Lonwin Chemical Group Limited
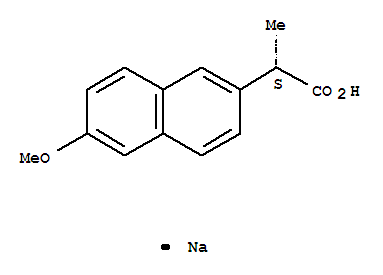
Naproxen sodium CAS NO 26159-34-2 at competitive price Product name:naproxen sodium CAS:26159-34-2 Assay:99% Standard:GMP&FDA approval naproxen sodium Another Name: (S)-6-Methoxy-alpha-methyl-2-naphthaleneacetic acid sodi
Hebei Kunsui Technology Co., Ltd.
1. A strong scientific research team . 2. Stable quality (with a complete scientific research center and testing center to ensure the quality stability of each batch of products). 3. Rich export experience (with practical experience in exporting to m
Cas:22204-53-1
Min.Order:1 Kilogram
FOB Price: $5.6 / 5.8
Type:Trading Company
inquiryHangzhou Dingyan Chem Co., Ltd
Items Standard Result Assay (Ursolic acid) 98%min 98.22% -----------------------------------------------------------------------------------------------------------
Cas:22204-53-1
Min.Order:1 Gram
FOB Price: $100.0 / 500.0
Type:Trading Company
inquiryHangzhou JINLAN Pharm-Drugs Technology Co., Ltd
We can provide GMP validation service that complies with SFDA, FDA, WHO and EU EMPA.Excellent registration team could help us easlily to register our products in different countries.If you and your customer are interested in some products or need C
Cas:22204-53-1
Min.Order:1 Gram
Negotiable
Type:Manufacturers
inquiryWuhan Fortuna Chemical Co.,Ltd
Unique advantages of Naproxen Cas 22204-53-1 Guaranteed purity High quality & competitive price Quality control Fast feedback Prompt shipment Appearance:White or off-white crystalline powder Storage:Cool dry place Package:25kg/dru
Cas:22204-53-1
Min.Order:25 Kilogram
Negotiable
Type:Trading Company
inquiryHefei TNJ chemical industry co.,ltd
LinkMan ALbert Yu Cellphone +8613696510409 Skype albertyu CAS No.: 22204-53-1 Other Names: DL-Naproxen MF: C14H14O3 EINECS No.: 244-
Henan Allgreen Chemical Co.,Ltd
he company has advanced technology, as well as a large number of excellent R & D team, to provide customers from the grams to one hundred kilograms and tons of high-quality products, competitive prices and quality se T rvice Appeara
Cas:22204-53-1
Min.Order:100 Gram
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:22204-53-1
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryDayang Chem (Hangzhou) Co.,Ltd.
Dayangchem’s R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. DayangChem can provide different quantiti
Chemwill Asia Co., Ltd.
Cas:22204-53-1
Min.Order:1 Metric Ton
FOB Price: $1.0
Type:Manufacturers
inquiryHubei DiBo chemical co., LTD
Name:Naproxene CAS NO:22204-53-1 Grade:Medical scientific research and export Molecular formula:C14H13O3 Molecular weight:229.2518 Product Quality 12 years of chemical raw materials Mature operation of the industry System stability Data s
Cas:22204-53-1
Min.Order:25 Kilogram
FOB Price: $2.0 / 3.0
Type:Other
inquiryHebei Nengqian Chemical Import and Export Co., LTD
With our good experience, we offer detailed technical support and advice to assist customers. We communicate closely with customers to establish their quality requirements. Consistent Quality Our plant has strict quality control in each manufacturin
Cas:22204-53-1
Min.Order:1 Kilogram
FOB Price: $3.0 / 10.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:22204-53-1
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Henan Sinotech Import&Export Corporation
