-
Name
Triethylphosphine
- EINECS 209-068-8
- CAS No. 554-70-1
- Article Data84
- CAS DataBase
- Density 0.88g/mLat 20°C
- Solubility Insoluble in water.
- Melting Point -3oC
- Formula C6H15P
- Boiling Point 126.516 °C at 760 mmHg
- Molecular Weight 118.159
- Flash Point 27.202 °C
- Transport Information UN 2924 3/PG 2
- Appearance clear liquid
- Safety 26-36/37/39-45-7/9-33-16
- Risk Codes 11-34-67-17
-
Molecular Structure
-
Hazard Symbols
 F,
F, C
C
- Synonyms HishicolinP 2;Triethylphosphorus;
- PSA 13.59000
- LogP 2.52800
Synthetic route

| Conditions | Yield |
|---|---|
| In dichloromethane mixing of the Mo compd. with the free phosphine (molar ratio 4.5:1) in CH2Cl2;; | A 100% B n/a |
| In toluene mixing of the Mo compd. with the free phosphine (molar ratio 2.1:1) in toluene;; | A 100% B n/a |


| Conditions | Yield |
|---|---|
| With phosphan; zinc(II) oxide In acetone at 0℃; for 12h; Solvent; Reagent/catalyst; Temperature; Irradiation; | 96% |

| Conditions | Yield |
|---|---|
| With phenylsilane; C28H16F24O8S2Si In (2)H8-toluene at 100℃; for 48h; Inert atmosphere; | 91% |
| With aluminium hydride In 1,2-dimethoxyethane at 0 - 65℃; for 12h; Inert atmosphere; Schlenk technique; | 70% |
| With chromium |
-
-
925-90-6
ethylmagnesium bromide

-
-
119841-34-8, 127780-84-1
cis-2,10-dimethyl-<1,2,3>benzothiadiphospholo<2,3-b><1,2,3>benzothiadiphosphole

-
A
-
554-70-1
triethylphosphine

-
B
-
736137-64-7
4-methyl-2-[(5-methyl-2-sulfanylphenyl)phosphanyl]benzenethiol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran | A n/a B 90% |

-
-
16940-66-2
sodium tetrahydroborate

-
-
121846-72-8
cis,trans-dicarbonylbis(triethylphosphine)(dithioformate-κ2-S)manganese

-
B
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| In ethanol; dichloromethane Addn. of a NaBH4 soln. (EtOH) to a stirred soln. of Mn-complex in CH2Cl2 under N2, stirring (1.5 h), colour turns from deep blue to bright red.; Evapn. of solvent in vac., pumping for 1 h to eleminate the free phosphine, extg. of red solid (hexane), filtn., addn. of EtOH, slow evapn. in vac., elem. anal.; | A 90% B n/a |


| Conditions | Yield |
|---|---|
| Stage #1: chloroethane With magnesium; methyl iodide In tetrahydrofuran; 5,5-dimethyl-1,3-cyclohexadiene; ethyl acetate at 40 - 50℃; for 2.5h; Inert atmosphere; Stage #2: With phosphorus trichloride In tetrahydrofuran; 5,5-dimethyl-1,3-cyclohexadiene; ethyl acetate at 5℃; for 4h; Concentration; Temperature; Inert atmosphere; | 89.9% |
-
-
14177-93-6, 15692-07-6, 13965-02-1
trans-platinum(triethylphosphine)2(chloro)2

-
B
-
116553-43-6
(Et3P)2Pt{η2-(PSiMe3)2}

-
C
-
15573-38-3
Tris(trimethylsilyl)phosphane

-
D
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| In toluene under protective gas; 8.8 mmol of the Li salt in toluene added to 4 mmol of the Pt complex in toluene at -20°C; mixt. warmed to 20°C and stirred for 2 d; filtration; filtrate evapd. in vac.; fractionated crystn.; | A 12% B 7% C n/a D n/a |

-
-
60-29-7
diethyl ether

-
-
4303-71-3
triphenylmethyl sodium

-
-
4317-06-0
tetraethylphosphonium iodide

-
A
-
519-73-3
triphenylmethane

-
B
-
554-70-1
triethylphosphine


| Conditions | Yield |
|---|---|
| With phosphonium iodide at 160℃; entsteht das Hydrojodid; |
-
-
64-17-5
ethanol

-
-
141-52-6
sodium ethanolate

-
A
-
292638-84-7
styrene

-
B
-
597-50-2
triethylphosphine oxide

-
C
-
104-51-8
1-butylbenzene

-
D
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| beim anschliessenden Erhitzen unter Stickstoff; |

