-
Name
2-Amino-5-chlorobenzophenone
- EINECS 211-949-7
- CAS No. 719-59-5
- Article Data107
- CAS DataBase
- Density 1.274 g/cm3
- Solubility insoluble
- Melting Point 96-98 °C(lit.)
- Formula C13H10ClNO
- Boiling Point 424.3 °C at 760 mmHg
- Molecular Weight 231.681
- Flash Point 210.4 °C
- Transport Information
- Appearance yellow powder
- Safety 26-36
- Risk Codes 36/37/38-22-20/21/22
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms (2-amino-5-chlorophenyl)(phenyl)methanone;Aminoketone;Benzophenone, 2-amino-5-chloro- (8CI);5-Chloro-2-aminobenzophenone;Benzophenone, 2-amino-5-chloro-;(2-amino-5-chloro-phenyl)-phenyl-methanone;(2-Amino-5-chlorophenyl)phenylmethanone;Methanone, (2-amino-5-chlorophenyl)phenyl-;2-Benzoyl-4-chloroaniline;2-Amino-5-chlorobenzylphenone;
- PSA 43.09000
- LogP 3.73440
Synthetic route

| Conditions | Yield |
|---|---|
| With trimethylsilyl iodide; water In acetonitrile at 20℃; for 0.2h; | 98% |
| Stage #1: 5-chloro-3-phenylanthranil With iron In ethanol for 0.5h; Reflux; Stage #2: With sulfuric acid In ethanol for 1h; Reflux; | 95.1% |
| With aluminium trichloride; zinc In tetrahydrofuran for 6h; Ambient temperature; | 92% |
-
-
5922-60-1
2-amino-5-chlorobenzonitrile

-
-
98-80-6
phenylboronic acid

-
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With 5,5'-dimethyl-2,2'-bipyridine; methanesulfonic acid; palladium(II) trifluoroacetate; water In 2-methyltetrahydrofuran at 80℃; for 36h; Schlenk technique; | 96% |
| With [2,2]bipyridinyl; methanesulfonic acid; palladium(II) trifluoroacetate In tetrahydrofuran; water at 80℃; for 36h; |

-
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With triethylene diamine nickel thiosulfate; water In chloroform at 20℃; for 0.166667h; Hydrolysis; | 92% |
-
-
5922-60-1
2-amino-5-chlorobenzonitrile

-
-
873-55-2
sodium benzenesulfonate

-
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With p-nitrobenzenesulfonic acid; palladium diacetate In tetrahydrofuran; water at 80℃; for 48h; Inert atmosphere; Schlenk technique; | 92% |

| Conditions | Yield |
|---|---|
| Stage #1: benzamide; 4-chloro-aniline With tin(ll) chloride at 123 - 250℃; for 4h; Stage #2: With potassium chloride at 95 - 130℃; for 4h; Stage #3: With phosphoric acid for 23h; Concentration; Temperature; Reflux; | 91% |
-
-
105448-12-2
7-Chlor-2-methylthio-5-phenyl-3H-1,3,4-benzotriazepin

-
A
-
74-93-1
methylthiol

-
B
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 10h; | A n/a B 83.4% |

| Conditions | Yield |
|---|---|
| With gallium(III) trichloride; boron trichloride In chlorobenzene at 100℃; for 20h; | 81% |
| With gallium(III) trichloride; boron trichloride 1.) CH2Cl2, 1,1,2,2-tetrachloroethane, 0 deg C, 2.) 1,1,2,2-tetrachloroethane, 100 deg C, 5 h; Yield given. Multistep reaction; |
-
-
719-64-2
5-chloro-3-phenylanthranil

