Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
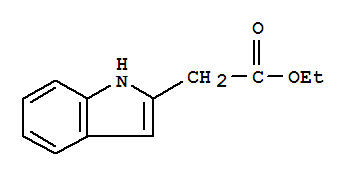
Cas:33588-64-6
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryWuhan Fortuna Chemical Co.,Ltd
Manufacturer supply Ethyl indole-2-acetate CAS 33588-64-6 with attractive price Company profile Wuhan Fortuna Chemical Co.,Ltd established in 2006, is a big integrative chemical enterprise being engaged in Pharmaceutical & its intermediates,
Cas:33588-64-6
Min.Order:10 Kilogram
FOB Price: $10.0
Type:Trading Company
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Cas:33588-64-6
Min.Order:1
Negotiable
Type:Other
inquiryHenan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 5 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so
Cas:33588-64-6
Min.Order:1 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryZhuozhou Wenxi import and Export Co., Ltd
WITH US,YOUR MONEY IN SAFE,YOUR BUSINESS IN SAFE 1)Quick Response Within 12 hours; 2)Quality Guarantee: All products are strictly tested by our QC, confirmed by QA and approved by third party lab in China, USA, Canada, Germany, UK, Italy, France et
Cas:33588-64-6
Min.Order:1 Kilogram
FOB Price: $139.0 / 210.0
Type:Trading Company
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:33588-64-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
ETHYL INDOLE-2-ACETATE CAS:33588-64-6 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic i
Cas:33588-64-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:33588-64-6
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Xiamen Hisunny Chemical Co.,Ltd
Best quality & Attractive price & Professional service; Trial & Pilot & Commercial Hisunny Chemical is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality intermediates, specia
Baoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present
Cas:33588-64-6
Min.Order:1 Gram
FOB Price: $879.0
Type:Trading Company
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Zibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:33588-64-6
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryXiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
Shanghai Minstar Chemical Co., Ltd
Product Name: ETHYL INDOLE-2-ACETATE Synonyms: ETHYL INDOLE-2-ACETATE;ETHYL 1H-INDOL-2-YLACETATE;Ethyl2-indoleacetate;2-(1-ethyl-1H-indol-2-yl)acetate;Ethyl 2-(1H-indol-2-yl)acetate;Indol-2-ylacetic acid ethyl ester;Ethyl 2-(2-Indolyl)acetate;2
Cas:33588-64-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryHunan chemfish Pharmaceutical co.,Ltd
Hunan chemfish Pharmaceutical co.,Ltd.located in Lugu High-tech industral park ,Hunan province . with its own R&D center and more than 10000㎡manufacture plant . Chemfish owns 40 reactors from 1000L to 8000L. With complete auxiliary equipment as
Cas:33588-64-6
Min.Order:0 Metric Ton
Negotiable
Type:Manufacturers
inquiryHangzhou Huarong Pharm Co., Ltd.
We Huarong Pharm can provide Customized Synthesis & Process R&D & APIs and intermediates Production & Quality Research & Registration Application, especially our GMP validation service which complies with SFDA, FDA, WHO and EU EMPA. O
Suzhou Sibian Chemical Technology Co., Ltd
The process is mature and can be mass produced to 100 kg, with good quality and purity up to 99%.The product has a large stock and can be supplied stably. Package:bottle Application:Drug R&D Transportation:Sealed drying(25℃) Port:ShangHai
Cas:33588-64-6
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryGIHI CHEMICALS CO.,LIMITED
Lower price, sample is available,SDS test documents are available,large stock in warehouseAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:Fine chemical intermediates, used as the main raw material for the synthe
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air Port:Shanghai;Guangzhou
Henan Allgreen Chemical Co.,Ltd
high quality Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea
Aecochem Corp.
Our clients, like BASF,CHEMO,Brenntag,ASR,Evonik,Merck and etc.Appearance:COA Storage:in stock Application:MSDS/TDS
Antimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
HANGZHOU TIANYE CHEMICALS CO., LTD.
We product this chemical more than 10 years . We are very experience to export it to many countries, Our superior & stable quality , competitive price gain warm reception from our customers. Application:Chemical intermediate
Hangzhou Fandachem Co.,Ltd
Ethyl indole-2-acetate cas 33588-64-6Appearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:intermediate Transportation:by air, by sea, by express
Hangzhou Sartort Biopharma Co., Ltd
Best quality with low price Storage:ln stock Package:25kg/Barrel Application:Chemicals Transportation:Express/Sea/Air Port:Shanghai
Henan Kanbei Chemical Co.,LTD
factory?direct?saleAppearance:White powder Storage:Sealed and preserved Package:200/Kilograms Application:healing drugs Transportation:By sea Port:Shanghai/tianjin
Hunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Cas:33588-64-6
Min.Order:0
Negotiable
Type:Trading Company
inquirySynthetic route
-
-
66073-33-4
4-(2-nitrophenyl)-3-oxobutyric acid ethyl ester