-
-
64-17-5
ethanol

-
-
141-52-6
sodium ethanolate

-
A
-
530-48-3
1,1-Diphenylethylene

-
B
-
597-50-2
triethylphosphine oxide

-
C
-
719-79-9
1,1-diphenylbutane

-
D
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| beim anschliessenden Erhitzen in einer Stickstoff-Atmosphaere unter vermindertem Druck; |


| Conditions | Yield |
|---|---|
| With phosphonium iodide at 180℃; entsteht das Hydrojodid; |
-
-
16616-89-0
dichlorotriethylphosphorane

-
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| With sodium amalgam; ethanol |
-
-
861373-39-9
fluoren-9-ylidene-triethylphosphoranylidene-hydrazine

-
A
-
2071-44-5
di-fluoren-9-ylidene-hydrazine

-
B
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| at 200 - 210℃; im Vakuum; |

| Conditions | Yield |
|---|---|
| With diethyl ether |

| Conditions | Yield |
|---|---|
| With diethyl ether; phosphorus tribromide das Reaktionsprodukt wird im Kohlendioxydstrom destilliert; | |
| With phosphorus trichloride at -20℃; |

| Conditions | Yield |
|---|---|
| With tetraphosphorus trisulfide; diethyl ether |

| Conditions | Yield |
|---|---|
| With diethyl ether; phosphorus trichloride entsteht eine Verbindung mit Zinkchlorid; |

| Conditions | Yield |
|---|---|
| With phosphorus trichloride und Behandeln des Reaktionsprodukts mit wss.NaOH; |

| Conditions | Yield |
|---|---|
| With phosphorus trichloride |
-
-
61223-96-9
triethyl-phenylthiocarbamoyl-phosphonium betaine

-
A
-
103-72-0
phenyl isothiocyanate

-
B
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| beim Erhitzen; |
-
-
82540-63-4
triethyl-diphenylacetyl-phosphonium betaine

-
A
-
525-06-4
diphenyl ketene

-
B
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| at 100℃; |
-
-
27063-22-5
tetraethylphosphonium; tetraethylphosphonium acetate

-
A
-
34557-54-5
methane

-
B
-
597-50-2
triethylphosphine oxide

-
C
-
78-93-3
butanone

-
D
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| beim Erhitzen; Produkt 5: Aethylen, Produkt 6: CO2; |

-
-
23666-92-4
triethylbenzyl phosphonium chloride

-
A
-
100-44-7
benzyl chloride

-
B
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| at 340℃; |

-
A
-
74-84-0
ethane

-
B
-
597-50-2
triethylphosphine oxide

-
C
-
100-41-4
ethylbenzene

-
D
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| bei der Destillation; Produkt 5: Styrol; |


-
A
-
74-85-1
ethene

-
B
-
597-50-2
triethylphosphine oxide

-
C
-
96-22-0
pentan-3-one

-
D
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| at 200 - 230℃; Produkt 5: CO2, Produkt 6: CO; |


-
A
-
74-84-0
ethane

-
B
-
597-50-2
triethylphosphine oxide

-
C
-
100-41-4
ethylbenzene

-
D
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| bei der Destillation; Produkt 5: Styrol; |


| Conditions | Yield |
|---|---|
| at 350 - 380℃; |

| Conditions | Yield |
|---|---|
| With sodium phosphide |
-
-
74-85-1
ethene

-
-
74-88-4
methyl iodide

-
A
-
4317-06-0
tetraethylphosphonium iodide

-
B
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| With phosphorous; cyclohexane at 250℃; under 661957 Torr; unter Einleiten von Wasserstoff; |

-
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| With iodine In diethyl ether at 20℃; for 48h; Addition; | 100% |

| Conditions | Yield |
|---|---|
| In pentane at 0℃; Addition; | 100% |

| Conditions | Yield |
|---|---|
| With pyridine In toluene at 85℃; for 21h; | 100% |

| Conditions | Yield |
|---|---|
| In toluene at 20℃; | 100% |
-
-
105218-70-0
1,1,2,2-tetrachloro-1,2-bis(t-butylamido)1,2-bis(t-butylamine)ditungsten(III)(3W-W)

-
-
554-70-1
triethylphosphine

-
-
123214-94-8, 157477-92-4
1,1,2,2-tetrachloro-1,2-bis(t-butylamido)1,2-bis(triethylphsphine)-ditungsten(III)(3W-W)

| Conditions | Yield |
|---|---|
| In pentane under dry N2; addn. of phosphine to W-complex in n-pentane, stirring (10 min); solvent removal (vac.); | 100% |
-
-
12146-37-1, 124717-04-0
(bicyclo[2.2.1]hepta-2,5-diene)tetracarbonylmolybdenum(0)

-
-
554-70-1
triethylphosphine

-
-
19217-80-2, 19217-81-3, 22614-45-5
cis-{molybdenum(0)(carbonyl)4(P(ethyl)3)2}