-
-
98-80-6
phenylboronic acid

-
A
-
719-59-5
2-Amino-5-chlorobenzophenone

-
B
-
108694-41-3
(5-chloro-2-(phenylamino)phenyl)(phenyl)methanone

| Conditions | Yield |
|---|---|
| With potassium carbonate; copper dichloride In dimethyl sulfoxide at 100℃; for 6h; Reagent/catalyst; Solvent; Sealed tube; | A 17% B 81% |


| Conditions | Yield |
|---|---|
| With antimony(III) oxide In dichloromethane at 15℃; for 2h; Temperature; | 78% |
| Stage #1: 4-chloro-aniline; benzoyl chloride With antimony(III) trioxide In dichloromethane at 15℃; for 2h; Friedel-Crafts Acylation; Stage #2: With nitric acid In dichloromethane at 10 - 45℃; for 11.5h; Temperature; | 77% |
| With dysprosium((III) oxide In tetrachloromethane at 15℃; for 2h; Temperature; | 76% |
-
-
86230-82-2
7-chloro-9-phenyl-1,2-dihydrooxazolo<2,3-b>-1,3,4-benzotriazepine

-
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With sulfuric acid In ethanol at 40℃; for 1.16667h; | 75% |
-
-
201230-82-2
carbon monoxide

-
-
63069-48-7
p-chloro-o-iodoaniline

-
-
98-80-6
phenylboronic acid

-
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With potassium carbonate at 100℃; under 2250.23 Torr; for 8h; Catalytic behavior; Suzuki-Miyaura Coupling; Autoclave; | 75% |

-
-
138964-58-6
5-chloro-2-[(phenylsulfonyl)amino]benzoic acid

-
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With dipotassium hydrogenphosphate; [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate; triphenylphosphine In 1,4-dioxane at 25℃; for 8h; Schlenk technique; Inert atmosphere; Sealed tube; Irradiation; | 73% |
-
-
1159372-20-9
(2-amino-5-chlorophenyl)(phenyl)methanol

-
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; oxygen; (R)-1,1'-binaphthyl-2,2'-diamine In toluene at 60℃; for 48h; | 61% |
| With 1,4-diaza-bicyclo[2.2.2]octane; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; oxygen; copper(l) chloride In toluene at 100℃; for 24h; | 58% |
| With copper(II) chloride monohydrate; oxygen; potassium carbonate In N,N-dimethyl-formamide at 60℃; for 24h; | |
| With copper(II) chloride monohydrate; potassium carbonate In N,N-dimethyl-formamide at 60℃; | |
| With copper(II) chloride monohydrate; potassium carbonate In N,N-dimethyl-formamide at 60℃; for 24h; |
-
-
23746-76-1
5-chloro-2-phenylindole

-
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With oxygen; caesium carbonate In dimethyl sulfoxide at 140℃; for 8h; | 59% |
-
-
70437-50-2
7-chloro-3-methyl-2-(methyl-thio)-5-phenyl-3H-1,3,4-benzotriazepine

-
A
-
74-93-1
methylthiol

-
B
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 10h; | A n/a B 57.7% |
-
-
107-10-8
propylamine

-
-
26381-66-8
5-chloro-2-trichloroacetylaminobenzophenone

-
A
-
719-59-5
2-Amino-5-chlorobenzophenone

-
B
-
37103-80-3
syn-5-chloro-2-trichloroacetamidobenzophenone n-propylimine

-
C
-
80170-54-3
6-chloro-3,4-dihydro-4-phenyl-3-(n-propyl)-4-trichloromethyl-2(1H)-quinazolinone

| Conditions | Yield |
|---|---|
| In ethanol at 60℃; for 3h; | A 16% B 57% C 13% |

-
-
1159372-20-9
(2-amino-5-chlorophenyl)(phenyl)methanol

-
C
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; oxygen; copper(II) bis(trifluoromethanesulfonate); (R)-1,1'-binaphthyl-2,2'-diamine In toluene at 60℃; for 24h; optical yield given as %ee; | A n/a B n/a C 57% |

-
-
1159372-20-9
(2-amino-5-chlorophenyl)(phenyl)methanol

-
B
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; oxygen; copper(II) bis(trifluoromethanesulfonate); (R)-1,1'-binaphthyl-2,2'-diamine In toluene at 80℃; for 11h; optical yield given as %ee; | A n/a B 57% |
-
-
26381-66-8
5-chloro-2-trichloroacetylaminobenzophenone

