 This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn't provide relevant sales information.
-
Name
Pentaerythritol
- EINECS 204-104-9
- CAS No. 115-77-5
- Article Data119
- CAS DataBase
- Density 1.396 g/cm3
- Solubility 1 g/18 mL (15 °C) in water
- Melting Point 260.5 °C
- Formula C5H12O4
- Boiling Point 380.4 °C at 760 mmHg
- Molecular Weight 136.148
- Flash Point 200.1 °C
- Transport Information
- Appearance white crystalline solid
- Safety 24/25
- Risk Codes 33
-
Molecular Structure
- Hazard Symbols
- Synonyms Pentaerythritol Tech Grade;Tetrahydroxymethylmethane;1,3-propanediol, 2,2-bis(hydroxymethyl)-;Methane tetramethylol;Pentaerythrite;Pentaerythritol(PE);Pentaerythritol, technical grade;Petaerythritol;Mo-pentaerythritol;New plasticizer Polyol benzoate;Sell Pentaerythritol;pentaerythritol (mono-penta);Mono- Pentaerythritol;Pentaerythritol;;Pentaerythritol(Micronized);Pentaerythritol for Sale;Pentaerithritol;Penetek;Pentek;1,3-Propanediol, 2, 2-bis (hydroxymethyl)-;2,2-bis(hydroxymethyl)propane-1,3-diol;Tetramethylolmethane;1,3-Propanediol,2,2-bis(hydroxymethyl)-;2,2-Bishydroxymethyl-1,3-propanediol;Tetrakis(hydroxymethyl)methane;Mono Pentaerythritol;
- PSA 80.92000
- LogP -2.05800
Synthetic route
-
-
50-00-0
formaldehyd

-
-
64-18-6
formic acid

-
-
75-07-0
acetaldehyde

-
A
-
115-77-5
Pentaerythritol

-
B
-
126-58-9
Dipentaerythritol

-
C
-
141-53-7
sodium formate

| Conditions | Yield |
|---|---|
| Stage #1: formaldehyd; acetaldehyde With sodium hydroxide at 45 - 65℃; under 1500.15 Torr; for 1.38333h; Inert atmosphere; Stage #2: formic acid pH=6; Product distribution / selectivity; | A 87.4% B 5% C 99.7% |


| Conditions | Yield |
|---|---|
| Stage #1: formaldehyd; acetaldehyde With sodium hydroxide In water Stage #2: With formic acid In water pH=5.7; Product distribution / selectivity; | 90% |
| With ortho-tungstic acid; calcium hydroxide In water at 30 - 48℃; for 1.16667h; Reagent/catalyst; | 7.7% |
| With sodium hydroxide at 10℃; for 4.16667h; Product distribution; pH=12.7; different reaction times; |
-
-
6228-26-8
Formaldehyd-bis(pentaerythrityl)acetal

-
A
-
6228-25-7
5,5-bis-hydroxymethyl-[1,3]dioxane

-
B
-
115-77-5
Pentaerythritol

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 80℃; for 2h; Title compound not separated from byproducts; | A 33.8 % Chromat. B 65% |
| With hydrogenchloride at 80℃; for 0.166667h; Title compound not separated from byproducts; | A 9.3 % Chromat. B 88.6 % Chromat. |
| With hydrogenchloride at 60℃; for 2h; Product distribution; further temperatures: 23, 40, 80 deg C; different times: 10, 20, 30, 60, 120 min; | A 21.9 % Chromat. B 76.8 % Chromat. |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; water |

| Conditions | Yield |
|---|---|
| With lead(II) hydroxide on calcium carbonate; water | |
| With calcium hydroxide; water |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; water in mehreren Stufen; |
-
-
5419-74-9
pentaerythritol bisacetate