-
B
-
121-46-0
bicyclo[2.2.1]hepta-2,5-diene

| Conditions | Yield |
|---|---|
| In tetrahydrofuran under argon; reaction in a calorimeter; | A 100% B n/a |


-
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| In benzene (under N2 or Ar) addn. of P-compd. to Pt-complex in benzene at room temp., standing overnight (ambient temp.); solvent removal (vac.); elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane The soln. of reagents in CH2Cl2 was stirred for 10 min at 25°C (N2 atm.);; hexane was added, concd., filtered, washed with Et2O-hexane, recrystd. from CH2Cl2-hexane;; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane byproducts: P(C6H5)3; mixing of the Mo compd. with the free phosphine (molar ratio 2.5:1) in CH2Cl2;; | 100% |

| Conditions | Yield |
|---|---|
| In dichloromethane-d2 PEt3 is added to Fe-Au-compd. in CD2Cl2 in a NMR-tube; monitored by (31)P-NMR-spectroscopy; | 100% |

| Conditions | Yield |
|---|---|
| In toluene mixing of the Mo compd. with the free phosphine (molar ratio 2.0:1) in toluene;; | A 100% B n/a |


| Conditions | Yield |
|---|---|
| In toluene PEt3 was added to a soln. of (Cu(OSiPh3))4*toluene in toluene (molar ratio 1.38:0.17) under Argon and the mixt. stirred for 16 h;; the solvent was evapd. in vac. and the resulting oil kept in a dynamic vac. at ca. 60°C for 4 h for slow solidification; elem. anal.;; | 100% |
-
-
204003-48-5
(PEt3)2Pt(2,2'-biphenyl)

-
-
1333-74-0
hydrogen

-
-
554-70-1
triethylphosphine

-
-
217080-23-4
trans-(PEt3)2Pt(α-biphenyl)H

| Conditions | Yield |
|---|---|
| In benzene-d6 H2-atmosphere (1 atm); stirring (80°C, 12 h); evapn.; | 100% |

-
-
488090-77-3
5,10,15-tris(pentafluorophenyl)corrole manganese(V) (N-mesityl)

-
-
554-70-1
triethylphosphine

-
-
488090-81-9
manganeser(III) 5,10,15-tris(pentafluorophenyl)corrole NPEt3(mesityl)

| Conditions | Yield |
|---|---|
| In toluene Irradiation (UV/VIS); (Ar), at 85°C for 21 h; | 100% |
-
-
488090-78-4
5,10,15-tris(pentafluorophenyl)corrole manganese(V) (N-2,4,6-trichlorophenyl)

-
-
554-70-1
triethylphosphine

-
-
488090-83-1
manganese(III) 5,10,15-tris(pentafluorophenyl)corrple NPEt3(2,4,6-trichlorophenyl)

| Conditions | Yield |
|---|---|
| In toluene Irradiation (UV/VIS); (Ar), at 20°C for < 1 min; | 100% |

| Conditions | Yield |
|---|---|
| In hexane (N2); standard Schlenk technique; hexane and PEt3 were added via syringeto mixt. of Ru complex and ligand; mixt. was stirred at room temp. for 20 h; volatiles evapd. with oil diffusion pump; crystd. from hexane at -30°C; recrystd. (pentane); | 100% |

-
-
164355-65-1, 164355-51-5
(η(3)-pentadienyl)Ir(PEt3)2

-
-
554-70-1
triethylphosphine

-
-
134967-62-7
((1,4,5-η)pentadienyl)Ir(PEt3)3

| Conditions | Yield |
|---|---|
| In benzene-d6 N2-atmosphere, NMR tube; detd. by NMR spectroscopy; | 100% |

-
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| In not given | 100% |
-
-
554-70-1
triethylphosphine

-
-
15647-86-6
triethylphosphane hydrobromide

| Conditions | Yield |
|---|---|
| With hydrogen bromide In water at 0 - 20℃; Inert atmosphere; | 100% |
| With hydrogen bromide In water at 0 - 20℃; Inert atmosphere; | 97% |
| With hydrogen bromide |

| Conditions | Yield |
|---|---|
| In hexane for 0.166667h; Inert atmosphere; Glovebox; | 100% |

| Conditions | Yield |
|---|---|
| In benzene-d6 at 20℃; for 0.0833333h; Inert atmosphere; Schlenk technique; | 100% |
-
-
1295-35-8
bis(1,5-cyclooctadiene)nickel (0)

-
-
1437769-76-0
3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)naphthalen-2-yl trifluoromethanesulfonate

-
-
7550-35-8
lithium bromide

-
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; hexane at 20℃; for 1h; Inert atmosphere; | 100% |


