-
Name
3-ISOCHROMANONE
- EINECS 224-493-9
- CAS No. 4385-35-7
- Article Data68
- CAS DataBase
- Density 1.196 g/cm3
- Solubility 易溶于丙酮,甲醇,二氯乙烷等有机溶剂
- Melting Point 80-82 °C(lit.)
- Formula C9H8O2
- Boiling Point 332.6 °C at 760 mmHg
- Molecular Weight 148.161
- Flash Point 137.7 °C
- Transport Information
- Appearance WHITE CRYSTALLINE POWDER
- Safety 26-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 3-Isochromanone(8CI);Acetic acid, (a-hydroxy-o-tolyl)-, d-lactone (7CI);1H-2-Benzopyran-3(4H)-one;2-Hydroxymethylphenylaceticacid lactone;3,6-Dihydro-4,5-benzo-2-pyrone;Benzeneacetic acid,2-(hydroxymethyl)-, d-lactone;NSC 160530;
- PSA 26.30000
- LogP 1.28590
Synthetic route

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; C52H46O2P2Pd2; triphenylphosphine In toluene at 60℃; under 760.051 Torr; for 2.16667h; Conversion of starting material; | 100% |
| With potassium carbonate; bis-triphenylphosphine-palladium(II) chloride In tetrahydrofuran at 25℃; under 760 Torr; for 24h; | 71% |

| Conditions | Yield |
|---|---|
| In tert-butyl alcohol | 95.1% |
| Multi-step reaction with 3 steps 1: methanol 2: zinc chloride; tetrachloromethane / Erhitzen des Reaktionsprodukts mit Kaliumcyanid in wss. Aethanol 3: aqueous sulfuric acid View Scheme |
-
-
98590-71-7
α-chloro-α'-cyano-o-xylene

-
A
-
4385-35-7
isochroman-3-one

-
B
-
67519-22-6
α-hydroxy-α'-cyano-o-xylene

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 150℃; under 3677.86 Torr; for 3h; | A 95% B n/a |

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 0℃; for 240h; | 90% |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane for 24h; Ambient temperature; | 90% |
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane | 90% |
-
-
201230-82-2
carbon monoxide

-
-
612-14-6
phthalyl alcohol

-
A
-
4385-35-7
isochroman-3-one

-
B
-
95-47-6
o-xylene

-
C
-
644-36-0
o-methylphenylacetic acid

| Conditions | Yield |
|---|---|
| With hydrogen iodide; tetrakis(triphenylphosphine) palladium(0) In acetone at 90℃; under 68400 Torr; for 42h; Carbonylation; reduction; | A 88% B n/a C 9% |
| With hydrogen iodide; tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane at 120℃; under 68400 Torr; for 42h; Carbonylation; reduction; | A 13% B n/a C 52% |


| Conditions | Yield |
|---|---|
| With water; N-ethyl-N,N-diisopropylamine In tert-Amyl alcohol at 40 - 70℃; under 3087.28 - 3475.15 Torr; Product distribution / selectivity; | 84.53% |
| With water; N-ethyl-N,N-diisopropylamine In tert-Amyl alcohol at 70℃; under 1551.49 Torr; Product distribution / selectivity; | 80.95% |
| With water; N-ethyl-N,N-diisopropylamine; triphenylphosphine; palladium dichloride In tert-Amyl alcohol at 70 - 75℃; under 3863.02 Torr; for 4h; Product distribution / selectivity; | 75.4% |
| With water; N-ethyl-N,N-diisopropylamine; hydrogen tetrachloropalladate; triphenylphosphine In tert-Amyl alcohol at 70℃; under 3102.97 - 3863.02 Torr; for 2 - 4h; Product distribution / selectivity; | 69.9% |
-
-
95335-46-9
2-chloromethylphenylacetic acid

-
-
4385-35-7
isochroman-3-one

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water at 50℃; for 0.5h; Temperature; | 84.1% |
| With hydrogenchloride; potassium hydrogencarbonate In fluorobenzene; cyclohexane | |
| With potassium hydrogencarbonate In water at 50℃; pH=7.2; pH-value; Temperature; Reagent/catalyst; | 39 g |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydroxide; N-ethyl-N,N-diisopropylamine; triphenylphosphine; dihydrotetrachloropalladium (H2PdCl4) In o-xylene; water | 83.9% |

| Conditions | Yield |
|---|---|
| With 4,4'-di-tert-butylbiphenyl; lithium In tetrahydrofuran 1.) 20 deg C, 0.5 h; 2.) -78 deg C, 1 h; | 82% |
| With naphthalene; lithium 1.) THF, 0 deg C, 2 h, 2.) THF, -40 deg C, 5 min; Yield given. Multistep reaction; | |
| With naphthalene; lithium 1) THF, 0 deg C, 1.5 h, 2) THF, -40 deg C, 5 min; Yield given. Multistep reaction; |
-
-
98590-71-7
α-chloro-α'-cyano-o-xylene

