-
Name
TRI-O-TOLYLPHOSPHINE
- EINECS 228-193-9
- CAS No. 6163-58-2
- Article Data31
- CAS DataBase
- Density 1.16[at 20℃]
- Solubility Soluble in alcohol. Slightly soluble in cold water. Insoluble in water.
- Melting Point 123-125 °C(lit.)
- Formula C21H21P
- Boiling Point 412.4 °C at 760 mmHg
- Molecular Weight 304.371
- Flash Point 214.6 °C
- Transport Information
- Appearance white to light yellow crystal powder
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Phosphine,tri-o-tolyl- (7CI,8CI);NSC 116667;Tri-2-tolylphosphine;Tri-o-tolylphosphine;Tris(2-toluene)phosphine;Tris(2-tolyl)phosphine;Tris(o-methylphenyl)phosphine;Tris(o-tolyl)phosphine;Tri-(O-Tol)phosphine;
- PSA 13.59000
- LogP 4.37000
Synthetic route
-
-
64-17-5
ethanol

-
-
878809-28-0
tris(2-tolyl)phosphane-borane(1:1)

-
A
-
150-46-9
triethyl borate

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| Inert atmosphere; Reflux; | A n/a B 96% |


| Conditions | Yield |
|---|---|
| Stage #1: 2-methylphenyl bromide With magnesium In tetrahydrofuran Reflux; Inert atmosphere; Stage #2: With phosphorus trichloride In tetrahydrofuran for 24h; Inert atmosphere; | 89% |
| Stage #1: 2-methylphenyl bromide With magnesium In tetrahydrofuran at 20℃; for 0.666667h; Stage #2: With phosphorus trichloride In tetrahydrofuran for 1.5h; | 85% |
| Stage #1: 2-methylphenyl bromide With iodine; magnesium In tetrahydrofuran at 20℃; for 2.5h; Schlenk technique; Inert atmosphere; Stage #2: With phosphorus trichloride In tetrahydrofuran at -40℃; for 0.5h; Stage #3: In tetrahydrofuran; ethanol for 5h; | 75% |

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane; magnesium at 20℃; for 4h; | 89% |
| With aluminum (III) chloride; N,N,N,N,-tetramethylethylenediamine; tetrabutyl-ammonium chloride; tert-butylammonium hexafluorophosphate(V) In acetonitrile at 60℃; Inert atmosphere; Glovebox; Electrolysis; | 69% |
| With chloro-trimethyl-silane; tetrabutylammomium bromide; copper; zinc In acetonitrile at 45℃; Electrochemical reaction; Inert atmosphere; | 58% |
| With oxalyl dichloride; hydrogen In chloroform-d1 at 130℃; under 60006 Torr; for 18h; Reagent/catalyst; | 51% |

| Conditions | Yield |
|---|---|
| With white phosphorus; 1,3-dicyano-5-fluoro-2,4,6-tris(diphenylamino)benzene; triethylamine In acetonitrile; benzene for 24h; Inert atmosphere; Glovebox; Sealed tube; Irradiation; | 43% |

-
-
15475-27-1, 4346-39-8
potassium diphenylphosphine

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| Stage #1: di-m-acetato-bis{2-(diortho-tolylphosphino)benzyl-C,P}dipalladium(II); potassium diphenylphosphine In tetrahydrofuran at 20℃; for 48h; Stage #2: With air | A 2% B 30% |

-
-
172418-37-0, 199609-05-7
di-m-chloro-bis{2-(di-ortho-tolylphosphino)benzyl-C,P}dipalladium(II)

-
-
15475-27-1, 4346-39-8
potassium diphenylphosphine

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| Stage #1: di-m-chloro-bis{2-(di-ortho-tolylphosphino)benzyl-C,P}dipalladium(II); potassium diphenylphosphine In tetrahydrofuran at 20℃; for 48h; Schlenk technique; Inert atmosphere; Stage #2: With air In tetrahydrofuran for 48h; Schlenk technique; | A 20% B 2% |


| Conditions | Yield |
|---|---|
| With 1-methyl-pyrrolidin-2-one; potassium hydroxide monohydrate; phosphorus at 100 - 120℃; for 1h; Reagent/catalyst; Temperature; Inert atmosphere; | 19% |

| Conditions | Yield |
|---|---|
| With diethyl ether; hydrogen; phosphorus trichloride |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In chloroform Irradiation (UV/VIS); decompn. at room temp. within minutes; |

