyridinium hydrobromide perbromide In methanol at 0℃; | 81% |

-
-
927-74-2
1-butyn-4-ol

| Conditions | Yield |
|---|---|
| With cerium(III) chloride In acetonitrile for 1.5h; Heating; | 90% |

| Conditions | Yield |
|---|---|
| In ammonia at -40℃; | 88% |
| In ammonia for 24h; | 80% |
-
-
218434-90-3
1-but-3-ynyloxymethyl-4-methoxy-benzene

-
-
927-74-2
1-butyn-4-ol

| Conditions | Yield |
|---|---|
| With carbon tetrabromide In methanol for 3h; ether cleavage; Heating; | 86% |

| Conditions | Yield |
|---|---|
| Multistep reaction; | 75% |

| Conditions | Yield |
|---|---|
| With bis(acetylacetonate)oxovanadium; methyl 3,5-bis((1H-1,2,4-triazol-1-yl)methyl)benzoate; oxygen; sodium acetate at 120℃; for 48h; | A 66% B 54% |

| Conditions | Yield |
|---|---|
| With aluminium; mercury dichloride In tetrahydrofuran | 55% |
| With zinc In tetrahydrofuran Addition; |

| Conditions | Yield |
|---|---|
| With ammonia | |
| With ammonia |
-
-
75-21-8
oxirane

-
-
1066-26-8
sodium acetylide

-
A
-
36697-85-5
2-but-3-ynyloxy-ethanol

-
B
-
927-74-2
1-butyn-4-ol

| Conditions | Yield |
|---|---|
| With ammonia |


| Conditions | Yield |
|---|---|
| With sodium |
-
-
76334-36-6
3-bromobut-3-en-1-ol

-
-
927-74-2
1-butyn-4-ol

| Conditions | Yield |
|---|---|
| With potassium hydroxide |

| Conditions | Yield |
|---|---|
| beim Behandeln von Natriumacetylenid; | |
| beim Behandeln von Natriumacetylenid; |



| Conditions | Yield |
|---|---|
| Stage #1: acetylene With acetylenemagnesium bromide In tetrahydrofuran at -10 - 10℃; for 0.5h; Stage #2: oxirane In tetrahydrofuran at 35 - 40℃; |

-
-
2134-29-4
3-Hydroxypropanal

-
-
90965-06-3
dimethyl 1-(1-diazo-2-oxopropyl)phosphonate

-
-
927-74-2
1-butyn-4-ol

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol at 20℃; for 5.5h; Seyferth-Gilbert Homologation; |

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane for 16h; Ambient temperature; | 100% |
| With pyridinium p-toluenesulfonate In dichloromethane | 99% |
| With toluene-4-sulfonic acid In dichloromethane at 0 - 23℃; for 1.08333h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| In pyridine; benzene at 0℃; for 14h; | 100% |
| With pyridine In dichloromethane at 0℃; | 100% |
| With pyridine In dichloromethane at 23℃; | 100% |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; silver nitrate In acetone at 23℃; for 2h; Inert atmosphere; | 100% |
| With N-Bromosuccinimide; silver nitrate In acetone at 20℃; for 2h; Inert atmosphere; | 98% |
| With N-Bromosuccinimide; silver nitrate In acetone at 25℃; for 1h; | 95% |

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide; sodium hydride In trichlorophosphate Heating; | 100% |
| With tetra-(n-butyl)ammonium iodide; sodium hydride In tetrahydrofuran at 23℃; for 5.5h; | 100% |
| With tetra-(n-butyl)ammonium iodide; sodium hydride In tetrahydrofuran; mineral oil at 0 - 23℃; for 18h; Inert atmosphere; Sealed tube; | 100% |
-
-
124-63-0
methanesulfonyl chloride

-
-
927-74-2
1-butyn-4-ol

-
-
72486-09-0
methanesulfonic acid but-3-ynyl ester

| Conditions | Yield |
|---|---|
| With triethylamine at 0 - 20℃; | 100% |
| With triethylamine In dichloromethane at 0℃; for 3h; | 99% |
| With triethylamine In dichloromethane at 0℃; for 1.25h; Inert atmosphere; | 99.8% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Butyn-1-ol With n-butyllithium In tetrahydrofuran; hexane at -78 - 20℃; for 1h; Stage #2: chloro-trimethyl-silane In tetrahydrofuran; hexane at -78 - 20℃; for 2h; | 100% |
| Stage #1: 3-Butyn-1-ol With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Schlenk technique; Inert atmosphere; Stage #2: chloro-trimethyl-silane In tetrahydrofuran; hexane at -78 - 20℃; for 2h; Schlenk technique; Inert atmosphere; | 100% |
| Stage #1: 3-Butyn-1-ol With n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: chloro-trimethyl-silane In tetrahydrofuran; hexane at -78 - 20℃; for 2h; | 98% |
-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
927-74-2
1-butyn-4-ol