-
A
-
4385-35-7
isochroman-3-one

-
D
-
67519-22-6
α-hydroxy-α'-cyano-o-xylene

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 100℃; for 6h; | A 76% B n/a C n/a D n/a |
| In water at 150℃; under 3677.86 Torr; for 3h; | A 75% B n/a C n/a D n/a |

-
-
75-85-4
tert-Amyl alcohol

-
-
612-12-4
o-Xylylene dichloride

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
4385-35-7
isochroman-3-one

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydroxide; triphenylphosphine; dihydrotetrachloropalladium (H2PdCl4) In o-xylene; water | 75.8% |

-
-
75-85-4
tert-Amyl alcohol

-
-
612-12-4
o-Xylylene dichloride

-
-
603-35-0
triphenylphosphine

-
-
4385-35-7
isochroman-3-one

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydroxide; carbon monoxide; N-ethyl-N,N-diisopropylamine; palladium(II) chloride In water | 75.4% |

-
-
22479-46-5
2-(ethoxycarbonylmethyl)benzoic acid

-
-
4385-35-7
isochroman-3-one

| Conditions | Yield |
|---|---|
| With dimethylsulfide borane complex In tetrahydrofuran at 40℃; Inert atmosphere; | 75% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; calcium hydroxide; carbon monoxide; triphenylphosphine In water; dichlorobis(triphenylphosphine)palladium[II]; tert-butyl alcohol | 72.9% |
| palladium(II) chloride In 1-methyl-pyrrolidin-2-one; water; tert-butyl alcohol | 66.4% |
| With hydrogenchloride; sodium hydroxide; carbon monoxide; N-ethyl-N,N-diisopropylamine; triphenylphosphine; palladium chloride (PdCl2) In o-xylene; water; tert-butyl alcohol |

-
-
4385-35-7
isochroman-3-one

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid In chloroform at 80℃; for 2h; | 70% |
-
-
941-69-5
N-phenyl-maleimide

-
-
163222-89-7, 163222-90-0
1,2-dihydrocyclobutabenzene-1-carboxylic acid

-
A
-
4385-35-7
isochroman-3-one

-
B
-
138235-90-2, 138331-82-5
(3aR,4S,9aR)-1,3-Dioxo-2-phenyl-2,3,3a,4,9,9a-hexahydro-1H-benzo[f]isoindole-4-carboxylic acid

-
-
138235-90-2, 138331-82-5
(3aS,4S,9aS)-1,3-Dioxo-2-phenyl-2,3,3a,4,9,9a-hexahydro-1H-benzo[f]isoindole-4-carboxylic acid

| Conditions | Yield |
|---|---|
| In benzene-d6 at 140℃; for 3h; | A 19% B 13% C 68% |


| Conditions | Yield |
|---|---|
| Stage #1: o-methylphenylacetic acid With sulfuryl dichloride; 2,2'-azobis(isobutyronitrile) In fluorobenzene at 75 - 80℃; for 4h; Stage #2: With potassium hydroxide; potassium hydrogencarbonate; potassium iodide In fluorobenzene; water at 60℃; pH=6.3; Conversion of starting material; | 68% |
| With iodobenzene; 3-chloro-benzenecarboperoxoic acid; potassium bromide In 2,2,2-trifluoroethanol at 20℃; for 24h; | 67% |
| Stage #1: o-methylphenylacetic acid With sulfuryl dichloride; 2,2'-azobis(isobutyronitrile) In chlorobenzene at 75 - 80℃; for 4h; Stage #2: With potassium hydroxide; potassium hydrogencarbonate; potassium iodide In water; chlorobenzene at 60℃; pH=6.5; Conversion of starting material; | 61.5% |
-
-
14736-50-6
o-(methoxycarbonylmethyl)benzoic acid

-
-
4385-35-7
isochroman-3-one

| Conditions | Yield |
|---|---|
| Stage #1: o-(methoxycarbonylmethyl)benzoic acid With borane In tetrahydrofuran at -15 - 20℃; for 16h; Stage #2: With toluene-4-sulfonic acid In toluene at 80℃; for 3h; | 66% |

-
-
4385-35-7
isochroman-3-one

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid In chloroform | 65% |
-
-
6346-00-5
2-(2-hydroxyethyl)benzyl alcohol