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| With ammonium chloride; zinc In tetrahydrofuran for 2h; | 95% |
| With palladium 10% on activated carbon; hydrogen In ethanol at 25℃; under 760.051 Torr; for 20h; Catalytic behavior; Reagent/catalyst; Solvent; | 91% |
| With ammonium acetate; titanium(III) chloride In acetone for 0.116667h; | 75% |
-
-
126742-92-5
(E)-4-[2-(1,1,1,3,3,3-Hexamethyl-disilazan-2-yl)-phenyl]-but-2-enoic acid ethyl ester

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride In tetrahydrofuran at 25℃; for 0.166667h; | 88% |

-
-
64-17-5
ethanol

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Stage #1: 2,2-dimethyl-5-[2-(2-nitrophenyl)acetyl]-1,3-dioxane-4,6-dione; ethanol for 5h; Reflux; Inert atmosphere; Stage #2: With ammonium chloride; zinc In tetrahydrofuran at 60℃; for 12h; | 88% |
-
-
128942-81-4
(Z)-ethyl 3-(2-iodophenylamino)-but-2-enoate

-
A
-
53855-47-3
ethyl 2-methyl-1H-indole-3-carboxylate

-
B
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0); silver orthophosphate In dimethyl sulfoxide at 100℃; for 3.5h; Heck reaction; | A 79% B 17% |
-
-
101315-54-2
triphenyl((ethyl(2-carbamoyl)acetate)-2-benzyl)phosphonium bromide

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In toluene for 0.75h; Heating / reflux; | 78% |
| With potassium tert-butylate In hexane; ethyl acetate; toluene | 78% |
| With potassium tert-butylate In toluene for 0.75h; Reflux; | 78% |
| With potassium tert-butylate In toluene for 0.25h; Heating; | 72% |
| With potassium tert-butylate In toluene for 0.25h; Heating; | 70% |

| Conditions | Yield |
|---|---|
| With silver fluoride In ethanol at 20℃; for 6h; Irradiation; Inert atmosphere; | 65% |
| With norborn-2-ene; bis(benzonitrile)palladium(II) dichloride; sodium hydrogencarbonate In water; N,N-dimethyl-formamide at 70℃; for 18h; Sealed tube; Inert atmosphere; | 57% |
| With tris[2-phenylpyridinato-C2,N]iridium(III); sodium hydrogencarbonate In water; acetonitrile for 18h; Schlenk technique; Inert atmosphere; Irradiation; | 55% |

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In ethanol at 20℃; for 24h; | 62% |
-
-
120-72-9
indole

-
-
3278-34-0
ethyl 2-(ethoxythiocarbonylthio)acetate

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| With dilauryl peroxide In 1,2-dichloro-ethane for 12h; Heating; | 60% |
| With triethyl borane; iron(II) sulfate In tetrahydrofuran; ethanol; dichloromethane at 20℃; | 30% |
| With tris[2-phenylpyridinato-C2,N]iridium(III); N,N-dimethyl-formamide at 24℃; Minisci Aromatic Substitution; Inert atmosphere; UV-irradiation; regioselective reaction; | 23% |

| Conditions | Yield |
|---|---|
| With tris(bipyridine)ruthenium(II) dichloride hexahydrate In methanol; dichloromethane; water Sealed tube; Irradiation; Cooling with ice; regioselective reaction; | 53% |

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; sodium thiosulfate; methyloxirane In various solvent(s) at 40℃; Substitution; Photolysis; | 36% |
-
-
14369-81-4
ethyl 2,3-butadienoate

-
-
113326-75-3
C-(4-oxo-4H<1>benzopyran-3-yl)-N-phenylnitrone

-
A
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| In benzene for 22h; Heating; | A 5% B 35% |

-
-
14369-81-4
ethyl 2,3-butadienoate

-
-
113326-75-3
C-(4-oxo-4H<1>benzopyran-3-yl)-N-phenylnitrone

-
A
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
B
-
213273-03-1
4-oxo-2-(N-phenyl)amino-4H-chromene-3-carbaldehyde

| Conditions | Yield |
|---|---|
| In benzene for 22h; Mechanism; Reactivity; Time; Reflux; regioselective reaction; | A n/a B n/a C 35% |

-
-
14369-81-4
ethyl 2,3-butadienoate

-
-
213273-02-0
C17H13NO3

-
A
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
B
-
1168003-46-0
1-ethoxycarbonyl-3-(2'-hydroxy-5'-methyl-benzoyl)-benzo[b]indolizine