-
A
-
115-77-5
Pentaerythritol

-
B
-
13051-30-4
pentaerythritol triacetate

-
C
-
597-71-7
pentaerythritol tetracetate

| Conditions | Yield |
|---|---|
| beim Aufbewahren; |

-
-
50-00-0
formaldehyd

-
-
75-07-0
acetaldehyde

-
A
-
115-77-5
Pentaerythritol

-
B
-
6228-26-8
Formaldehyd-bis(pentaerythrityl)acetal

| Conditions | Yield |
|---|---|
| With methyllithium Title compound not separated from byproducts; | |
| With methyllithium; ethyl acetate 2) 1 h, reflux; Yield given. Multistep reaction. Yields of byproduct given. Title compound not separated from byproducts; | |
| als Nebenprodukt bei der Darstellung von Pentaerythrit; Isolierung aus technischen Pentaerytrit; |


| Conditions | Yield |
|---|---|
| With alkali Trennung ueber die Salpersaeureester; | |
| With alkali Trennung von Pentaerythrit ueber die Salpetersaeureester; | |
| With calcium hydroxide; water at 20℃; |


| Conditions | Yield |
|---|---|
| With calcium hydroxide; water |

| Conditions | Yield |
|---|---|
| With calcium hydroxide; water |

| Conditions | Yield |
|---|---|
| 2-(Diethylamino)ethanol In water at 100℃; for 20h; Product distribution; | |
| 2-(Diethylamino)ethanol In water at 100℃; for 20h; | |
| With sodium hydroxide In water at 25℃; for 3h; Irradiation; | |
| With potassium carbonate 1.) H2O, 20 deg C, 10 h, irradiation, 2.) 25 deg C, 7-14 d; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| calcium hydroxide In water at 25℃; Irradiation; | |
| calcium hydroxide In water at 25℃; Product distribution; Irradiation; various inorganic catalysts; |

-
-
50-00-0
formaldehyd

-
A
-
79-14-1
glycolic Acid

-
B
-
115-77-5
Pentaerythritol

-
C
-
909878-64-4
meso-erythritol

-
D
-
473-81-4
glyceric acid

-
E
-
107-21-1
ethylene glycol

-
F
-
56-81-5
glycerol

| Conditions | Yield |
|---|---|
| With zeolite of type X under 1 Torr; for 144h; Product distribution; Irradiation; other objects of study: dependence of product distribution on time of irradiation, thermal pretreatment of zeolite, and water content of zeolite; |

-
-
6228-26-8
Formaldehyd-bis(pentaerythrityl)acetal

-
A
-
6228-25-7
5,5-bis-hydroxymethyl-[1,3]dioxane

-
B
-
50-00-0
formaldehyd

-
C
-
115-77-5
Pentaerythritol

| Conditions | Yield |
|---|---|
| Heating; heating more than 149 deg C; |

-
-
50-00-0
formaldehyd

-
-
75-07-0
acetaldehyde

-
A
-
68516-39-2
2,2-di(hydroxymethyl)-acetic acid

-
B
-
115-77-5
Pentaerythritol

-
C
-
4704-94-3
2-hydroxymethyl-1,3-propanediol

-
D
-
2831-90-5
α,α,α-tris(hydroxymethyl)acetic acid

-
E
-
68658-38-8
2-Hydroxymethyl-2-(methoxymethoxymethyl)-1,3-propandiol

-
F
-
504-63-2
trimethyleneglycol

| Conditions | Yield |
|---|---|
| With calcium hydroxide Mechanism; |

-
-
50-00-0
formaldehyd

-
-
75-07-0
acetaldehyde

-
A
-
115-77-5
Pentaerythritol

-
B
-
3818-32-4
tri(hydroxymethyl)acetaldehyde

-
C
-
126-58-9
Dipentaerythritol

-
D
-
78-24-0
tripentaerythritol

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 30℃; Product distribution; Mechanism; pH=12.5; different initial acetaldehyde concentrations; |