-
A
-
4385-35-7
isochroman-3-one

-
B
-
4702-34-5
isochroman-1-one

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole; [2,2]bipyridinyl; tetrakis(acetonitrile)copper(I) trifluoromethanesulfonate; 9-azabicyclo<3.3.1>nonane-N-oxyl In acetonitrile at 22℃; for 2h; regioselective reaction; | A 18% B 58% |
| With iodosylbenzene; potassium bromide In water for 2h; sonication; | |
| With [(η5-C5Me5)Ir(6,6'-dihydroxy-2,2'-bipyridine)(H2O)]OTf2 In water for 20h; Inert atmosphere; Reflux; Green chemistry; Overall yield = 88 %; |
-
-
52267-51-3
benzyl diazoacetate

-
-
4385-35-7
isochroman-3-one

| Conditions | Yield |
|---|---|
| dirhodium tetraacetate In dichloromethane for 24h; | 56% |
-
-
201230-82-2
carbon monoxide

-
-
612-14-6
phthalyl alcohol

-
A
-
4385-35-7
isochroman-3-one

-
B
-
644-36-0
o-methylphenylacetic acid

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); hydrogen iodide In acetone at 90℃; under 68400 Torr; for 42h; | A 56% B 9 % Spectr. |


| Conditions | Yield |
|---|---|
| With n-butyllithium In diethyl ether; hexane 1) 24 h, room temp. or 4 h reflux; | 53% |

| Conditions | Yield |
|---|---|
| With N,N,N,N,-tetramethylethylenediamine; α,α′-bis(2-pyridyl(tert-butyl)phosphino)-o-xylene; palladium diacetate In N,N-dimethyl-formamide at 115℃; for 12h; Inert atmosphere; | 46% |

| Conditions | Yield |
|---|---|
| palladium(II) chloride In water; tert-butyl alcohol | 24% |
-
-
98590-71-7
α-chloro-α'-cyano-o-xylene

-
-
4385-35-7
isochroman-3-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 100℃; for 6h; | 3% |
-
-
408535-88-6
(2-methoxymethyl-phenyl)-acetonitrile

-
-
4385-35-7
isochroman-3-one

| Conditions | Yield |
|---|---|
| With hydrogen bromide | |
| With sulfuric acid |

| Conditions | Yield |
|---|---|
| With thionyl chloride In 5,5-dimethyl-1,3-cyclohexadiene; N,N-dimethyl-formamide at 20 - 30℃; for 4h; Solvent; | 100% |
| With hydrogenchloride at 20 - 26℃; for 2.5h; | 98% |
| With thionyl chloride at 20℃; Cooling with ice; | 96% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
4385-35-7
isochroman-3-one

-
-
87532-04-5
((1H-isochromen-3-yl)oxy)trimethylsilane

| Conditions | Yield |
|---|---|
| Stage #1: isochroman-3-one With lithium diisopropyl amide In tetrahydrofuran; hexane at -78℃; for 0.533333h; Inert atmosphere; Stage #2: chloro-trimethyl-silane In tetrahydrofuran; hexane at -78 - 20℃; Inert atmosphere; Enzymatic reaction; | 99% |
| With n-butyllithium; diisopropylamine 1.) THF, -78 deg C, 10 min, 2.) 3 h, warm. to RT; Yield given. Multistep reaction; |
-
-
4385-35-7
isochroman-3-one

-
-
74-89-5
methylamine

-
-
218131-53-4
2-(2-(hydroxymethyl)phenyl)-N-methylacetamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 8h; | 99% |
| In tetrahydrofuran at 20℃; for 8h; | 93% |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; for 3h; Inert atmosphere; | 99% |
| With lithium aluminium tetrahydride In tetrahydrofuran at 20℃; for 3h; Inert atmosphere; | 99% |
| With phenylsilane; potassium hydroxide In tetrahydrofuran for 4h; Schlenk technique; Inert atmosphere; Reflux; | 60% |
| With samarium diiodide; water In tetrahydrofuran at 20℃; for 1h; Inert atmosphere; | 65 %Spectr. |

| Conditions | Yield |
|---|---|
| With phosphorus tribromide | 98% |
| With hydrogen bromide | 90% |
| With hydrogen bromide at 20℃; for 48h; Ring cleavage; esterification; |
-
-
4385-35-7
isochroman-3-one

-
-
95360-33-1
methyl 2-(chloromethyl)phenylacetate

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol; dichloromethane | 98% |
| With thionyl chloride; potassium hydrogencarbonate In methanol; toluene | |
| Multi-step reaction with 2 steps 1: thionyl chloride / 4 h / Reflux; Heating 2: 1 h / 20 °C / Cooling with ice View Scheme |