-
-
141-43-5
ethanolamine

-
A
-
719-59-5
2-Amino-5-chlorobenzophenone

-
B
-
27772-51-6
9-chloro-10b-phenyl-2,3,6,19b-tetrahydro-5H-oxazolo<3,2-c>quinazolin-5-one

-
C
-
80171-01-3
6-chloro-3,4-dihydro-3-(2-hydroxyethyl)-4-phenyl-4-trichloromethyl-2(1H)-quinazolinone

| Conditions | Yield |
|---|---|
| In ethanol for 3h; Heating; | A 17% B 43% C 38% |

-
-
67863-85-8
3-Amino-6-chlor-4-hydroxy-4-phenyl-2-thioxo-1,2,3,4-tetrahydrochinazolin

-
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 10h; Heating; | 41.5% |
| Multi-step reaction with 2 steps 1: 1) CH3ONa, 2) HCl (g) / 1) acetone, 7 d, RT, 2) benzene 2: 5 M HCl / Heating; hydrolysis View Scheme |
-
-
112608-11-4
3-Amino-6-chlor-4-hydroxy-2-methylthio-4-phenyl-3,4-dihydrochinazolinhydrochlorid

-
A
-
74-93-1
methylthiol

-
B
-
719-59-5
2-Amino-5-chlorobenzophenone

-
C
-
112608-09-0
3-Amino-6-chlor-4-hydroxy-2-oxo-4-phenyltetrahydrochinazolin

-
D
-
112608-08-9
N-(2-Benzoyl-4-chlorphenyl)monothiocarbamidsaeure-S-methylester

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 0.5h; Heating; reflux; | A n/a B 37% C n/a D n/a |
| With hydrogenchloride Product distribution; Heating; hydrolysis; |

-
-
26381-66-8
5-chloro-2-trichloroacetylaminobenzophenone

-
-
78-96-6, 1674-56-2
3-amino-2-propanol

-
A
-
719-59-5
2-Amino-5-chlorobenzophenone

-
B
-
80171-03-5
9-chloro-2-methyl-10b-phenyl-2,3,6,10b-tetrahydro-5H-oxazolo<3,2-c>quinazolin-5-one

-
C
-
80171-02-4
6-chloro-3,4-dihydro-3-(2-hydroxypropyl)-4-phenyl-4-trichloromethyl-2(1H)-quinazolinone

| Conditions | Yield |
|---|---|
| In propan-1-ol for 8h; Heating; | A 28% B 31% C 36% |

-
-
78823-09-3
2'-Benzoyl-4'-chloro-N-(N-phthaloyl-DL-alanyl)sarcosinanilide

-
A
-
719-59-5
2-Amino-5-chlorobenzophenone

-
B
-
78823-10-6
2'-Benzoyl-4'-chloro-N-(DL-alanyl)sarcosinanilide

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In ethanol; chloroform for 15h; Ambient temperature; | A 29% B 36% |
-
-
26381-66-8
5-chloro-2-trichloroacetylaminobenzophenone

-
-
75-31-0
isopropylamine

-
A
-
719-59-5
2-Amino-5-chlorobenzophenone

-
B
-
70547-52-3
1-(2-benzoyl-4-chlorophenyl)-3-isopropylurea

-
C
-
150878-29-8
6-chloro-3,4-dihydro-3-isopropyl-4-hydroxy-4-phenyl-2(1H)-quinazolinone

-
D
-
80170-55-4
6-chloro-3,4-dihydro-3-isopropyl-4-phenyl-4-trichloromethyl-2(1H)-quinazolinone

| Conditions | Yield |
|---|---|
| In N,N,N,N,N,N-hexamethylphosphoric triamide at 100℃; for 5h; | A 5% B 12% C 35% D 7% |

-
-
26381-66-8
5-chloro-2-trichloroacetylaminobenzophenone

-
-
108-91-8
cyclohexylamine

-
A
-
17387-32-5
5-chloro-2-dichloroacetamidobenzophenone

-
B
-
25514-22-1
1-(2-benzoyl-4-chlorophenyl)-3-cyclohexylurea

-
C
-
719-59-5
2-Amino-5-chlorobenzophenone

-
D
-
80170-81-6
6-chloro-3-cyclohexyl-3,4-dihydro-4-hydroxy-4-phenyl-2(1H)-quinazolinone

| Conditions | Yield |
|---|---|
| In N,N,N,N,N,N-hexamethylphosphoric triamide at 100℃; for 7h; Further byproducts given; | A 5% B 31% C 8% D 28% |

