Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
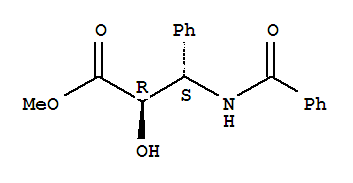
Cas:32981-85-4
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquirySimagchem Corporation
Welcome to Simagchem, your partner in China as a premier supply of bulk specialty chemicals for industry and life science. We introduce experienced quality product and exceptional JIT service with instant market intelligence in China to benefit our
Cas:32981-85-4
Min.Order:1 Kilogram
Negotiable
Type:Manufacturers
inquiryHangzhou Think Chemical Co. Ltd
Docetaxel Side Chain Methyl (2R,3S)-3-(benzoylamino)-2-hydroxy-3-phenylpropanoate CAS:32981-85-4 HANGZHOU THINK CHEMICAL CO., LTD. (THINKCHEM) is an integrative corporation of trade, research and contract manufacture. With about ten yea
Cas:32981-85-4
Min.Order:100 Gram
Negotiable
Type:Other
inquiryHebei yanxi chemical co.,LTD.
Our advantage: 1: High quality and Competitive price 2: Quality control, GC&HPLC&NMR or as your requested 3: Packing as your requested, and double check before shipment 4: Prompt shipment with professional documents 5: Excellent after-
Cas:32981-85-4
Min.Order:1 Kilogram
FOB Price: $1.0 / 3.0
Type:Trading Company
inquiryHenan Tianfu Chemical Co., Ltd.
Product Name: Methyl (2R,3S)-3-(benzoylamino)-2-hydroxy-3-phenylpropanoate Synonyms: PACLITAXEL SIDE CHAIN 4;PACLITAXEL SIDE CHAIN NO 4;C-13 SIDE CHAIN FOR PACLITAXEL II;(2R,3S)-1-Benzoyl-2-Hydroxy-3-Aminophenylpionic Acid Ester;METHYL (2R,3S)-3-B
Cas:32981-85-4
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1. All inquiries will be replied within 12 hours. 2. Dedication to quality, supply & service. 3. Strictly on selecting raw materials. 4. Reasonable & competitive price, fast lead time. 5. Sample is available for your eva
Cas:32981-85-4
Min.Order:1 Kilogram
FOB Price: $9.0 / 99.0
Type:Trading Company
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:32981-85-4
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryLIDE PHARMACEUTICALS LIMITED
Advantage : LIDE PHARMACEUTICALS LTD. is a mid-small manufacturing-type enterprise, engaged in pharmaceutical intermediates of R&D, custom-made and production, and also involving trading chemicals for export. We have established the R&
Cas:32981-85-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:32981-85-4
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
Methyl (2R,3S)-3-(benzoylamino)-2-hydroxy-3-phenylpropanoate CAS:32981-85-4 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, s
Cas:32981-85-4
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:32981-85-4
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Cas:32981-85-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryXiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Cas:32981-85-4
Min.Order:0
Negotiable
Type:Manufacturers
inquiryAfine Chemicals Limited
Company Introduction 1. Established in 2005, with two independent business divisions: Fine chemicals division; Pharmaceutical division. 2. Main product: Optical brightener Textile auxiliary Dye stuff Pigments
Cas:32981-85-4
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:32981-85-4
Min.Order:0 Metric Ton
Negotiable
Type:Other
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Cas:32981-85-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:32981-85-4
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquirySiwei Development Group Ltd.
Product name: Methyl (2R,3S)-N-Benzoylphenylisoserinate CAS No.:32981-85-4 Molecule Formula:C17H17NO4 Molecule Weight:299.32 Purity: 99.0% Package: 25kg/drum Description:White powder Manufacture Standards:Enterprise Standard TEST
Cas:32981-85-4
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Cas:32981-85-4
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Cas:32981-85-4
Min.Order:1 Milligram
Negotiable
Type:Trading Company
inquiryHubei CuiRan Biotechnology Co., Ltd
Hubei CuiRan Biotechnology Co., Ltd is a leading company in the research, development, manufacture and marketing of High Quality Phytochemicals and Extracts(especially Active Ingredients from Traditional Chinese Medicine,Traditional Chinese Medicine)
Cas:32981-85-4
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquiryHUNAN CHEMAPI BIOLOGICAL TECHNOLOGY CO.,LTD.
Hunan Kaimir Biotechnology Co., Ltd. is a professional technology company specializing in the research and production of raw materials and intermediates. In 2017, it was recognized as a high-tech enterprise and a small technological innovation giant
Cas:32981-85-4
Min.Order:1 Metric Ton
Negotiable
Type:Other
inquiryHangzhou Zhongqi chem Co.,Ltd.
Located in Hangzhou National Hi-Tech Industrial Development Zone, zhongqichem is a technical company mainly focus on the Custom synthesis, manufacturing, sales of chemicals to various industries. Benefiting from the outstanding customer service and h
Cas:32981-85-4
Min.Order:0
Negotiable
Type:Other
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
Cas:32981-85-4
Min.Order:0
Negotiable
Type:Manufacturers
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:white powder Storage:stored in a cool, dry and ventilated place to provent sun and rain Package:25kg/drum, or as per your request. Application:Used as Pharmaceutical Intermediate
Cas:32981-85-4
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Cas:32981-85-4
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryKono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Cas:32981-85-4
Min.Order:0
Negotiable
Type:Other
inquiryHangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air Port:Shanghai;Guangzhou
Cas:32981-85-4
Min.Order:0
Negotiable
Type:Manufacturers
inquiryJINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Cas:32981-85-4
Min.Order:100 Gram
Negotiable
Type:Lab/Research institutions
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Cas:32981-85-4
Min.Order:0
Negotiable
Type:Manufacturers
inquirySynthetic route
-
-
145041-37-8
methyl (2R,3S)-3-benzoylamino-2-benzyloxy-3-phenylpropanethioate