-
-
50-00-0
formaldehyd

-
-
75-07-0
acetaldehyde

-
A
-
115-77-5
Pentaerythritol

-
B
-
107-89-1
acetaldol

-
C
-
126-58-9
Dipentaerythritol

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 10℃; for 5h; Product distribution; Mechanism; different initial aldehyde concentrations and reaction times; effect of additional pentaerythritol; |

-
-
50-00-0
formaldehyd

-
-
75-07-0
acetaldehyde

-
A
-
115-77-5
Pentaerythritol

-
B
-
2134-29-4
3-Hydroxypropanal

-
C
-
40364-80-5
bis-(hydroxymethyl)-acetaldehyde

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate at 30℃; for 2.5h; Product distribution; |

-
-
50-00-0
formaldehyd

-
-
75-07-0
acetaldehyde

-
A
-
115-77-5
Pentaerythritol

-
B
-
4376-78-7
Tetrapentaerythrit 2,2,6,6,10,10,14,14-Octakis(hydroxymethyl)-4,8,12-trioxapentadecan-1,15-diol

| Conditions | Yield |
|---|---|
| With methyllithium Yield given. Title compound not separated from byproducts; |

-
-
50-00-0
formaldehyd

-
-
75-07-0
acetaldehyde

-
A
-
115-77-5
Pentaerythritol

-
B
-
2163-42-0
2-methyl-1.3-propanediol

-
C
-
4704-94-3
2-hydroxymethyl-1,3-propanediol

-
D
-
504-63-2
trimethyleneglycol

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sulfuric acid; hydrogen; 5% activated charcoal-supported ruthenium catalyst 1.) 25 deg C, 30 min, 2.) 50 psi, 25 deg C, 4 h; Yield given. Multistep reaction. Yields of byproduct given. Title compound not separated from byproducts; | |
| With sodium hydroxide; sulfuric acid; hydrogen; Ru-carbon 1.) 25 deg C, 30 min, 2.) 50 psi, 25 deg C 4 h; Yield given. Multistep reaction. Yields of byproduct given; |



| Conditions | Yield |
|---|---|
| bei monatelangem Stehen; |


| Conditions | Yield |
|---|---|
| je nach Bedingungen wechselnden Mengen; |


| Conditions | Yield |
|---|---|
| at 0 - 40℃; Kinetics; |



-
-
50-00-0
formaldehyd

-
-
75-07-0
acetaldehyde

-
A
-
115-77-5
Pentaerythritol

-
B
-
6228-26-8
Formaldehyd-bis(pentaerythrityl)acetal

-
C
-
126-58-9
Dipentaerythritol

| Conditions | Yield |
|---|---|
| In water at 10℃; Rate constant; Mechanism; Product distribution; and equilibrium constants; |

-
-
115-77-5
Pentaerythritol

-
-
108-94-1
cyclohexanone

-
-
183-10-8
7,11,18,21-tetraoxa-trispiro[5.2.2.5.2.2]heneicosane

| Conditions | Yield |
|---|---|
| With sulfuric acid at 80℃; | 100% |
| With pyrographite In toluene for 8h; Heating; | 94% |
| peroxodisulfate ion; zirconium(IV) oxide In toluene for 4h; Heating; | 94% |
-
-
115-77-5
Pentaerythritol

-
-
78-84-2
isobutyraldehyde

-
-
69695-04-1
3,9-diisopropyl-2,4,8,10-tetraoxaspiro<5.5>undecane

| Conditions | Yield |
|---|---|
| With sulfuric acid at 80℃; | 100% |
| With indium(III) chloride at 120℃; for 3h; | 89% |
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| In diethyl ether; dichloromethane; tetramethyl ammoniumhydroxide; water | 100% |
| tetramethyl ammoniumhydroxide In water at 20℃; for 20h; Cooling with ice; | 100% |
| With sodium hydroxide Ambient temperature; | 99% |
-
-
115-77-5
Pentaerythritol