-
-
26381-66-8
5-chloro-2-trichloroacetylaminobenzophenone

-
-
108-91-8
cyclohexylamine

-
A
-
25514-22-1
1-(2-benzoyl-4-chlorophenyl)-3-cyclohexylurea

-
B
-
719-59-5
2-Amino-5-chlorobenzophenone

-
C
-
80170-81-6
6-chloro-3-cyclohexyl-3,4-dihydro-4-hydroxy-4-phenyl-2(1H)-quinazolinone

-
D
-
80170-56-5
6-chloro-3-cyclohexyl-3,4-dihydro-4-phenyl-4-trichloromethyl-2(1H)-quinazolinone

| Conditions | Yield |
|---|---|
| In N,N,N,N,N,N-hexamethylphosphoric triamide at 100℃; for 7h; Further byproducts given; | A 31% B 8% C 28% D 3% |


| Conditions | Yield |
|---|---|
| With aluminium trichloride for 5h; Heating; | 31% |
-
-
13788-59-5
N-(2-benzoyl-4-chlorophenyl)acetamide

-
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
21259-25-6
N-Benzoyl-2-amino-5-chlorobenzophenone

-
-
719-59-5
2-Amino-5-chlorobenzophenone

| Conditions | Yield |
|---|---|
| With sulfuric acid | |
| With sodium hydroxide In methanol for 4h; Heating; | 1.96 g |
-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
106-95-6
allyl bromide

-
-
4142-77-2
2-Allylamino-5-chlor-benzophenon

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetrabutylammomium bromide In tetrahydrofuran at 60℃; for 0.5h; | 100% |
-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
77-78-1
dimethyl sulfate

-
-
1022-13-5
5-chloro-2-(methylamino)benzophenone

| Conditions | Yield |
|---|---|
| With sodium hydroxide; tetrabutylammomium bromide In tetrahydrofuran at 60℃; for 1h; | 100% |
| With sodium hydroxide; tetrabutylammomium bromide; potassium carbonate In tetrahydrofuran at 30 - 35℃; for 2.5h; | 96% |

| Conditions | Yield |
|---|---|
| With 3-methyl-1-sulfoimidazolium trichloroacetate; trichloroacetic acid In neat (no solvent) at 100℃; Friedlaender Quinoline Synthesis; | 100% |
| With sulfuric acid; silica gel at 100℃; for 2h; Friedlaender synthesis; | 99% |
| KAl(SO4)2*12H2O/SiO2 at 30℃; for 1.5h; Friedlaender synthesis; | 99% |

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 153℃; for 19h; Sealed tube; | 100% |
| at 160℃; for 4h; Time; | 93% |
| With Amberlite Na sr1L at 115℃; for 0.0666667h; Microwave irradiation; Neat (no solvent); | 92% |
-
-
3132-99-8
m-bromobenzoic aldehyde

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
1257084-12-0
2-(3-bromophenyl)-6-chloro-4-phenylquinazoline

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With ammonium acetate In ethanol at 60℃; for 1h; | 94% |
| With 1,1,1,3',3',3'-hexafluoro-propanol; ammonium acetate at 55℃; for 0.5h; | 92% |
-
-
104-88-1
4-chlorobenzaldehyde

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
49797-11-7
6-chloro-2(4-chloro-phenyl)-4-phenyl-quinazoline

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With ammonium acetate In ethanol at 60℃; for 1h; Catalytic behavior; Solvent; Temperature; Time; | 96% |
| With ammonium acetate; iodine In neat (no solvent) at 40℃; for 2.5h; Green chemistry; | 95% |
-
-
100-52-7
benzaldehyde

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
30169-34-7
6-chloro-2,4-diphenyl-quinazoline