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen In methanol under 4654.46 Torr; | 100% |
| With hydrogen; 1% Pd/C In methanol under 4654.46 Torr; | 100% |
| With hydrogenchloride; hydrogen; palladium on activated charcoal In ethanol at 60℃; for 1h; | 78% |
| With hydrogen; palladium on activated charcoal In ethanol at 60℃; | 78% |
-
-
99458-15-8
Methyl (2R,3S)-(+)-2-hydroxy-3-azido-3-phenyl-propionate

-
-
98-88-4
benzoyl chloride

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| Stage #1: Methyl (2R,3S)-(+)-2-hydroxy-3-azido-3-phenyl-propionate; benzoyl chloride With dmap; triethylamine In ethyl acetate at 20℃; for 4h; Stage #2: With hydrogen; palladium on activated charcoal In methanol; ethyl acetate for 48h; Further stages.; | 92% |
| Stage #1: Methyl (2R,3S)-(+)-2-hydroxy-3-azido-3-phenyl-propionate; benzoyl chloride With dmap; triethylamine In methanol; ethyl acetate at 20℃; for 7h; Stage #2: With hydrogen; palladium on activated carbon In methanol; ethyl acetate for 48h; | 92% |
| Stage #1: Methyl (2R,3S)-(+)-2-hydroxy-3-azido-3-phenyl-propionate; benzoyl chloride; dmap In methanol; ethyl acetate at 20℃; for 7h; Stage #2: With hydrogen; palladium 10% on activated carbon In methanol; ethyl acetate for 48h; | 88% |
-
-
146848-86-4
methyl (2R,3S)-2-acetoxy-3-phenyl-3-benzoylaminopropanoate

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 20℃; for 1h; | 92% |
-
-
146848-91-1
(4S,5R)-2,4-diphenyl-2-oxazoline-5-carboxylic acid methyl ester