-
-
78592-82-2
4-(tert-butyldimethylsilyloxy)-1-butyne

| Conditions | Yield |
|---|---|
| With dmap; triethylamine Substitution; Silylation; | 100% |
| With 1H-imidazole In tetrahydrofuran at 20℃; for 3h; | 100% |
| With 1H-imidazole In diethyl ether at 100℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With sodium hydride In tert-butyl methyl ether at -5 - 20℃; for 16.25h; | 100% |
| With sodium hydride In tetrahydrofuran; diethyl ether | 70% |
| With sodium hydride 1.) Et2O, HMPA, RT, 30 min, 2.) Et2O, HMPA, reflux, 2.5 h; Yield given. Multistep reaction; | |
| Stage #1: 3-Butyn-1-ol With sodium hydride In tetrahydrofuran; mineral oil at 0℃; Inert atmosphere; Stage #2: allyl bromide With tetra-(n-butyl)ammonium iodide In tetrahydrofuran; hexane at 20℃; Inert atmosphere; | |
| Stage #1: 3-Butyn-1-ol With sodium hydride In tetrahydrofuran; mineral oil at 0℃; for 0.666667h; Inert atmosphere; Schlenk technique; Stage #2: allyl bromide With tetra-(n-butyl)ammonium iodide In tetrahydrofuran; mineral oil at 0 - 25℃; for 16h; Inert atmosphere; Schlenk technique; |
-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
927-74-2
1-butyn-4-ol

-
-
88158-68-3
4-(tert-butyldiphenylsilyloxy)-1-butyne

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 20℃; for 2h; | 100% |
| With 1H-imidazole In dichloromethane at 20℃; for 2h; | 100% |
| With 1H-imidazole In N,N-dimethyl-formamide for 0.5h; Ambient temperature; | 100% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); N-butylamine In benzene at 20℃; for 12h; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); N-butylamine In benzene for 16h; | 99% |
| Stage #1: cis-1,2-Dichloroethylene With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); N-butylamine In benzene for 0.416667h; Condensation; Stage #2: 3-Butyn-1-ol In benzene at 20℃; for 3h; Condensation; | 80% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); triethylamine for 16h; Sonogashira Cross-Coupling; Reflux; Inert atmosphere; Schlenk technique; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; for 10h; Inert atmosphere; | 99% |
| With copper(l) iodide; potassium carbonate; bis(η3-allyl-μ-chloropalladium(II)); Tedicyp In N,N-dimethyl-formamide at 100℃; Sonogashira reaction; | 97% |
-
-
927-74-2
1-butyn-4-ol

-
-
2873-29-2
D-glucal triacetate

-
-
135195-22-1, 142344-14-7
but-3-yn-1-yl 4,6-di-O-acetyl-2,3-dideoxy-α/β-D-erythro-hex-2-enopyranoside

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In dichloromethane Ambient temperature; | 100% |
| With 3-butyl-1-methyl-1H-imidazol-3-ium hexafluorophosphate; dysprosium(III) trifluoromethanesulfonate at 20℃; for 2h; | 90% |
-
-
176168-47-1
3-iodo-4-phenylthiophene

-
-
927-74-2
1-butyn-4-ol

| Conditions | Yield |
|---|---|
| With triethylamine; copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) In acetonitrile Heating; | 100% |
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0) In triethylamine; acetonitrile for 18h; Heating; | 100% |
-
-
927-74-2
1-butyn-4-ol

-
-
211029-67-3
(3-iodo-pyridin-4-yl)-carbamic acid tert-butyl ester

| Conditions | Yield |
|---|---|
| With copper(l) iodide; triethylamine; bis-triphenylphosphine-palladium(II) chloride at 20℃; | 100% |
-
-
927-74-2
1-butyn-4-ol

-
-
87600-97-3
4,6-dichloropyrimidine-5-carboxylic acid chloride

-
-
285991-67-5
4,6-dichloro-pyrimidine-5-carboxylic acid but-3-ynyl ester

| Conditions | Yield |
|---|---|
| With triethylamine at 0 - 20℃; | 100% |
| With triethylamine Substitution; | 87% |
-
-
927-74-2
1-butyn-4-ol

-
-
278167-18-3
N,N-dimethyl 3-trimethylsilyl-4-iodo-1H-pyrrole-1-sulfonamide

| Conditions | Yield |
|---|---|
| With copper(l) iodide; diethylamine; bis-triphenylphosphine-palladium(II) chloride at 20℃; for 7h; cross-coupling; Sonogashira reaction; | 100% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; trans-bis(triphenylphosphine)palladium dichloride; triethylamine at 0℃; for 24h; Sonogashira coupling; Inert atmosphere; Reflux; | 100% |
| With tris-(dibenzylideneacetone)dipalladium(0); triethylamine; triphenylphosphine; copper(l) iodide at 70℃; for 24h; | 82% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; for 8h; Sonogashira Cross-Coupling; Inert atmosphere; | 73% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 20℃; for 12h; Inert atmosphere; | |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine at 117℃; for 3h; Sonogashira Cross-Coupling; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; triethylamine; tetrakis(triphenylphosphine) palladium(0) In dichloromethane at 50℃; Sonogashira reaction; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran for 4h; Inert atmosphere; Reflux; | 85% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide Schlenk technique; | 82% |
-
-
1122-91-4
4-bromo-benzaldehyde