-
-
170996-99-3
[2,2-bis(trifluoromethyl)-5-oxo-1,3-oxathiolan-4-yl]-acetyl chloride

| Conditions | Yield |
|---|---|
| In toluene for 72h; Heating; | 100% |
-
-
115-77-5
Pentaerythritol

-
-
103-71-9
phenyl isocyanate

-
-
125614-20-2
tetrakis[(N-phenylcarbamoyloxy)methyl]methane

| Conditions | Yield |
|---|---|
| With MoCl2O2(dmf)2 In N,N-dimethyl-formamide at 60℃; for 0.333333h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| In methanol for 4h; Reflux; | 100% |
| In methanol for 4h; Reflux; | |
| In methanol for 4h; Reflux; |

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; potassium hydroxide In toluene at 70℃; for 18h; | 99.6% |

| Conditions | Yield |
|---|---|
| With pyridine at 0 - 20℃; for 48h; | 99.5% |
| With pyridine at 0 - 20℃; for 48h; | 99.5% |
| With pyridine at 20℃; for 16h; | 89% |
-
-
115-77-5
Pentaerythritol

-
-
100-52-7
benzaldehyde

-
-
2064-95-1
3,9-diphenyl-2,4,8,10-tetraoxa-spiro[5,5]undecane

| Conditions | Yield |
|---|---|
| With pyrographite In benzene for 1.5h; Heating; | 99% |
| With toluene-4-sulfonic acid In benzene for 4h; Condensation; acetalization; Heating; | 99% |
| With montmorillonite K-10 In benzene for 0.6h; Heating; | 98% |
-
-
115-77-5
Pentaerythritol

-
-
123-11-5
4-methoxy-benzaldehyde

-
-
1130782-84-1, 1130782-85-2
3,9-di(4-methoxyphenyl)-2,4,8,10-tetraoxaspiro<5,5>undecane

| Conditions | Yield |
|---|---|
| With Sulfate; zirconium(IV) oxide In toluene for 0.8h; Heating; | 99% |
| With pyrographite In benzene for 1.8h; Heating; | 96% |
| With tetrachlorosilane In acetonitrile at 20℃; | 95% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide at 120℃; for 5h; | 99% |
| (i) /BRN= 906677/, aq. KOH, (ii) /BRN= 906677/, KOEt, EtOH; Multistep reaction; | |
| With potassium hydroxide In dimethyl sulfoxide at 100℃; under 759.8 Torr; | |
| With potassium hydroxide In dimethyl sulfoxide |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrachloromethane for 16h; Ambient temperature; | 99% |

| Conditions | Yield |
|---|---|
| With Sulfate; zirconium(IV) oxide In toluene for 1.5h; Heating; | 99% |
-
-
115-77-5
Pentaerythritol

-
-
865835-76-3
C6H5CH2OCH2CH2CH2CB10H10CCH2CH2COOH

| Conditions | Yield |
|---|---|
| With N-(3-dimethylaminopropyl)-N-ethylcarbodiimide; 4-(dimethylamino)pyridinium tosylate In pyridine; dichloromethane CH2Cl2-pyridine 3:2, room temp., > 12 h; column chromy.; | 99% |
-
-
1189-71-5
isocyanate de chlorosulfonyle

-
-
115-77-5
Pentaerythritol

-
-
24794-44-3
pentaerythritol tetracarbamate

| Conditions | Yield |
|---|---|
| Stage #1: isocyanate de chlorosulfonyle; Pentaerythritol In acetonitrile at 0 - 20℃; for 1h; Stage #2: With water In acetonitrile Cooling with ice; | 99% |
| Stage #1: isocyanate de chlorosulfonyle; Pentaerythritol In acetonitrile at 25℃; for 2h; Cooling with ice; Stage #2: With water In acetonitrile at 20℃; for 1h; Cooling with ice; |

| Conditions | Yield |
|---|---|
| Stage #1: Pentaerythritol; cis-Octadecenoic acid In water at 130 - 135℃; Inert atmosphere; Stage #2: butanedioic acid, monoethyl ester In water at 130 - 135℃; for 2h; Inert atmosphere; | 99% |


| Conditions | Yield |
|---|---|
| Stage #1: Pentaerythritol; cis-Octadecenoic acid In water at 130 - 135℃; Inert atmosphere; Stage #2: methyl hydrogen succinate In water at 130 - 135℃; for 2h; Inert atmosphere; | 99% |