Name: Naproxen Formula: C14H14O3 Molecular Weight: 230.26 Synonyms: 2-Naphthaleneacetic acid, 6-methoxy-α-methyl-, (S)-;Naprontag;(2S)-2-(6-methoxynaphthalen-2-yl)propanoic acid;Nafasol;Proxen LE;(+)-2-(Methoxy-2-naphth
Hubei Langyou International Trading Co., Ltd
Our Adwantage: 1.We have stock so we can delivery quickly at the very day when receive the payment. 2.Best price, first class service, high successful delivery rate. A discount would be given when you make a large order. 3.High quality gua
Cas:22204-53-1
Min.Order:10 Gram
Negotiable
Type:Other
inquiryHANWAYS CHEMPHARM CO.,LIMITED
1.Prompt Goods; 2.Flexible payment terms; 3.Documents & Certificate support. Name: Naproxene Molecular Structure: Formula: C14H14O3 Molecular Weight : 230.26 Synonyms: ()-2-(methoxy-2-naphthyl)-propionicacid;()-2-(methoxy-2-naphthyl)-prop
Cas:22204-53-1
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:22204-53-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryXi'an Faithful Biotech Co., Ltd.
We are the manufacturers and suppliers of API in China, and warehouse in Germany and USA of California, which can quickly and safely deliver to your address 1.High quality and competitive price. 2.Free sample for your evaluation. 3.Promptly delivery
Cas:22204-53-1
Min.Order:10 Gram
FOB Price: $0.6
Type:Trading Company
inquiryHangzhou Fonlynn Health Technology Co., Ltd.
Packing: According to customer requirements Delivery time: In stock or depands Port of shipment: Ningbo/Shanghai/Qingdao OEM/ODM:Welcome Sample:We can offer our existing samples at once Appearance:White crystal powder / Refer to COA Storage:Ref
Cas:22204-53-1
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present
Cas:22204-53-1
Min.Order:1 Kilogram
FOB Price: $34.0 / 38.0
Type:Trading Company
inquiryQingdao Beluga Import and Export Co., LTD
Naproxen CAS:22204-53-1 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates, s
Cas:22204-53-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
LIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the R&
Cas:22204-53-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hebei Mojin Biotechnology Co.,Ltd
Hebei Mojin Biotechnology Co., Ltd, Our company is a professional in lead acetate, diphenyl ethylamine and other chemical raw materials and chemical reagents research and development production enterprises. Our business covers more than 30 countrie
Cas:22204-53-1
Min.Order:1 Kilogram
FOB Price: $100.0
Type:Trading Company
inquiryHangzhou Maytime Bio-Tech Co.,Ltd.
Our advance: 1. higest quality and good package 2.fast delivery 3.better payment term 4.fast response to customer within 6 hours 5.good business credit in Global. Hangzhou Maytime Bio-Tech Co.,Ltd was was founded in 2010, the main market in So
Triumph International Development Limilted
Appearance:white powder/crystal Storage:Store in a cool,dry place and keep away from direct strong light Package:As customer request Application:Used for research and industrial manufacture. Transportation:Common products:Sea/Ai
Afine Chemicals Limited
Our Services 1. New Molecules R&D 2. Own test center HPLC NMR GC LC-MS 3. API and Intermediates from China reputed manufacturers 4. Documents support COA MOA MSDS DMF open part Our advantages 1. Government awarded company. Top 100 enter
Synthetic route