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In benzene Kinetics; monitored by UV; |
-
-
1094249-98-5
[(o-C6H4Me)3PH][HB(p-C6F4H)3]

-
A
-
148892-95-9
tris(2,3,5,6-tetrafluorophenyl)borane triethylphosphine oxide

-
B
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| at 25 - 60℃; vacuum; |
-
-
1094249-98-5
[(o-C6H4Me)3PH][HB(p-C6F4H)3]

-
A
-
148892-95-9
tris(2,3,5,6-tetrafluorophenyl)borane triethylphosphine oxide

-
B
-
1333-74-0
hydrogen

-
C
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In neat (no solvent, solid phase) (N2); keeping boron compd. at room temp. in vac. for 4 d; | |
| In not given (N2); heating boron compd. at 60°C for few hs; |

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
32461-51-1
tris(2-methylphenyl)phosphine selenide

| Conditions | Yield |
|---|---|
| With selenium In toluene for 6h; Heating; | 100% |
| With selenium In chloroform for 5h; Heating; | 23% |
| With potassium selenocyanate In methanol at 20℃; | |
| With selenium In toluene at 100℃; for 48h; Inert atmosphere; | |
| With selenium In chloroform-d1 at 70℃; for 16h; Glovebox; Sealed tube; Inert atmosphere; |

-
-
2298-07-9
4-Bromo-1-naphthylamine

-
-
292638-85-8
acrylic acid methyl ester

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
436864-73-2
3-(4-amino-1-naphthyl)propanoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydroxide; potassium acetate; Pd-C; palladium diacetate In methanol; water; N,N-dimethyl-formamide | 100% |
| With hydrogenchloride; sodium hydroxide; potassium acetate; Pd-C; palladium diacetate In methanol; water; N,N-dimethyl-formamide | 100% |

-
-
1122-70-9
2-methyl-6-vinylpyridine

-
-
255847-81-5
1-[(4-cyano-5-methyl-4-phenyl)hexyl]-4-[2-(2-bromophenoxy)ethyl]piperazine

-
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| With triethylamine; palladium diacetate In water; N,N-dimethyl-formamide | 100% |

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
24554-43-6, 157183-46-5
Pd(P(C6H4CH3)3)2(Br)2

| Conditions | Yield |
|---|---|
| With lithium bromide In methanol N2 or Ar-atmosphere; | 100% |
-
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| With tetrafluoroboric acid In diethyl ether at 20℃; Inert atmosphere; | 100% |
-
-
64443-05-6
tetrakis(actonitrile)copper(I) hexafluorophosphate

-
-
77429-58-4
tris(2-pyridyl)methane

-
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| Stage #1: tetrakis(actonitrile)copper(I) hexafluorophosphate; tris(2-pyridyl)methane In dichloromethane for 0.166667h; Inert atmosphere; Stage #2: tris-(o-tolyl)phosphine In dichloromethane at 20℃; for 12h; Inert atmosphere; | 100% |


| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In tetrahydrofuran; water for 0.533333h; | 99% |
| With water; Selectfluor In acetonitrile at 20℃; for 0.0833333h; | 99% |
| With fluorosulfonylchloride In dichloromethane for 1h; Ambient temperature; | 98% |

-
-
21226-24-4
4-(benzenesulfonylamino)phenyl iodide

-
-
79-10-7
acrylic acid

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
501682-73-1
3-[4-(benzenesulfonylamino)phenyl]acrylic acid

| Conditions | Yield |
|---|---|
| With triethylamine; tris-(dibenzylideneacetone)dipalladium(0) In N,N-dimethyl-formamide | 99% |


| Conditions | Yield |
|---|---|
| In n-heptane; dichloromethane in CH2Cl2/heptane=1:10 v/v, excess phosphine, room temp. (UV-control); vol. reduction (Ar-stream), crystn. (-15°C); | 99% |
-
-
148892-95-9
tris(2,3,5,6-tetrafluorophenyl)borane triethylphosphine oxide

-
-
1333-74-0
hydrogen

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
1094249-98-5
[(o-C6H4Me)3PH][HB(p-C6F4H)3]

| Conditions | Yield |
|---|---|
| In not given | 99% |
| In hexane High Pressure; (N2); mixing hexane soln. of phosphine deriv. and borane deriv. in C6H5Br, freezing at -196°C, exposure to H2, warming to room temp., stirring overnight; pressure of H2 after warming to room temp. was 4 atm; evapn., recrystn. by addn. of hexanes to concd. CH2Cl2 soln. and keepingat -35°C for wk, elem. anal.; |