-
-
927-74-2
1-butyn-4-ol

-
-
544707-13-3
4-(4-hydroxy-1-butyn-1-yl)benzaldehyde

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 20℃; | 100% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 20℃; for 48h; Sonogashira-Hagihara coupling; | 91% |
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide In tetrahydrofuran at 20℃; Sonogashira Cross-Coupling; |
-
-
530-62-1
1,1'-carbonyldiimidazole

-
-
927-74-2
1-butyn-4-ol

-
-
909707-34-2
but-3-yn-1-yl 1H-imidazole-1-carboxylate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; dichloromethane at 20℃; for 2h; | 100% |
| In dichloromethane at 0 - 20℃; for 17h; Inert atmosphere; | 88% |
-
-
2296-23-3
2-iodo-4-cyanophenol

-
-
927-74-2
1-butyn-4-ol

-
-
919088-04-3
2-(2-hydroxy-ethyl)-benzofuran-5-carbonitrile

| Conditions | Yield |
|---|---|
| With bis-triphenylphosphine-palladium(II) chloride; copper(l) iodide; triethylamine In tetrahydrofuran at 20℃; for 15h; | 100% |
-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
927-74-2
1-butyn-4-ol

-
-
190774-49-3
but-3-ynyl-1-oxycarbonyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: bis(trichloromethyl) carbonate; 3-Butyn-1-ol In tetrahydrofuran at 0 - 20℃; Stage #2: With pyridine In tetrahydrofuran at 0 - 20℃; for 2h; | 100% |
| In diethyl ether for 12h; | |
| In tetrahydrofuran for 12h; Cooling with ice; | |
| With sodium carbonate In tetrahydrofuran at 0 - 20℃; for 12h; |

| Conditions | Yield |
|---|---|
| With triethylamine; palladium diacetate; copper(l) iodide; triphenylphosphine at 20℃; for 3h; | 100% |
-
-
27607-77-8
trimethylsilyl trifluoromethanesulfonate

-
-
927-74-2
1-butyn-4-ol

-
-
58434-99-4
1-<(trimethylsilyl)oxy>-4-(trimethylsilyl)-3-butyne

| Conditions | Yield |
|---|---|
| With zinc trifluoromethanesulfonate; triethylamine In dichloromethane at 23℃; for 12h; | 100% |
-
-
191790-03-1
(((C6H5)2PCH2)2)Pt(CH3CN)(CH(Cl)COOC2H5)(1+)*BF4(1-)=[(((C6H5)2PCH2)2)Pt(CH3CN)(CH(Cl)COOC2H5)]BF4

-
-
927-74-2
1-butyn-4-ol

-
-
191790-05-3
(((C6H5)2PCH2)2)Pt(C4H6O)(CH(Cl)COOC2H5)(1+)*BF4(1-)=[(((C6H5)2PCH2)2)Pt(C4H6O)(CH(Cl)COOC2H5)]BF4

| Conditions | Yield |
|---|---|
| In dichloromethane 20°C (15 h); | 100% |
-
-
848440-23-3
(3-bromo-2-fluorophenyl)carbamic acid tert-butyl ester

-
-
927-74-2
1-butyn-4-ol

-
-
1099766-43-4
tert-butyl [3-(4-hydroxybut-1-yn-1-yl)-2-fluorophenyl]carbamate

| Conditions | Yield |
|---|---|
| With copper(l) iodide; tetrakis(triphenylphosphine) palladium(0); diisopropylamine for 3h; Reflux; | 100% |
-
-
927-74-2
1-butyn-4-ol

-
-
1050479-30-5
sodium but-3-yn-1-yl sulfate

| Conditions | Yield |
|---|---|
| With pyridine; chlorosulfonic acid In tetrachloromethane at 0 - 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 20℃; for 0.5h; Mitsunobu reaction; | 100% |
-
-
112543-78-9
(1R,2R,3S,4R)-4-azido-1-<((tert-butyldiphenylsilyl)oxy)methyl>-2,3-(isopropylidenedioxy)cyclopentane

-
-
927-74-2
1-butyn-4-ol

-
-
1161113-46-7
(1R,2S,3R,4R)-4-[(tert-butyldiphenylsilyloxy)methyl]-1-[4-(2-hydroxyethyl)-1H-1,2,3-triazol-1-yl]-2,3-isopropylidenedioxycyclopentane

| Conditions | Yield |
|---|---|
| With copper; copper(II) sulfate In water; tert-butyl alcohol at 125℃; for 0.0166667h; Microwave irradiation; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Butyn-1-ol With N,N,N,N,N,N-hexamethylphosphoric triamide; n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Inert atmosphere; Stage #2: C18H33NO3Si In tetrahydrofuran; hexane at -78 - 20℃; for 3h; | 100% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View






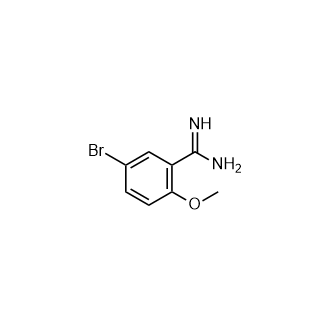









 Xi,
Xi, F
F