-
B
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water | A 100% B n/a |
-
-
23981-80-8
naproxen

-
A
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
B
-
23979-41-1
(R)-2-(6-methoxy-2-naphthyl)propionic acid

| Conditions | Yield |
|---|---|
| With 1-deoxy-1-(octylamino)-d-glucitol In acetonitrile for 6h; Heating; | A 99% B n/a |
| With phosphate buffer; sodium dodecyl-sulfate; vancomycin at 20℃; Product distribution; electrophoretic enantioseparations, various substrates; | |
| With ammonium acetate Purification / work up; Resolution of racemate; |

-
-
27602-79-5
2-(6-methoxynaphth-2-yl)acrylic acid

-
-
7440-44-0
pyrographite

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With triethylamine | 98% |

-
-
26159-34-2
Naprelan

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol at 20℃; | 98% |
| With hydrogenchloride In water pH=< 2; Inert atmosphere; | 94.3% |

-
A
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
B
-
55375-92-3
(R)-(-)-1,2,3,6,7,11b-hexahydro-4H-pyrazino[2,1-a]isoquinolin-4-one

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 20℃; for 0.5h; pH=1; | A 97.9% B n/a |
-
-
27602-79-5
2-(6-methoxynaphth-2-yl)acrylic acid

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With C48H50Cl4N2O2P2Ru3; hydrogen; sodium hydrogencarbonate In methanol at 20℃; under 3750.38 Torr; for 24h; Autoclave; enantioselective reaction; | 97% |
| With hydrogen; (S)-Ru(OAc)2-BINAP In methanol at 15 - 30℃; under 102600 Torr; for 12h; | 92% |
| With C42H48CoP2; hydrogen; triethylamine; zinc In methanol at 50℃; under 25858.1 Torr; for 16h; enantioselective reaction; | 91% |

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 2-(1-bromoethyl)-2-(6-methoxylnaphth-2-yl)-5,5-dimethyl-1,3-epoxyhexane With zinc 2-(6-methoxylnaphthyl)propionate In toluene for 5.5h; Reflux; Stage #2: With sodium hydroxide In toluene for 3.5h; Reflux; Stage #3: With water; pyrographite In toluene for 0.5h; Reflux; | 96.9% |

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water | 96.3% |
-
-
23981-47-7
6-methoxy-2-naphthylacetic acid

-
-
74-88-4
methyl iodide

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 6-methoxy-2-naphthylacetic acid With n-butyllithium; N1,N3-bis((R)-1-phenyl-2-(piperidin-1-yl)ethyl)propane-1,3-diamine In tetrahydrofuran; hexanes at 0℃; Inert atmosphere; Stage #2: methyl iodide In tetrahydrofuran; hexanes at -90℃; for 0.0333333h; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 96% |
| Stage #1: 6-methoxy-2-naphthylacetic acid With n-butyllithium; N1,N3-bis((R)-1-phenyl-2-(piperidin-1-yl)ethyl)propane-1,3-diamine In tetrahydrofuran; hexane at -78 - 0℃; for 0.333333h; Inert atmosphere; Stage #2: methyl iodide In tetrahydrofuran; hexane at -78℃; for 0.0333333h; Inert atmosphere; enantioselective reaction; | 94% |
-
-
32305-59-2
S-2-(6-methoxy-2-naphthyl)propionaldehyde

-
-
109-68-2
2-pentene

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With sodium dihydrogenphosphate; sodium chlorite In water at 20℃; for 1h; | 96% |

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid In 1,4-dioxane; water at 80℃; for 8h; | 96% |
-
-
126899-42-1
2-(6-Methoxy-naphthalen-2-yl)-2-methyl-malonic acid

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With tris hydrochloride In water at 37℃; for 24h; pH=7.0; Reagent/catalyst; Enzymatic reaction; enantioselective reaction; | 95% |
-
-
1330660-68-8
(S)-3-(2-(6-methoxynaphthalen-2-yl)propanoyl)oxazolidin-2-one

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With water; dihydrogen peroxide; lithium hydroxide In tetrahydrofuran at 20℃; for 12h; | 94% |
| Stage #1: (S)-3-(2-(6-methoxynaphthalen-2-yl)propanoyl)oxazolidin-2-one With lithium hydroxide monohydrate In tetrahydrofuran; water at 20℃; for 3h; Stage #2: With hydrogenchloride In tetrahydrofuran; water pH=4; |

-
-
27602-79-5
2-(6-methoxynaphth-2-yl)acrylic acid

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| In methanol | 93.7% |

-
-
72658-10-7
tert-butyl trimethylsilyl methyl ketene acetal

-
-
129731-74-4
6-methoxynaphthalen-2-yl trifluoromethanesulfonate

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: tert-butyl trimethylsilyl methyl ketene acetal; 6-methoxynaphthalen-2-yl trifluoromethanesulfonate With (R)-2-(dicyclohexylphosphino)-2'-(2-naphthylmethoxy)-5,5',6,6',7,7',8,8'-octahydro-1,1'-binaphthyl; lithium acetate In dodecane; α,α,α-trifluorotoluene at 50℃; for 60h; Inert atmosphere; Stage #2: In dichloromethane; trifluoroacetic acid optical yield given as %ee; | 93% |
| Stage #1: tert-butyl trimethylsilyl methyl ketene acetal; 6-methoxynaphthalen-2-yl trifluoromethanesulfonate With (N,N,N',N'-tetramethylethylenediamine)dimethylpalladium(II); (R)-2-(dicyclohexylphosphino)-2'-(2-naphthylmethoxy)-5,5',6,6',7,7',8,8'-octahydro-1,1'-binaphthyl; lithium acetate at 50℃; for 60h; Inert atmosphere; Stage #2: With trifluoroacetic acid In dichloromethane at 25℃; for 4h; | 93% |
-
-
26159-36-4
naproxol