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol for 2h; Heating; | 90% |
| With hydrogenchloride In methanol for 5h; Heating; | 85% |
| With hydrogenchloride In methanol for 5h; Heating; | 85% |
-
-
186581-53-3, 908094-01-9
diazomethane

-
-
132201-33-3
(2R,3S)-N-benzoyl-3-phenylisoserine

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether | 90% |
| In methanol; diethyl ether |
-
-
99458-16-9
(2R,3S)-(+)-methyl 3-azido-2-benzoly-3-phenylpropionate

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In methanol | 88% |
| With hydrogen; toluene-4-sulfonic acid; palladium on activated charcoal | 76% |
| With hydrogen; palladium on activated charcoal In methanol under 2585.7 Torr; for 4h; | 74% |
| With hydrogen; palladium on activated charcoal In methanol at 20℃; under 760 Torr; for 48h; | 71% |
| With hydrogen; palladium on activated charcoal 1.) CH3OH, 15 h, 2.) CH3OH, RT, 72 h; Yield given. Multistep reaction; |
-
-
67-56-1
methanol

-
-
98-88-4
benzoyl chloride

-
-
195624-97-6
(2R,3S)-3-acetylamino-2-hydroxy-3-phenylpropanoic acid isopropyl ester

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| Stage #1: methanol; (2R,3S)-3-acetylamino-2-hydroxy-3-phenylpropanoic acid isopropyl ester With hydrogenchloride for 20h; Heating; Stage #2: benzoyl chloride With triethylamine In dichloromethane at 0 - 20℃; | 88% |

-
-
67-56-1
methanol

-
-
98-88-4
benzoyl chloride

-
-
132201-32-2
(2R,3S)-3-phenylisoserine hydrochloride

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| Stage #1: benzoyl chloride; (2R,3S)-3-phenylisoserine hydrochloride With sodium hydroxide In water at 20℃; for 1h; pH=9 - 12; Stage #2: methanol Concentration; | 85.6% |

-
-
67-56-1
methanol

-
-
100-44-7
benzyl chloride

-
-
195624-97-6
(2R,3S)-3-acetylamino-2-hydroxy-3-phenylpropanoic acid isopropyl ester

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| Stage #1: methanol; (2R,3S)-3-acetylamino-2-hydroxy-3-phenylpropanoic acid isopropyl ester With hydrogenchloride for 20h; Addition; Heating; Stage #2: benzyl chloride With triethylamine In dichloromethane at 0 - 20℃; for 2h; Substitution; Further stages.; | 85% |

-
-
144541-45-7
methyl (2S,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With thionyl chloride In dichloromethane for 5h; Inert atmosphere; Reflux; | 85% |
| Multi-step reaction with 2 steps 1.1: SOCl2 / CH2Cl2 / Heating 2.1: HCl / H2O; methanol / Heating 2.2: NaHCO3 / H2O; methanol / 50 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 90 percent / DEAD; PPh3 / benzene / 20 h / 20 °C 2: 90 percent / 1N aq. HCl / methanol / 2 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: 77 percent / Ph3P, diethylazodicarboxylate / tetrahydrofuran / Ambient temperature 2: 1.) aq. HCl, 2.) aq. NaHCO3 / 1) CH3OH, room temperature, 1.5 h; 2) room temperature, 16 h View Scheme | |
| Multi-step reaction with 2 steps 1: 1) SOCl2 / 1) CHCl3, 45 deg C, 5 h, 2) CHCl3, reflux, 48 h 2: 80 percent / 1N HCl / methanol / 2 h / Heating View Scheme |
-
-
67-56-1
methanol

-
-
222727-06-2
S-Ethyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanethioate

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With silver trifluoroacetate Ambient temperature; | 84% |
| With silver trifluoroacetate at 20℃; for 1h; Methylation; | 84% |
-
-
33069-62-4
taxol

-
A
-
32981-86-5
10-deacetylbaccatin III

-
B
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
C
-
71629-92-0
10-deacetyl-7-epi-baccatin III

| Conditions | Yield |
|---|---|
| With lithium iodide In methanol for 10h; Ambient temperature; | A 58% B 82% C 40% |
| With lithium iodide In methanol for 10h; Ambient temperature; | A 58% B 82% C 40% |