-
-
1154042-85-9, 1154049-74-7
[Pd(1-dicyclohexylphosphino-2-di-tert-butylphosphinoethylferrocene)(H)(4-methoxybenzothiolate)]

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
583872-37-1
[Pd(1-dicyclohexylphosphino-2-di-tert-butylphosphinoethylferrocene)(P(o-tolyl)3)]

| Conditions | Yield |
|---|---|
| With sodium t-butanolate In toluene 1 h, 100°C; | 99% |
-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
878809-28-0
tris(2-tolyl)phosphane-borane(1:1)

| Conditions | Yield |
|---|---|
| Stage #1: tris-(o-tolyl)phosphine With dimethylsulfide borane complex In dichloromethane at 20℃; Inert atmosphere; Stage #2: With ammonium chloride In dichloromethane; water | 99% |

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
28978-10-1
chloro{tris(o-tolyl)phosphine}gold(I)

| Conditions | Yield |
|---|---|
| Stage #1: hydrogen tetrachloroaurate(III) tetrahydrate With propyl sulfide In ethanol at 40℃; for 0.166667h; Stage #2: tris-(o-tolyl)phosphine In ethanol at 40℃; for 1h; | 99% |
-
-
1109-15-5
tris(pentafluorophenyl)borate

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
196953-02-3
1-(4-chlorophenyl)buta-2,3-dien-1-one

| Conditions | Yield |
|---|---|
| In chloroform-d1 Inert atmosphere; Glovebox; | 99% |


| Conditions | Yield |
|---|---|
| With C26H46ClO3Ru2S2(1+)*CF3O3S(1-); hydrogen In dichloromethane at 20℃; under 760.051 Torr; for 11h; Catalytic behavior; Inert atmosphere; | A 99% B 98% |

-
-
1423-15-0
perfluorophenyl azide

-
-
341-02-6
trityl tetrafluoroborate

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
109-66-0
pentane

| Conditions | Yield |
|---|---|
| Stage #1: perfluorophenyl azide; trityl tetrafluoroborate; tris-(o-tolyl)phosphine In dichloromethane at 20℃; for 12h; Inert atmosphere; Schlenk technique; Glovebox; Stage #2: pentane | 99% |

-
-
13472-85-0
2-methoxy-5-bromopyridine

-
-
1663-39-4
tert-Butyl acrylate

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
339555-37-2
tert-butyl (2E)-3-(6-methoxypyridin-3-yl)prop-2-enoate

| Conditions | Yield |
|---|---|
| With sodium hypochlorite; triethylamine; palladium diacetate In toluene | 98.4% |

-
-
1111-67-7, 15192-76-4, 26656-82-6
copper(I) thiocyanate

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
75-05-8
acetonitrile

| Conditions | Yield |
|---|---|
| In acetonitrile CuSCN ground with P(o-Tol)3 dry, then with MeCN; dried in air; detd. by IR; | 98.1% |
| In methanol CuSCN reacted with P(o-Tol)3 in MeOH for 4 h; recrystd. from MeCN; elem. anal.; | 80% |


-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
69861-71-8
bis(tri-ortho-tolylphosphine)palladium(0)

| Conditions | Yield |
|---|---|
| In methanol for 3h; Heating / reflux; | 98% |

| Conditions | Yield |
|---|---|
| In acetone at 25℃; | 98% |
-
-
1165952-92-0
cyclohexa-1,4-diene

-
-
341-02-6
trityl tetrafluoroborate

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
A
-
519-73-3
triphenylmethane

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 0.5h; Inert atmosphere; Schlenk technique; Glovebox; | A 98% B 97% |

-
-
341-02-6
trityl tetrafluoroborate

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
766-97-2
4-n-methylphenylacetylene

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 2h; Inert atmosphere; Schlenk technique; Glovebox; | A 92% B 98% |

-
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide In dichloromethane; water at 0 - 23℃; for 0.5h; Inert atmosphere; Schlenk technique; | 98% |