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; laccase from Trametes versicolor; oxygen In water; acetone at 20℃; for 168h; Enzymatic reaction; | 92% |
| With Iron(III) nitrate nonahydrate; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; potassium chloride; oxygen In 1,2-dichloro-ethane at 25℃; Schlenk technique; | 90% |
| With chromium(VI) oxide; sulfuric acid In acetone at 0℃; for 0.5h; | 69% |
-
-
31220-35-6
(S)-6-methoxy-α-methyl-2-naphthaleneacetic acid ethyl ester

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide In 1,4-dioxane at 60℃; for 2h; pH=10 - 14; | 92% |
| With monoclonal antibody N116-27; tris hydrochloride; sodium chloride In acetonitrile at 37℃; pH=8.2; Enzyme kinetics; |

-
-
27602-79-5
2-(6-methoxynaphth-2-yl)acrylic acid

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| In methanol | 92% |

-
-
27655-95-4
(d,l)-2-(5-bromo-6-methoxy-2-naphthyl)propionic acid

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; aluminium trichloride In chloroform; water; toluene; 1,3,5-trimethyl-benzene | 91% |
| With hydrogenchloride; aluminium trichloride In chloroform; water; toluene; 1,3,5-trimethyl-benzene | 91% |
-
-
1181399-48-3
(S)-phenethyl 2-(6-methoxynaphthalen-2-yl)propanoate

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; ammonium formate; palladium diacetate In methanol at 65℃; for 17h; | 91% |
-
-
32305-59-2
S-2-(6-methoxy-2-naphthyl)propionaldehyde

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With sodium chlorite; sodium dihydrogenphosphate; 2-methyl-but-2-ene; tert.-butyl lithium In water; tert-butyl alcohol at 20℃; for 1h; | 91% |
| With sodium chlorite; disodium hydrogenphosphate; 2-methyl-but-2-ene In water; tert-butyl alcohol for 1h; | 90% |
| With sodium chlorite; potassium dihydrogenphosphate; 2-methyl-but-2-ene In water; tert-butyl alcohol at 20℃; for 20.8h; | 184.7 mg |
-
-
26159-35-3
(S)-naproxen methyl ester

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water; acetic acid at 85℃; | 90% |
| Acid hydrolysis; | |
| With sodium hydroxide for 3h; Heating; | |
| With naproxen esterase(NP); water Enzymatic reaction; |

-
A
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
B
-
95530-58-8
(R)-4-isopropyloxazolidin-2-one

| Conditions | Yield |
|---|---|
| With lithium hydroxide; dihydrogen peroxide In tetrahydrofuran; water at 20℃; for 12h; | A 74% B 90% |
-
-
878194-08-2
(4R,2'S)-3-[2'-(6-methoxy-naphthalen-2-yl)propionyl]-4-phenyl-oxazolidin-2-one

-
A
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
B
-
90319-52-1
(4R)-phenyl-2-oxazolidinone

| Conditions | Yield |
|---|---|
| Stage #1: (4R,2'S)-3-[2'-(6-methoxy-naphthalen-2-yl)propionyl]-4-phenyl-oxazolidin-2-one With lithium hydroxide monohydrate; dihydrogen peroxide In tetrahydrofuran; water at 20℃; for 12h; Inert atmosphere; Stage #2: With hydrogenchloride In water pH=3; Inert atmosphere; | A 90% B 89% |
-
-
301663-72-9
(2S,3R,4S)-2,3-Dihydroxy-4-(6-methoxynaphthalen-2-yl)-2-phenylpentane

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: (2S,3R,4S)-2,3-Dihydroxy-4-(6-methoxynaphthalen-2-yl)-2-phenylpentane With sodium periodate In dichloromethane for 1h; Oxidation; oxidative cleavage; Stage #2: With jones reagent In acetone for 1h; Oxidation; Further stages.; | 88% |