-
-
105454-04-4
7-epipaclitaxel

-
A
-
31077-81-3
7-epi-baccatin III

-
B
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
C
-
71629-92-0
10-deacetyl-7-epi-baccatin III

| Conditions | Yield |
|---|---|
| With lithium iodide In methanol for 14h; Ambient temperature; | A 56% B 78% C 17% |

-
-
148930-54-5
(2aR,4S,4aS,6R,9S,11S,12S,12bS)-6,12b-bis(acetyloxy)-9-({(2R,3S)-3-(benzoylamino)-3-phenyl-2-[(triethylsilyl)oxy]propanoyl}oxy)-4,11-dihydroxy-4a,8,13,13-tetramethyl-5-oxo-2a,3,4,4a,5,6,9, 10,11,12,12a,12bdodecahydro-1H-7,11-methanocyclodeca[3,4]benzo[1,2-b]oxet-12-yl benzoate

-
A
-
32981-86-5
10-deacetylbaccatin III

-
B
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
C
-
71629-92-0
10-deacetyl-7-epi-baccatin III

| Conditions | Yield |
|---|---|
| With lithium iodide In methanol Ambient temperature; 2 weeks; | A 22% B 71% C 34% |

-
-
67-56-1
methanol

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With caesium carbonate In tetrahydrofuran at 20℃; for 1.5h; | 51% |
-
-
67-56-1
methanol


-
A
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
B
-
135821-94-2
methyl (2S,3R)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| In methanol Title compound not separated from byproducts; | |
| With sodium methylate Title compound not separated from byproducts; |

-
-
67-56-1
methanol

-
A
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
B
-
503178-23-2
3-Benzoylamino-2-hydroxy-3-phenyl-propionic acid methyl ester

-
C
-
135821-94-2
methyl (2S,3R)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| In methanol Title compound not separated from byproducts; | |
| With sodium methylate Title compound not separated from byproducts; |

-
-
67-56-1
methanol

-
A
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
B
-
503178-23-2
3-Benzoylamino-2-hydroxy-3-phenyl-propionic acid methyl ester

-
C
-
135821-94-2
methyl (2S,3R)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| In methanol Title compound not separated from byproducts; | |
| With sodium methylate Title compound not separated from byproducts; |

-
-
67-56-1
methanol


-
A
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
B
-
503178-23-2
3-Benzoylamino-2-hydroxy-3-phenyl-propionic acid methyl ester

| Conditions | Yield |
|---|---|
| In methanol Title compound not separated from byproducts; | |
| With sodium methylate |

-
-
67-56-1
methanol


-
A
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
B
-
503178-23-2
3-Benzoylamino-2-hydroxy-3-phenyl-propionic acid methyl ester

-
C
-
135821-94-2
methyl (2S,3R)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With sodium methylate Title compound not separated from byproducts; |

-
-
124-41-4
sodium methylate

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| In methanol at 25℃; for 1h; | 79 mg |
-
-
98-88-4
benzoyl chloride

-
-
157240-36-3
(2R,3S)-3-phenylisoserine methyl ester

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| Yield given; | |
| With sodium hydrogencarbonate In ethyl acetate | |
| With sodium hydrogencarbonate In tetrahydrofuran; water at 0℃; for 2h; | 0.123 g |
| With triethylamine | |
| In dichloromethane at 20℃; for 5h; Time; Cooling with ice; | 267 g |
-
-
98-88-4
benzoyl chloride

-
-
147058-27-3
methyl (2R,3S)-2-hydroxy-3-phenyl-3-<(S)-1-methylbenzylamino>propanoate

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With sodium hydroxide; hydrogen; palladium on activated charcoal 1.) MeOH, room temp., 30 h, 2.) THF, water, 0 deg C, 30 min; Yield given. Multistep reaction; |