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With 1-butyl-3-methylimidazolium tetrachloridoferrate(III); ammonium acetate In neat (no solvent) at 40℃; for 2.5h; Green chemistry; | 95% |
| With ammonium acetate In ethanol at 60℃; for 1h; | 95% |
-
-
123-11-5
4-methoxy-benzaldehyde

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
84570-84-3
6-chloro-2-(4-methoxyphenyl)-4-phenylquinazoline

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With ammonium acetate In ethanol at 60℃; for 1h; | 95% |
| With Maltose; N,N'-Dimethylurea; ammonium acetate; ammonium chloride at 90℃; for 2.5h; | 91% |
-
-
90-02-8
salicylaldehyde

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
52537-82-3
2-(6-chloro-4-phenylquinazoline-2-yl)phenol

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With 1-butyl-3-methylimidazolium tetrachloridoferrate(III); ammonium acetate In neat (no solvent) at 40℃; for 2.5h; Green chemistry; | 91% |
| With ammonium acetate In neat (no solvent) at 60℃; Catalytic behavior; Green chemistry; | 91% |
| With Maltose; N,N'-Dimethylurea; ammonium acetate; ammonium chloride at 90℃; for 2.5h; | 88% |
| With ammonium acetate; 1-methyl-1H-imidazolium trifluoroacetate at 80℃; for 2h; Ionic liquid; aerobic oxidation; | 87% |
-
-
874-42-0
2,4-dichlorobenzaldeyhde

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
1257084-13-1
6-chloro-2-(2,4-dichlorophenyl)-4-phenylquinazoline

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With ammonium acetate; 1-methyl-1H-imidazolium trifluoroacetate at 80℃; for 2h; Ionic liquid; aerobic oxidation; | 92% |
| With ammonium acetate In ethanol at 60℃; for 1h; | 90% |
-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
552-89-6
2-nitro-benzaldehyde

-
-
1257084-14-2
6-chloro-2(2-nitro-phenyl)-4-phenyl-quinazoline

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With ammonium acetate; iodine In neat (no solvent) at 40℃; for 2.5h; Green chemistry; | 93% |
| With 1-butyl-3-methylimidazolium tetrachloridoferrate(III); ammonium acetate In neat (no solvent) at 40℃; for 2.5h; Green chemistry; | 91% |
-
-
455-19-6
4-Trifluoromethylbenzaldehyde

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
1372890-80-6
6-chloro-4-phenyl-2-(4-(trifluoromethyl)phenyl)quinazoline

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With Maltose; N,N'-Dimethylurea; ammonium acetate; ammonium chloride at 90℃; for 2.33333h; | 93% |
-
-
454-89-7
3-Trifluoromethylbenzaldehyde

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
49797-17-3
6-chloro-4-phenyl-2-(3-(trifluoromethyl)phenyl)quinazoline

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With Maltose; N,N'-Dimethylurea; ammonium acetate; ammonium chloride at 90℃; for 3.33333h; | 91% |
-
-
104-87-0
4-methyl-benzaldehyde

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
185547-80-2
6-chloro-4-phenyl-2-(p-tolyl)-quinazoline

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With ammonium acetate; iodine In neat (no solvent) at 40℃; for 0.5h; Green chemistry; | 97% |
| With ammonium acetate In toluene at 110℃; for 4h; Reflux; Green chemistry; | 95% |

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With 1,1,1,3',3',3'-hexafluoro-propanol; ammonium acetate at 55℃; for 1.5h; | 85% |

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With 1,1,1,3',3',3'-hexafluoro-propanol; ammonium acetate at 55℃; for 1h; | 90% |

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With 1,1,1,3',3',3'-hexafluoro-propanol; ammonium acetate at 55℃; for 1.5h; | 86% |

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With 1,1,1,3',3',3'-hexafluoro-propanol; ammonium acetate at 55℃; for 1.5h; | 84% |
-
-
99-61-6
3-nitro-benzaldehyde