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: (S)-2-(6-Methoxy-naphthalen-2-yl)-propionic acid (2R,3S,4S,5S)-2,3,4,5,6-pentahydroxy-hexyl ester With hydroxyl chlorite In methanol for 2h; Heating; Stage #2: With sodium hydroxide In water | 87% |
-
-
197143-65-0
(S)-4-tert-Butyl-3-[(S)-2-(6-methoxy-naphthalen-2-yl)-propionyl]-oxazolidin-2-one

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With lithium hydroxide; dihydrogen peroxide In tetrahydrofuran at 25 - 30℃; for 1h; | 85.4% |
-
-
186581-53-3, 908094-01-9
diazomethane

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
26159-35-3
(S)-naproxen methyl ester

| Conditions | Yield |
|---|---|
| In diethyl ether | 100% |
-
-
67-56-1
methanol

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
26159-35-3
(S)-naproxen methyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid | 100% |
| With sulfuric acid for 4h; | 100% |
| With thionyl chloride at 20℃; for 16h; Cooling with ice; Inert atmosphere; | 99% |
-
-
64-17-5
ethanol

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
31220-35-6
(S)-6-methoxy-α-methyl-2-naphthaleneacetic acid ethyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid | 100% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane 1.) 0 deg C, 1 h, 2.) r.t., 6 h; | 91% |
| With sulfuric acid at 50℃; for 18h; | 84% |
-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
84236-26-0
S-(+)-2-(5-Bromo-6-Methoxy-2-Naphthyl)-Propionic Acid

| Conditions | Yield |
|---|---|
| With bromine In dichloromethane at 20℃; for 1h; | 100% |
| With aluminum tri-bromide; [bis(acetoxy)iodo]benzene In acetonitrile at 23℃; | 93% |
| Multi-step reaction with 4 steps 1: 53 percent / oxalyl chloride / toluene / 4 h / 90 °C 2: 85 percent / triethylamine / CH2Cl2 / 0.25 h / -10 °C 3: 85 percent / Br2 / 1,2-dichloro-ethane / 0.75 h / 0 °C 4: 95 percent / CH3COOH, concd. HCl, water / 85 °C View Scheme | |
| With sodium hypochlorite; potassium bromide In methanol; water |
-
-
5292-43-3
bromoacetic acid tert-butyl ester

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
646509-90-2
((tert-butyl)oxycarbonyl)methyl (2S)-2-(6-methoxy(2-naphthyl))propanoate

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In N,N-dimethyl-formamide at 20℃; for 48h; | 100% |
| With sodium hydrogencarbonate In N,N-dimethyl-formamide at 20℃; for 48h; |
-
-
19982-08-2
memantine*

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
1185878-08-3
memantine-(S)-naproxen salt

| Conditions | Yield |
|---|---|
| In methanol at 20℃; | 100% |
-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
1112063-73-6
(1S,2S)-2-((dimethylamino)methyl)-1-(3-methoxyphenyl)cyclohexanol (S)-naproxen salt

| Conditions | Yield |
|---|---|
| In methanol at 20℃; | 100% |
-
-
67-56-1
methanol

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
30012-51-2
methyl 2-(6-methoxy-2-naphthyl)propionate

| Conditions | Yield |
|---|---|
| With sulfuric acid for 6h; Reflux; | 100% |
| With sulfuric acid for 18h; Reflux; | 98% |
| Stage #1: (2S)-2-(6-methoxy(2-naphthyl))propanoic acid With sulfuryl dichloride In dichloromethane at 50℃; for 1h; Stage #2: methanol With triethylamine In dichloromethane at 20℃; | 93% |
-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
5680-79-5
glycine ethyl ester hydrochloride

-
-
847454-70-0
N-[2-(S)-(6-methoxy-2-naphthyl)propanoyl]glycine methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: (2S)-2-(6-methoxy(2-naphthyl))propanoic acid With 1,1'-carbonyldiimidazole In chloroform at 20℃; for 1h; Inert atmosphere; Stage #2: glycine ethyl ester hydrochloride In chloroform Inert atmosphere; | 100% |
| With 4-pyrrolidin-1-ylpyridine; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; Inert atmosphere; | 60% |
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In acetonitrile for 3h; |
-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
505-86-2
cetyltrimethylammonium hydroxide

| Conditions | Yield |
|---|---|
| In propan-1-ol | 100% |
-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
26159-35-3
(S)-naproxen methyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid In methanol at 80℃; for 8h; stereoselective reaction; | 100% |
| With sulfuric acid In methanol at 20℃; for 12h; Inert atmosphere; | 88% |

| Conditions | Yield |
|---|---|
| In acetone | 99.9% |
-
-
112-43-6
10-Undecen-1-ol