-
-
115-77-5
Pentaerythritol

-
-
552-89-6
2-nitro-benzaldehyde

-
-
102174-89-0
3,9-bis-(2-nitro-phenyl)-2,4,8,10-tetraoxa-spiro[5.5]undecane

| Conditions | Yield |
|---|---|
| With copper(II) chloride dihydrate In N,N-dimethyl-formamide at 70℃; for 1.33333h; | 98.8% |
| With toluene-4-sulfonic acid In toluene at 140℃; for 3h; | 98.8% |
| With toluene-4-sulfonic acid for 0.0666667h; microwave irradiation at 600 W power; | 97.5% |
-
-
115-77-5
Pentaerythritol

-
-
4095-10-7
dimethyldibromosilane

-
-
14896-25-4
3,3,9,9,-tetramethyl-3,9-disila-2,4,8,10-tetraoxaspiro<5,5>undecane

| Conditions | Yield |
|---|---|
| In diethylene glycol dimethyl ether at 50 - 130℃; for 6h; Solvent; Temperature; | 98.6% |

| Conditions | Yield |
|---|---|
| With zinc/copper couple at 160℃; under 600.06 Torr; for 2.33333h; Temperature; Pressure; Reagent/catalyst; | 98.3% |
| at 240℃; | |
| With phenol at 180 - 230℃; |

| Conditions | Yield |
|---|---|
| With lithium hydroxide; triethyl phosphite at 130 - 160℃; for 2h; Temperature; Reagent/catalyst; Time; | 98.2% |
-
-
115-77-5
Pentaerythritol

-
-
19759-75-2
3-(n-dodecylthio)-propanoic acid,methyl ester

-
-
29598-76-3
pentaerythritol tetrakis(β-dodecylmercapto)propionate

| Conditions | Yield |
|---|---|
| With [BCl2(1-methylimidazole)][Al2Cl7] at 120℃; for 3h; Reagent/catalyst; Temperature; Large scale; | 98.2% |

| Conditions | Yield |
|---|---|
| With methanesulfonic acid at 25℃; for 2h; Inert atmosphere; | 98% |
| With aluminum triflate at 20℃; for 0.00833333h; | 95% |
| aluminium dodecatungsten phosphate at 20℃; for 0.583333h; | 94% |
-
-
115-77-5
Pentaerythritol

-
-
104-87-0
4-methyl-benzaldehyde

-
-
53731-74-1
3,9-di-p-tolyl-2,4,8,10-tetraoxa-spiro[5.5]undecane

| Conditions | Yield |
|---|---|
| With Sulfate; zirconium(IV) oxide In toluene for 1h; Heating; | 98% |
| With pyrographite In benzene for 1.5h; Heating; | 97% |
| With tetrachlorosilane In acetonitrile at 20℃; | 96% |
-
-
115-77-5
Pentaerythritol

-
-
124-63-0
methanesulfonyl chloride

-
-
7511-62-8
2,2-bis(((methylsulfonyl)oxy)methyl)propane-1,3-diyl dimethanesulfonate

| Conditions | Yield |
|---|---|
| With pyridine Cooling with ice; Inert atmosphere; | 98% |
| Stage #1: Pentaerythritol With triethylamine In dichloromethane at 0℃; for 0.166667h; Stage #2: methanesulfonyl chloride In dichloromethane at 20℃; for 12h; | 87.3% |
| With pyridine | |
| With pyridine Inert atmosphere; Schlenk technique; | |
| With triethylamine In toluene at 0 - 20℃; Dean-Stark; |