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
49797-14-0
6-Chloro-2-(3-nitro-phenyl)-4-phenyl-quinazoline

| Conditions | Yield |
|---|---|
| With copper(II) choride dihydrate; ammonium acetate In ethanol for 4h; Reflux; | 100% |
| With 1,1,1,3',3',3'-hexafluoro-propanol; ammonium acetate at 55℃; for 1.16667h; | 90% |
-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
1159372-20-9
(2-amino-5-chlorophenyl)(phenyl)methanol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In isopropyl alcohol at 60℃; for 3.5h; | 99% |
| With sodium tetrahydroborate In methanol at 50℃; for 1h; | 98% |
| With sodium tetrahydroborate In ethanol | 98% |

| Conditions | Yield |
|---|---|
| sulfuric acid for 0.116667h; Friedlaender quinoline synthesis; microwave irradiation; | 99% |
| With 3-methyl-1-sulfoimidazolium trichloroacetate; trichloroacetic acid In neat (no solvent) at 100℃; Friedlaender Quinoline Synthesis; | 99% |
| With phosphoric acid triphenyl ester at 130℃; for 4h; | 97% |
-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
123-54-6
acetylacetone

-
-
22609-02-5
1-(6-chloro-2-methyl-4-phenylquinolin-3-yl)ethanone

| Conditions | Yield |
|---|---|
| With sulfuric acid; silica gel at 100℃; for 0.75h; Friedlaender synthesis; | 99% |
| With 3-methyl-1-sulfoimidazolium trichloroacetate; trichloroacetic acid In neat (no solvent) at 100℃; Friedlaender Quinoline Synthesis; | 99% |
| With tin(ll) chloride at 20℃; for 0.5h; | 98% |
-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
407-25-0
trifluoroacetic anhydride

-
-
69559-40-6
N-(2-benzoyl-4-chlorophenyl)-2,2,2-trifluoroacetamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 12h; | 99% |
| With triethylamine In dichloromethane at -78 - 20℃; for 0.166667h; |
-
-
108-94-1
cyclohexanone

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
116690-69-8
6-chloro-10-phenyl-1,2,3,4-tetrahydroacridine

| Conditions | Yield |
|---|---|
| With CuBTC metal-organic-framework at 89.84℃; for 5h; Friedlaender reaction; Neat (no solvent); | 99% |
| With 1-butylimidazolium tetrafluoroborate at 100℃; for 3h; Friedlaender reaction; | 98% |
| With tin(ll) chloride at 20℃; for 2.33333h; | 96% |
-
-
141-97-9
ethyl acetoacetate

-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
22609-01-4
ethyl 6-chloro-2-methyl-4-phenylquinoline-3-carboxylate

| Conditions | Yield |
|---|---|
| With magnesium(II) perchlorate In ethanol at 60℃; for 18h; Friedlaender synthesis; | 99% |
| scandium tris(dodecyl sulfate) In water at 40℃; Friedlaender annulation; | 99% |
| With cuprous triflate at 20℃; for 2h; Friedlaender synthesis; neat (no solvent); | 99% |
-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
762-42-5
dimethyl acetylenedicarboxylate

-
-
10037-28-2
dimethyl 4-phenyl-6-chloroquinoline-2,3-dicarboxylate

| Conditions | Yield |
|---|---|
| With propylphosphonium tetrachloroindate ionic liquid supported on nanosilica In neat (no solvent) at 110℃; for 0.5h; Reagent/catalyst; Temperature; | 99% |
| With indium(III) chloride at 80℃; for 0.833333h; regioselective reaction; | 96% |
| Stage #1: dimethyl acetylenedicarboxylate With pyridine at 0 - 10℃; for 0.25h; Stage #2: 5-chloro-2-aminobenzophenone at 20℃; for 12h; | 89% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; iodine In ISOPROPYLAMIDE; water at 80℃; for 4h; Sealed tube; | 99% |
-
-
719-59-5
2-Amino-5-chlorobenzophenone