-
A
-
93-58-3
benzoic acid methyl ester

-
B
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol Ambient temperature; |


-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With ammonium hydroxide In methanol Ambient temperature; | |
| With alkaline solution |
-
-
144494-74-6
(R)-3-benzamido-3-phenylpropanoic acid methyl ester

-
A
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
B
-
144541-45-7
methyl (2S,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With (+)-(2S,8aS)-(camphorylsulfonyl)oxaziridine; lithium chloride; lithium diisopropyl amide 1) -42 deg C, 2) -100 to -78 deg C; Yield given. Multistep reaction. Yields of byproduct given; | |
| With MoO5*pyridine*HMPA; potassium hexamethylsilazane 1.) toluene, THF, -78 deg C to -25 deg C; 2.) toluene, THF, -60 deg C, 3 h; Yield given. Multistep reaction. Yields of byproduct given; |

-
A
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
B
-
151764-53-3
(2R,3R)-(-)-2-hydroxy-3-(benzoylamino)-3-phenylpropanoic acid methyl ester

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In methanol | |
| With hydrogen; palladium on activated charcoal In methanol Title compound not separated from byproducts; |
-
-
157722-43-5
(2R,3S)-3-Benzoylamino-3-phenyl-2-trimethylsilanyloxy-propionic acid methyl ester

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With potassium fluoride In water |
-
-
32981-85-4, 132074-01-2, 135821-94-2, 144541-45-7
syn-(+/-)-2-hydroxy-3-(benzoylamino)-3-phenylpropanoic acid methyl ester

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate
-
-
67-56-1
methanol

-
-
33069-62-4
taxol

-
A
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
B
-
27548-93-2
baccatin III

-
C
-
105454-04-4
7-epipaclitaxel

| Conditions | Yield |
|---|---|
| at 58 - 116℃; Thermodynamic data; apparent activation energy Ea, other solvents, temperatures; |

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
76513-69-4
(2-trimethylethylsilylethoxy)methyl chloride

-
-
99458-19-2
(2R,3S)-N-benzoyl-O-<<2-(trimethylsilyl)ethoxy>methyl>-3-phenylisoserine methyl ester

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane for 40h; | 96% |
-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
124-63-0
methanesulfonyl chloride

-
-
274681-89-9
(2R,3S)-3-benzoylamino-2-methanesulfonyloxy-3-phenylpropanoic acid methyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 0 - 20℃; | 93% |
| With triethylamine In tetrahydrofuran at 0 - 20℃; for 1.5h; Mesylation; | 86% |
-
-
2186-92-7
p-Anisaldehyde dimethyl acetal

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
157826-09-0
Methyl (2R,4S,5R)-3-benzoyl-2-(p-methoxyphenyl)-4-phenyl-1,3-oxazolidine-5-carboxylate

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In cyclohexane; toluene | 91% |
-
-
1515-81-7
1-methoxy-4-(methoxymethyl)benzene

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
157826-09-0
Methyl (2R,4S,5R)-3-benzoyl-2-(p-methoxyphenyl)-4-phenyl-1,3-oxazolidine-5-carboxylate

| Conditions | Yield |
|---|---|
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile Heating; | 90% |
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile at 60℃; | 69% |
-
-
116-11-0
2-Methoxypropene

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
202390-84-9
Methyl (4S,5R)-3-benzoyl-2,2-dimethyl-4-phenyl-1,3-oxazolidine-5-carboxylate