| Conditions | Yield |
|---|---|
| With pyridine; <(chlorosulfinyloxy)methylene>dimethylammonium chloride In dichloromethane at 20℃; for 6h; | 98% |
| With sulfuric acid; toluene-4-sulfonic acid at 105℃; for 0.15h; Neat (no solvent); Microwave irradiation; | 96% |
| at 180℃; | |
| In toluene at 158℃; for 10h; Catalytic behavior; Reagent/catalyst; Green chemistry; | 98 %Spectr. |
| With (cetyltrimethylammonium)1/3[Cu46(C9H3O6)24(OH)12](PW12O40)3·xH2O (COK-15b) In toluene at 155℃; for 6h; Green chemistry; | 94 %Spectr. |
-
-
16967-79-6
trichloroethylene epoxide

-
-
115-77-5
Pentaerythritol

-
-
33216-80-7
tetra(dichloroacetato)pentaerythritol

| Conditions | Yield |
|---|---|
| With magnesium sulfate for 6h; Heating; | 98% |
-
-
115-77-5
Pentaerythritol

-
-
587-04-2
3-Chlorobenzaldehyde

-
-
35583-09-6
3,9-di(m-chlorophenyl)-2,4,8,10-tetraoxaspiro[5.5]undecane

| Conditions | Yield |
|---|---|
| peroxodisulfate ion; zirconium(IV) oxide In toluene for 0.5h; Heating; | 98% |
| silica sulfate In benzene for 0.5h; microwave irradiation; | 98% |
| With pyrographite In toluene for 0.8h; Heating; | 97% |
-
-
115-77-5
Pentaerythritol

-
-
149-57-5
2-Ethylhexanoic acid

-
-
7299-99-2
pentaerythritol tetra(2-ethylhexanoate)