-
-
501-65-5
diphenyl acetylene

-
-
1208258-68-7
6-chloro-2,3,4-triphenylquinoline

| Conditions | Yield |
|---|---|
| With 18-crown-6 ether; trifluorormethanesulfonic acid; palladium diacetate; 1,2-bis-(diphenylphosphino)ethane In toluene; acetonitrile at 100℃; for 12h; Reagent/catalyst; Solvent; Sealed tube; | 98.1% |
| With propylphosphonium tetrachloroindate ionic liquid supported on nanosilica In neat (no solvent) at 110℃; for 1h; | 67% |
| With acetic acid; palladium(II) bromide In benzonitrile; 1,2-dichloro-ethane at 130℃; for 18h; Sealed tube; | 51% |

| Conditions | Yield |
|---|---|
| In pyridine; hydrogenchloride | 98% |
| 39% |
2-Amino-5-chlorobenzophenone Chemical Properties
IUPAC Name: (2-amino-5-chlorophenyl)-phenylmethanone
Empirical Formula: C13H10ClNO
Molecular Weight: 231.6776g/mol
EINECS: 211-949-7
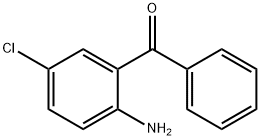
Structure of 2-Amino-5-chlorobenzophenone (CAS NO.719-59-5):
Index of Refraction: 1.636
Molar Refractivity: 65.17 cm3
Molar Volume: 181.7 cm3
Polarizability: 25.83×10-24cm3
Surface Tension: 52.2 dyne/cm
Density: 1.274 g/cm3
Flash Point: 210.4 °C
Enthalpy of Vaporization: 67.87 kJ/mol
Melting Point: 96-98 °C(lit.)
Boiling Point: 424.3 °C at 760 mmHg
Vapour Pressure: 2.09E-07 mmHg at 25°C
Stability: Stable. Incompatible with strong oxidizing agents.
Product Categories: Benzene derivatives;FINE Chemical & INTERMEDIATES;Aromatic Benzophenones & Derivatives (substituted);ketone;Diazepam
Canonical SMILES: C1=CC=C(C=C1)C(=O)C2=C(C=CC(=C2)Cl)N
InChI: InChI=1S/C13H10ClNO/c14-10-6-7-12(15)11(8-10)13(16)9-4-2-1-3-5-9/h1-8H,15H2
InChIKey: ZUWXHHBROGLWNH-UHFFFAOYSA-N
2-Amino-5-chlorobenzophenone Toxicity Data With Reference
| 1. | ipr-mus LD50:681 mg/kg | IJSIDW Indian Journal of Pharmaceutical Sciences. (Indian Journal of Pharmaceutical Sciences, Kalina, Santa Cruz 44 (1982),1. |
2-Amino-5-chlorobenzophenone Consensus Reports
Reported in EPA TSCA Inventory.
2-Amino-5-chlorobenzophenone Safety Profile
Moderately toxic by intraperitoneal route. When heated to decomposition it emits toxic vapors of NOx and Cl−.
Hazard Codes:  Xi,Xn
Xi,Xn
The Risk Statements information of 2-AMINO-5-CHLOROBENZOPHENONE :
22: Harmful if swallowed
20/21/22: Harmful by inhalation, in contact with skin and if swallowed
36/37/38: Irritating to eyes, respiratory system and skin
The Safety Statements information of 2-AMINO-5-CHLOROBENZOPHENONE :
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36: Wear suitable protective clothing
WGK Germany: 3
RTECS: PC4933500
HS Code: 29223900
2-Amino-5-chlorobenzophenone Specification
2-Amino-5-chlorobenzophenone , its cas register number is 719-59-5. It also can be called (2-Amino-5-chlorophenyl)phenylmethanone ; 2-Benzoyl-4-chloroaniline ; 3-14-00-00214 (Beilstein Handbook Reference) ; 5-Chloro-2-aminobenzophenone . 2-Amino-5-chlorobenzophenone (CAS NO.719-59-5) is a yellow powder.
Related Products
- 2-Amino-5-chlorobenzophenone
- 719-60-8
- 71962-74-8
- 71963-18-3
- 71963-21-8
- 71963-77-4
- 719-64-2
- 71-96-5
- 71965-10-1
- 71965-15-6
- 71965-21-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View