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; W(OTf)6; hydrogen; acetic acid at 50℃; under 760.051 Torr; for 12h; | 98% |
| With palladium on activated carbon; W(OTf)6; hydrogen In acetic acid at 50℃; under 760.051 Torr; for 12h; | 97% |

| Conditions | Yield |
|---|---|
| With piperidine at 140℃; for 1h; Knoevenagel condensation; | 97% |

| Conditions | Yield |
|---|---|
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; sodium acetate In toluene at 110℃; for 36h; Inert atmosphere; Glovebox; Molecular sieve; Schlenk technique; | 97% |
-
-
4385-35-7
isochroman-3-one

| Conditions | Yield |
|---|---|
| Stage #1: isochroman-3-one With sodium azide; p-toluenesulfonyl chloride In acetonitrile at 20℃; Stage #2: With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 0 - 20℃; | 96% |
| With 4-toluenesulfonyl azide; 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 0℃; | 81% |
| With 4-acetamidobenzenesulfonyl azide; 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 0 - 20℃; for 1h; | 60% |
-
-
4385-35-7
isochroman-3-one

-
-
107-31-3
Methyl formate

-
-
117428-51-0
methyl (E)-3-methoxy-2-(2-chloromethylphenyl)acrylate

| Conditions | Yield |
|---|---|
| With acetic acid Heating; | 96% |

| Conditions | Yield |
|---|---|
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; sodium acetate In toluene at 110℃; for 36h; Inert atmosphere; Glovebox; Molecular sieve; Schlenk technique; | 95% |
-
-
4385-35-7
isochroman-3-one

-
-
42900-89-0
3,4-dihydro-1H-2-benzopyran-3-ol

| Conditions | Yield |
|---|---|
| With diisobutylaluminium hydride In toluene at -60 - -10℃; | 94% |
| With diisobutylaluminium hydride In dichloromethane at -78℃; for 3h; | 91% |
| With lithium aluminium tetrahydride In tetrahydrofuran at -78℃; for 1h; Reduction; | |
| With diisobutylaluminium hydride In dichloromethane; toluene at -78℃; for 2h; | |
| With diisobutylaluminium hydride In dichloromethane at -78℃; for 2h; Inert atmosphere; |
-
-
4385-35-7
isochroman-3-one

-
-
13737-35-4
o-(bromomethyl)phenylacetic acid

| Conditions | Yield |
|---|---|
| With hydrogen bromide In acetic acid | 93% |
| With hydrogen bromide In acetic acid at 20 - 70℃; for 3h; | 93% |
| With hydrogen bromide; acetic acid at 70℃; for 1h; | 88% |
| With hydrogen bromide In acetic acid 1.) 20 deg C, 2 h; 2.) 70 deg C, 1 h; | |
| With hydrogen bromide In water |
-
-
4385-35-7
isochroman-3-one

-
-
14773-42-3
3-(3,4-dimethoxyphenyl)propylamine

-
-
166537-41-3
N-<3-(3,4-Dimethoxyphenyl)propyl><2-(hydroxymethyl)phenyl>acetamid

| Conditions | Yield |
|---|---|
| In ethanol for 3h; Heating; | 92% |
-
-
4385-35-7
isochroman-3-one

-
-
100-46-9
benzylamine

-
-
6772-73-2
1,4-dihydro-2-(phenylmethyl)-3(2H)-isoquinolinone

| Conditions | Yield |
|---|---|
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; sodium acetate In toluene at 110℃; for 36h; Inert atmosphere; Glovebox; Molecular sieve; Schlenk technique; | 92% |
| With aluminium trichloride at 160℃; for 20h; | 73% |
-
-
4385-35-7
isochroman-3-one

-
-
74-88-4
methyl iodide

-
-
122571-80-6
3,4-dihydro-4-methyl-(3H)-2-benzopyran-3-one

| Conditions | Yield |
|---|---|
| Stage #1: isochroman-3-one With N,N,N,N,N,N-hexamethylphosphoric triamide; n-butyllithium In tetrahydrofuran; hexane at -78℃; for 1h; Stage #2: methyl iodide at -78℃; for 1h; | 90% |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In methanol at 40℃; for 24h; Solvent; Temperature; Concentration; Reagent/catalyst; | 90% |
-
-
4385-35-7
isochroman-3-one