| Conditions | Yield |
|---|---|
| With pyridine; <(chlorosulfinyloxy)methylene>dimethylammonium chloride In dichloromethane at 20℃; for 6h; | 98% |
| With sulfuric acid | 82% |
| Stage #1: Pentaerythritol; 2-Ethylhexanoic acid at 170 - 250℃; for 6h; Stage #2: With 2-ethylhexanoic acid anhydride at 150 - 170℃; for 0.5h; Reagent/catalyst; |
pentaerythritol Specification
The pentaerythritol, with the CAS registry number 115-77-5,is also known as Tetrakis(hydroxymethyl)methane; 2,2-Bis(hydroxymethyl)-1,3-propanediol. It belongs to the product categories of Inorganic & organic chemicals;Organics. Its EINECS number is 204-104-9. This chemical's molecular formula is C5H12O4 and molecular weight is 136.15. What's more,Its systematic name is pentaerythritol.It is a odorless white solid,sinks and mixes slowly with water,stable,incompatible with strong acids,strong oxidizing agents, acid chlorides, acid anhydrides,combustible.It is danger of cumulative effects, so you should avoid contact with skin and eyes.
Physical properties about pentaerythritol are:
(1)ACD/LogP: -2.592; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -2.59; (4)ACD/LogD (pH 7.4): -2.59; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 1.00; (8)ACD/KOC (pH 7.4): 1.00; (9)#H bond acceptors: 4; (10)#H bond donors: 4; (11)#Freely Rotating Bonds: 8; (12)Index of Refraction: 1.532; (13)Molar Refractivity: 31.319 cm3; (14)Molar Volume: 101.141 cm3; (15)Surface Tension: 70.5199966430664 dyne/cm; (16)Density: 1.346 g/cm3; (17)Flash Point: 200.145 °C; (18)Enthalpy of Vaporization: 72.706 kJ/mol; (19)Boiling Point: 380.408 °C at 760 mmHg; (20)Vapour Pressure: 0 mmHg at 25°C.
Preparation of Pentaerythritol:
Pentaerythritol is manufactured by reaction of formaldehyde and acetaldehyde in the presence of a basic catalyst, generally an alkali or alkaline-earth hydroxide.
In the process (Fig. 1), the main concern in mixing is to avoid loss of temperature control in this exothermic reaction, which can load to excessive by-product formation and/or reduced yields of pentaerythritol. The reaction time depends on the reaction temperature and may vary from about 0.5 to 4 hours at final temperatures of about 65 and 35 °C, respectively. The reactor product, neutralized with acetic or formic acid, is then stripped of excess formaldehyde and water to produce a highly concentrated solution of pentaerythritol reaction products. This is then cooled under carefully controlled crystallization conditions so that the crystals can be readily separated from the liquors by subsequent filtration.
Staged reactions, where only part of the initial reactants is added, either to consecutive reactors or with a time lag to the same reactor, may be used to reduce dipentaerythritol content. This technique increases the effective formaldehyde-to-acetaldehyde mole ratio, maintaining the original stoichiometric one. It also permits easier thermal control of the reaction. Both batch and continuous reaction systems are used.
Dipentaerythritol and tripentaerythritol are obtained as by-products of the pentaerythritol process and may be further purified by fractional crystallization or extraction.
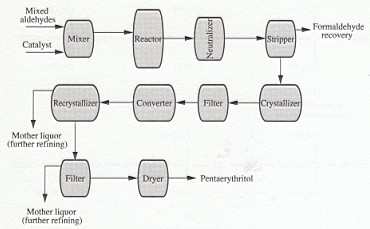
FIGURE 1: Manufacture of pentaerythritol
You can still convert the following datas into molecular structure:
(1)SMILES:OCC(CO)(CO)CO;
(2)Std. InChI:InChI=1S/C5H12O4/c6-1-5(2-7,3-8)4-9/h6-9H,1-4H2;
(3)Std. InChIKey:WXZMFSXDPGVJKK-UHFFFAOYSA-N.
The toxicity data of pentaerythritol as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | oral | 11300mg/kg (11300mg/kg) | BEHAVIORAL: TREMOR BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) BEHAVIORAL: ATAXIA | Toxicology and Applied Pharmacology. Vol. 6, Pg. 351, 1964. |
| mouse | LD50 | oral | 25500mg/kg (25500mg/kg) | BEHAVIORAL: ATAXIA BEHAVIORAL: TREMOR BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) | Toxicology and Applied Pharmacology. Vol. 6, Pg. 351, 1964. |
| rabbit | LD50 | oral | 18500mg/kg (18500mg/kg) | BEHAVIORAL: GENERAL ANESTHETIC GASTROINTESTINAL: ULCERATION OR BLEEDING FROM STOMACH LIVER: OTHER CHANGES | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 36(2), Pg. 25, 1971. |
| rat | LC | inhalation | > 11gm/m3/6H (11000mg/m3) | Toxicology and Applied Pharmacology. Vol. 6, Pg. 351, 1964. | |
| rat | LD50 | oral | 19500mg/kg (19500mg/kg) | LIVER: OTHER CHANGES BEHAVIORAL: GENERAL ANESTHETIC GASTROINTESTINAL: ULCERATION OR BLEEDING FROM STOMACH | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 36(2), Pg. 25, 1971. |
Related Products
- pentaerythritol
- Pentaerythritol dibehenate
- Pentaerythritol dinitrate
- PENTAERYTHRITOL GLYCIDYL ETHER
- Pentaerythritol rosinate
- Pentaerythritol tetra(2-ethylhexanoate)
- Pentaerythritol tetraacetate
- Pentaerythritol tetraacrylate
- Pentaerythritol tetrakis(2-mercaptoacetate)
- Pentaerythritol tetrakis(3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate)
- 115783-35-2
- 1157834-49-5
- 1157-84-2
- 1157859-81-8
- 115-78-6
- 1157869-05-0
- 115787-50-3
- 115794-67-7
- 115-79-7
- 115-80-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View