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In toluene at 85℃; | 89% |
| With pyridinium p-toluenesulfonate In toluene at 80℃; for 10h; Etherification; Cyclization; | 89% |
| With toluene-4-sulfonic acid at 80℃; |
-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
227962-26-7
(4S,5S)-2,4-diphenyl-2-oxazoline-5-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With dmap; trifluoromethylsulfonic anhydride In dichloromethane at -30℃; for 1h; | 85% |
| With dmap; trifluoromethylsulfonic anhydride In dichloromethane at -30℃; for 1h; Cyclization; Dehydration; | 84% |
| With dmap; sulfuryl dichloride In dichloromethane at 0℃; Cyclization; | 65% |
| Multi-step reaction with 2 steps 1: 93 percent / Et3N / tetrahydrofuran / 0 - 20 °C 2: DBU / CHCl3 / 2 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: 86 percent / Et3N / tetrahydrofuran / 1.5 h / 0 - 20 °C 2: DBU / CHCl3 / 2 h / Heating View Scheme |
-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
107-30-2
chloromethyl methyl ether

-
-
99458-17-0
(2R,3S)-N-benzoyl-O-(methoxymethyl)-3-phenylisoserine methyl ester

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane for 40h; | 82% |
-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
132201-33-3
(2R,3S)-N-benzoyl-3-phenylisoserine

| Conditions | Yield |
|---|---|
| With potassium carbonate In methanol at 23℃; for 12h; | 80% |
-
-
2186-92-7
p-Anisaldehyde dimethyl acetal

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
202390-85-0
Methyl (2S,4S,5R)-3-benzoyl-2-(p-methoxyphenyl)-4-phenyl-1,3-oxazolidine-5-carboxylate

| Conditions | Yield |
|---|---|
| With camphor-10-sulfonic acid In toluene | 79% |
-
-
2186-92-7
p-Anisaldehyde dimethyl acetal

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
A
-
202390-85-0
Methyl (2S,4S,5R)-3-benzoyl-2-(p-methoxyphenyl)-4-phenyl-1,3-oxazolidine-5-carboxylate

-
B
-
157826-09-0
Methyl (2R,4S,5R)-3-benzoyl-2-(p-methoxyphenyl)-4-phenyl-1,3-oxazolidine-5-carboxylate

| Conditions | Yield |
|---|---|
| With camphor-10-sulfonic acid In toluene at 130℃; Cycloaddition; | A 79% B 18% |

-
-
3587-60-8
Benzyloxymethyl chloride

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
99458-18-1
(2R,3S)-N-benzoyl-O-<(benzyloxy)methyl>-3-phenylisoserine methyl ester

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane for 40h; | 75% |
-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
-
150-78-7
1,4-dimethoxybezene

-
-
157826-09-0
Methyl (2R,4S,5R)-3-benzoyl-2-(p-methoxyphenyl)-4-phenyl-1,3-oxazolidine-5-carboxylate

| Conditions | Yield |
|---|---|
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile at 70℃; for 0.116667h; Substitution; Cyclization; | 75% |
-
-
2186-92-7
p-Anisaldehyde dimethyl acetal

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| Stage #1: p-Anisaldehyde dimethyl acetal; methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate With pyridinium p-toluenesulfonate In toluene for 6h; Inert atmosphere; Reflux; Stage #2: With methanol; sodium hydroxide In tetrahydrofuran at 0 - 25℃; for 3h; | 75% |
-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

-
A
-
146848-91-1
(4S,5R)-2,4-diphenyl-2-oxazoline-5-carboxylic acid methyl ester

-
B
-
235432-33-4
3-N-benzoyl-4-phenyl-(4S,5R)-2-oxo-1,2,3-oxathiazolidine methyl ester

-
C
-
235432-34-5
3-N-benzoyl-4-phenyl-(4S,5R)-2-oxo-1,2,3-oxathiazolidine methyl ester

| Conditions | Yield |
|---|---|
| With thionyl chloride; triethylamine In benzene at 0 - 5℃; for 0.25h; Cyclization; | A 3% B 68% C 14% |

-
-
91352-76-0
2,4-dimethoxybenzaldehyde dimethylacetal

-
-
32981-85-4
methyl (2R,3S)-3-benzoylamino-2-hydroxy-3-phenylpropanoate

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In tetrahydrofuran at 100℃; for 1.5h; Condensation; Title compound not separated from byproducts; | A 64% B n/a |

Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View