-
-
18655-52-2
3-(3-methoxyphenyl)propan-1-amine

| Conditions | Yield |
|---|---|
| In ethanol for 3h; Heating; | 88% |

| Conditions | Yield |
|---|---|
| With heptahydridobis(triphenylphosphine)rhenium In tetrahydrofuran at 150℃; for 5h; Inert atmosphere; teflon-cocked sealed tube; | 88% |

| Conditions | Yield |
|---|---|
| With allyl(cyclopentadiene)palladium(II); 1,3-bis(dicyclohexylphosphine)propane In 1,2-dimethoxyethane at 80℃; for 18h; | 87% |
-
-
4385-35-7
isochroman-3-one

-
-
623-49-4
ethyl cyanoformate

-
-
72206-74-7
ethyl 3-oxoisochroman-4-carboxylate

| Conditions | Yield |
|---|---|
| Stage #1: isochroman-3-one With lithium diisopropyl amide Inert atmosphere; Stage #2: ethyl cyanoformate Claisen condensation; | 87% |
-
-
4385-35-7
isochroman-3-one

-
-
36455-21-7
3-benzyloxy-4-methoxyphenylethylamine

-
-
98988-21-7
N-<2-(3-benzyloxy-4-methoxyphenyl)-ethyl>-(2-hydroxymethyl)phenylacetamide

| Conditions | Yield |
|---|---|
| In ethanol for 24h; Ambient temperature; | 86% |

| Conditions | Yield |
|---|---|
| With bis[dichloro(pentamethylcyclopentadienyl)iridium(III)]; sodium acetate In toluene at 110℃; for 36h; Inert atmosphere; Glovebox; Molecular sieve; Schlenk technique; | 85% |
3-Isochromanone Specification
The 3H-2-Benzopyran-3-one,1,4-dihydro- with CAS registry number of 4385-35-7 is also known as 3-Isochromanone. The IUPAC name is 1,4-Dihydroisochromen-3-one. It belongs to product categories of Benzopyrans; Building Blocks; Heterocyclic Building Blocks. Its EINECS registry number is 224-493-9. In addition, the formula is C9H8O2 and the molecular weight is 148.16. This chemical is a beige powder that may cause inflammation to the skin or other mucous membranes. It should be sealed in cool, dry place away from oxidants.
Physical properties about 3H-2-Benzopyran-3-one,1,4-dihydro- are: (1)ACD/LogP: 0.75; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.75; (4)ACD/LogD (pH 7.4): 0.75; (5)ACD/BCF (pH 5.5): 2.19; (6)ACD/BCF (pH 7.4): 2.19; (7)ACD/KOC (pH 5.5): 60.91; (8)ACD/KOC (pH 7.4): 60.91; (9)#H bond acceptors: 2; (10)Index of Refraction: 1.562; (11)Molar Refractivity: 40.16 cm3; (12)Molar Volume: 123.8 cm3; (13)Surface Tension: 43.5 dyne/cm; (14)Density: 1.196 g/cm3; (15)Flash Point: 137.7 °C; (16)Enthalpy of Vaporization: 57.54 kJ/mol; (17)Boiling Point: 332.6 °C at 760 mmHg; (18)Vapour Pressure: 0.000144 mmHg at 25 °C.
Preparation of 3H-2-Benzopyran-3-one,1,4-dihydro-: it is prepared by reaction of 2-methyl-benzyl alcohol. The reaction needs reagent butyllithium and solvents diethyl ether, hexane at room temperature for 24 hours. The yield is about 53%.

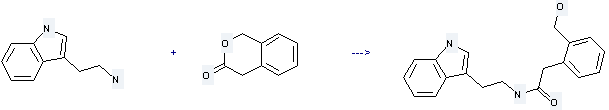
Uses of 3H-2-Benzopyran-3-one,1,4-dihydro-: it is used to produce 2-(2-hydroxymethyl-phenyl)-N-(2-indol-3-yl-ethyl)-acetamide by reaction with 2-indol-3-yl-ethylamine. The reaction occurs with solvent ethanol and other condition of heating for 20 hours. The yield is about 83%.
When you are using this chemical, please be cautious about it. As a chemical, it is irritating to eyes, respiratory system and skin. During using it, wear suitable gloves and eye/face protection. If contact with eyes accidently, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
1. Canonical SMILES: C1C2=CC=CC=C2COC1=O
2. InChI: InChI=1S/C9H8O2/c10-9-5-7-3-1-2-4-8(7)6-11-9/h1-4H,5-6H2
3. InChIKey: ILHLUZUMRJQEAH-UHFFFAOYSA-N
Related Products
- 3-Isochromanone
- 438553-83-4
- 438554-27-9
- 4385-56-2
- 4385-62-0
- 4385-63-1
- 4385-75-5
- 4385-76-6
- 438577-61-8
- 438577-62-9
- 4385-77-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View