-
-
292638-85-8
acrylic acid methyl ester

-
-
6163-58-2
tris-(o-tolyl)phosphine

-
-
258532-94-4
methyl (E)-3-(3-oxo-4-(2-(benzyloxy)ethyl)-3,4-dihydro-2H-1,4-benzoxazin-8-yl)acrylate

| Conditions | Yield |
|---|---|
| With triethylamine; lithium chloride; palladium diacetate In N,N-dimethyl-formamide | 97% |


| Conditions | Yield |
|---|---|
| With potassium acetate; palladium diacetate In water; N,N-dimethyl-formamide | 97% |


| Conditions | Yield |
|---|---|
| With lithium iodide In methanol N2 or Ar-atmosphere; refluxing (30 min); filtering (room temp.), washing (MeOH, Et2O), drying (vac.); elem. anal.; | 97% |

-
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; Schlenk technique; Inert atmosphere; Glovebox; | 97% |
-
-
13203-60-6
1,2-bis(2-phenylethynyl)benzene

-
-
1109-15-5
tris(pentafluorophenyl)borate

-
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In pentane at 20℃; | 97% |

-
-
64443-05-6
tetrakis(actonitrile)copper(I) hexafluorophosphate

-
-
14044-48-5
2-(2-quinolyl)benzimidazole

-
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In dichloromethane | 97% |

-
-
1469887-23-7
2-(2-fluoropyridin-1-ium-1-yl)-1,1-bis((trifluoromethyl)sulfonyl)ethan-1-ide

-
-
6163-58-2
tris-(o-tolyl)phosphine

| Conditions | Yield |
|---|---|
| In acetonitrile at 20℃; for 0.25h; Inert atmosphere; | 97% |
Tris(2-methylphenyl)phosphine Specification
The CAS register number of Tris(2-methylphenyl)phosphine is 6163-58-2. It also can be called as Phosphine,tris(2-methylphenyl)- and the IUPAC name about this chemical is tris(2-methylphenyl)phosphane. The molecular formula about this chemical is C21H21P and the molecular weight is 304.37. It belongs to the following product categories, such as Ligand; Phosphine Ligands; Synthetic Organic Chemistry; Benzenes and so on. This chemical is usually used as a ligand for the coupling reaction.
Physical properties about Tris(2-methylphenyl)phosphine are: (1)ACD/LogP: 7.07; (2)# of Rule of 5 Violations: 1; (3)ACD/LogD (pH 5.5): 7.07; (4)ACD/LogD (pH 7.4): 7.07; (5)ACD/BCF (pH 5.5): 139115.45; (6)ACD/BCF (pH 7.4): 139115.45; (7)ACD/KOC (pH 5.5): 167188.17; (8)ACD/KOC (pH 7.4): 167188.17; (9)#Freely Rotating Bonds: 3; (10)Polar Surface Area: 13.59Å2; (11)Flash Point: 214.6 °C; (12)Enthalpy of Vaporization: 63.92 kJ/mol; (13)Boiling Point: 412.4 °C at 760 mmHg; (14)Vapour Pressure: 1.24E-06 mmHg at 25°C.
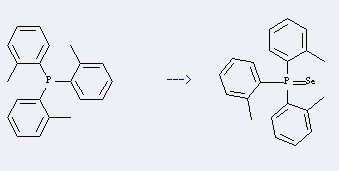
Uses of p-Chloropropiophenone: it can be used to produce Tri-ortho-phosphinselenid at heating. This reaction will need reagent selenium black and solvent toluene with reaction time of 6 hours. The yield is about 100%.
When you are using this chemical, please be cautious about it as the following:
This chemical is irritating to eyes, respiratory system and skin. When you are using it, wear suitable protective clothing, gloves and eye/face protection. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: c3(c(P(c1ccccc1C)c2ccccc2C)cccc3)C
(2)InChI: InChI=1/C21H21P/c1-16-10-4-7-13-19(16)22(20-14-8-5-11-17(20)2)21-15-9-6-12-18(21)3/h4-15H,1-3H3
(3)InChIKey: COIOYMYWGDAQPM-UHFFFAOYAJ
(4)Std. InChI: InChI=1S/C21H21P/c1-16-10-4-7-13-19(16)22(20-14-8-5-11-17(20)2)21-15-9-6-12-18(21)3/h4-15H,1-3H3
(5)Std. InChIKey: COIOYMYWGDAQPM-UHFFFAOYSA-N
Related Products
- Tris(2-methylphenyl)phosphine
- 61636-08-6
- 616-38-6
- 616-39-7
- 61639-74-5
- 616-40-0
- 616-42-2
- 61642-40-8
- 616-43-3
- 61644-00-6
- 61644-18-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View