-
-
554-70-1
triethylphosphine

-
-
15529-90-5
(triethylphosphine)chlorogold(I)

| Conditions | Yield |
|---|---|
| Stage #1: gold(III) tetrachloride trihydrate With 2,2'-thiobis-ethanol In water Stage #2: triethylphosphine In ethanol; water at 0 - 20℃; for 2h; | 100% |
-
-
554-70-1
triethylphosphine

-
-
677-11-2, 54082-58-5
triethyldifluorophosphorane

| Conditions | Yield |
|---|---|
| With xenon fluoride In dichloromethane at 20℃; for 1h; Inert atmosphere; | 99% |
| With 1-pentanamine hydrofluoride; N,N,N,N-tetraethylammonium tetrafluoroborate In acetonitrile anodic oxidation; | 80% |
| (i) sulfur, benzene, (ii) SbF3; Multistep reaction; |
-
-
104-15-4
toluene-4-sulfonic acid

-
-
67-64-1
acetone

-
-
554-70-1
triethylphosphine

-
-
126580-26-5
Toluene-4-sulfonatetriethyl-(1-hydroxy-1-methyl-ethyl)-phosphonium;

| Conditions | Yield |
|---|---|
| 1) -78 deg C, 4h, 2) RT; | 99% |


| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 0.833333h; Inert atmosphere; | 99% |
| In dichloromethane at 25℃; for 24h; Inert atmosphere; | 98.4% |
| In diethyl ether Ambient temperature; |
-
-
12560-37-1
[Co2Rh2(CO)12]

-
-
21050-13-5
bis(triphenylphosphine)iminium chloride

-
-
554-70-1
triethylphosphine

-
A
-
53433-12-8
[bis(triphenylphospine)nitrogen(1+)][Co(CO)4]

-
-
14871-47-7, 15631-52-4
trans-ClRh(CO)(PEt3)2

| Conditions | Yield |
|---|---|
| In dichloromethane N2-atmosphere; addn. of (PPN)Cl to Co/Rh complex, then addn. of 2 equivs. PEt3; solvent removal (vac.), hexane addn., filtration, washing (hexane); | A 98.3% B 99% |


-
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| In further solvent(s) Schlenk flask; in aniline, flask was sealed and heated at 50°C with stirring for 2 d; mixt. was filtered, layered with MeCN, ppt. was filtered off, washed with Et2O; NMR; | 99% |

-
-
554-70-1
triethylphosphine

| Conditions | Yield |
|---|---|
| In acetonitrile byproducts: P(C6H5)3; under N2; monitored by (1)H-NMR; | 99% |
Triethylphosphine Chemical Properties
IUPAC Name: Triethylphosphane (554-70-1)
Synonyms: Aurora ka-1111 ; Cytop(R) 320 ; Triethylphosphine ; (C2H5)3P ; Phosphorus triethyl ; Ttl409 ; Triethylphosphane ; Triethyl-phosphane
CAS: 554-70-1
MF: C6H15P
MW: 118.16
MS: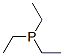
EINECS: 209-068-8
Product Categories: Tertiary Phosphines ; Organics ; Phosphine Ligands ; Catalysis and Inorganic Chemistry ; Phosphorus Compounds
Mol File: 554-70-1.mol
Flash Point: 27.2 °C
Enthalpy of Vaporization: 34.93 kJ/mol
Boiling Point: 126.5 °C at 760 mmHg
Vapour Pressure: 14.1 mmHg at 25°C
Density: 0.88 g/mL at 20 °C
Refractive index : n20/D 1.456(lit.)
Sensitive: Air & Moisture Sensitive
Merck: 14,9675
Triethylphosphine Uses
Triethyl phosphine (554-70-1) is used in organic syntheses.
Triethylphosphine Safety Profile
Hazard Codes: F![]() ,C
,C ![]()
Risk Statements: 11-34-67-17
11: Highly Flammable
17: Spontaneously flammable in air
34: Triethyl phosphine (554-70-1) can causes burns.
67: Vapors may cause drowsiness and dizziness
Safety Statements: 26-36/37/39-45-7/9-33-16
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37/39: Wear suitable protective clothing, gloves and eye/face protection
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
7/9: Keep container tightly closed and in a well-ventilated place
33: Take precautionary measures against static discharges
16: Keep Triethyl phosphine (554-70-1) away from sources of ignition - No smoking
RIDADR : UN 2924 3/PG 2
WGK Germany: 3
RTECS : SZ3400000
F : 10-13-23
HazardClass: 4.2
PackingGroup: I
Related Products
- Triethylphosphine
- Triethylphosphine oxide
- 55473-29-5
- 554-73-4
- 55476-36-3
- 55477-35-5
- 55478-23-4
- 55478-55-2
- 55480-45-0
- 55-48-1
- 55481-86-2
- 55481-88-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View