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
142634-66-0
(S)-naproxen undec-10-en-1-yl ester

| Conditions | Yield |
|---|---|
| toluene-4-sulfonic acid In benzene Heating; | 99% |
-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
26159-36-4
naproxol

| Conditions | Yield |
|---|---|
| Stage #1: (2S)-2-(6-methoxy(2-naphthyl))propanoic acid With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 0.5h; Stage #2: With sodium tetrahydroborate; water In tetrahydrofuran at 0℃; for 0.5h; | 99% |
| Stage #1: (2S)-2-(6-methoxy(2-naphthyl))propanoic acid With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; Inert atmosphere; Reflux; Stage #2: With rochelle salt In tetrahydrofuran at 0℃; Inert atmosphere; | 89% |
| With lithium aluminium tetrahydride In tetrahydrofuran at 20℃; for 16h; | 87% |
-
-
57561-39-4
methyl(2-hydroxyethyl)carbamic acid tert-butyl ester

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride; dmap In dichloromethane at 0 - 20℃; for 24h; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: (2S)-2-(6-methoxy(2-naphthyl))propanoic acid With dicyclohexyl-carbodiimide In dichloromethane at 0℃; for 0.5h; Inert atmosphere; Stage #2: cholesterol With dmap In dichloromethane at 0℃; for 8h; | 99% |
-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
2684-60-8
2-diazopropane

| Conditions | Yield |
|---|---|
| In dichloromethane at 19℃; for 2h; chemoselective reaction; | 99% |

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| With copper diacetate; sodium carbonate; (3aR,3a'R,8aS,8a'S)-2,2'-(propane-2,2-diyl)bis(8,8a-dihydro-3aH-indeno[1,2-d]oxazole) In dichloromethane; water at 30℃; for 12h; enantioselective reaction; | 99% |
-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: L-valine isopropyl ester hydrochloride With ammonium hydroxide In water Stage #2: (2S)-2-(6-methoxy(2-naphthyl))propanoic acid In chloroform at 20℃; for 0.333333h; | 98.5% |

-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

| Conditions | Yield |
|---|---|
| In methanol | 98.3% |
-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
89617-86-7
(S)-2-(1-chloro-2-methoxynaphthalen-6-yl)propanoic acid

| Conditions | Yield |
|---|---|
| With N-chloro-succinimide; dimethyl sulfoxide In chloroform at 25℃; for 12h; Reagent/catalyst; Schlenk technique; | 98% |
| With sulfuryl dichloride at 20℃; for 1h; | 89% |
| Multi-step reaction with 3 steps 1: 100 percent / diethyl ether 2: 80 percent / sodium hypochlorite, aq. HCl / CH2Cl2 / 1 h / 15 °C 3: 37percent aq. Hcl / acetone / 3 h / Heating View Scheme | |
| With sodium hypochlorite In methanol; water |
-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid

-
-
154472-05-6
(2R)-(-)-2-phenyl-2-(piperidin-1-yl)ethanol

| Conditions | Yield |
|---|---|
| Stage #1: (2R)-(-)-2-phenyl-2-(piperidin-1-yl)ethanol With methanesulfonyl chloride; triethylamine In chloroform at 0 - 5℃; for 0.5h; Stage #2: (2S)-2-(6-methoxy(2-naphthyl))propanoic acid With triethylamine In chloroform at 5 - 20℃; for 16h; enantiospecific reaction; | 98% |
-
-
22204-53-1
(2S)-2-(6-methoxy(2-naphthyl))propanoic acid


-
-
646509-45-7
(2R)-7-(nitrooxy)-4,8-dioxabicyclo(3.3.0)oct-2-yl(2S)-2-(6-methoxy (2-naphthyl)) propanoate

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; | 97.6% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View








 Xn
Xn