-
C
-
213273-05-3
6-Methyl-4-oxo-2-phenylamino-4H-chromene-3-carbaldehyde

| Conditions | Yield |
|---|---|
| In benzene for 21h; Mechanism; Reactivity; Time; Reflux; regioselective reaction; | A n/a B 32% C n/a |

-
-
14369-81-4
ethyl 2,3-butadienoate

-
-
213273-01-9
C16H10ClNO3

-
A
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
B
-
1168003-50-6
1-ethoxycarbonyl-3-(2'-hydroxy-5'-chloro-benzoyl)-benzo[b]indolizine

-
C
-
213273-04-2
2-anilino-6-chloro-3-formylchromone

| Conditions | Yield |
|---|---|
| In benzene for 20h; Mechanism; Reactivity; Time; Reflux; regioselective reaction; | A n/a B 29% C n/a |

-
-
108629-63-6
2-indol-2-yl-acetimidic acid ethyl ester; hydrochloride

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| With water |
-
-
120-72-9
indole

-
-
623-48-3
ethyl iodoacetae

-
A
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
B
-
778-82-5
ethyl 3-indoleacetate

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; iron(II) sulfate In dimethyl sulfoxide Product distribution; Ambient temperature; intramolecular selectivity, further with thiophene, pyrrole, N-methylpyrrole, furan, ICH2CN, BrCH(CO2Et)2; |

-
-
113373-69-6
ethyl-1,2,3,5-tetrahydro-4-oxo-2-phenyl-4H-1-benzazepine-3-carboxylate

-
-
201024-81-9
C,N-diphenylnitrone

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| With water In tetrahydrofuran at 20℃; for 24h; Yield given; |
-
-
5344-90-1
2-Aminobenzyl alcohol

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 88 percent / acetonitrile / 7 h / Heating 2: 71 percent / CH2Cl2 / 3 h 3: 70 percent / t-BuOK / toluene / 0.25 h / Heating View Scheme | |
| Multi-step reaction with 3 steps 1: hexane; acetonitrile 2: ethanol; hexane; dichloromethane 3: potassium tert-butylate / hexane; ethyl acetate; toluene View Scheme | |
| Multi-step reaction with 3 steps 1: acetonitrile / 3 h / Reflux 2: dichloromethane / 3 h / 20 °C 3: potassium tert-butylate / toluene / 0.75 h / Reflux View Scheme |
-
-
78133-84-3
(2-aminobenzyl)triphenylphosphonium bromide

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 71 percent / CH2Cl2 / 3 h 2: 70 percent / t-BuOK / toluene / 0.25 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: 60 percent / CH2Cl2 / 3 h 2: 72 percent / KOtBu / toluene / 0.25 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: ethanol; hexane; dichloromethane 2: potassium tert-butylate / hexane; ethyl acetate; toluene View Scheme | |
| Multi-step reaction with 2 steps 1: dichloromethane / 3 h / 20 °C 2: potassium tert-butylate / toluene / 0.75 h / Reflux View Scheme |
-
-
5344-90-1
2-Aminobenzyl alcohol

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 88 percent / acetonitrile / 6 h / Heating 2: 60 percent / CH2Cl2 / 3 h 3: 72 percent / KOtBu / toluene / 0.25 h / Heating View Scheme |
-
-
14369-81-4
ethyl 2,3-butadienoate

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 70 percent Spectr. / tetrahydrofuran / 48 h / 20 °C 2: water / tetrahydrofuran / 24 h / 20 °C View Scheme |
-
-
201024-81-9
C,N-diphenylnitrone

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 70 percent Spectr. / tetrahydrofuran / 48 h / 20 °C 2: water / tetrahydrofuran / 24 h / 20 °C View Scheme |
-
-
126742-78-7
1,1,1-Trimethyl-N-(2-methylphenyl)-N-(trimethylsilyl)silanamine

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 65 percent / NBS, AIBN / CCl4 / 7 h / Heating 3: 88 percent / Bu4NF / tetrahydrofuran / 0.17 h / 25 °C View Scheme |
-
-
126742-80-1
2-bromomethyl-N,N-bis-trimethylsilylaniline

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: 88 percent / Bu4NF / tetrahydrofuran / 0.17 h / 25 °C View Scheme |
-
-
95-53-4
o-toluidine

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 82 percent / BuLi / tetrahydrofuran; hexane / 3 h / 0 - 25 °C 2: 65 percent / NBS, AIBN / CCl4 / 7 h / Heating 4: 88 percent / Bu4NF / tetrahydrofuran / 0.17 h / 25 °C View Scheme |
-
-
22751-23-1
(2-nitrophenyl)acetyl chloride

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: i-Pr2NEt / CH2Cl2 / 1) 0 deg C, 1h, 2) RT, 1h 2: Heating 3: 75 percent / aq. ammonium acetate, aq. TiCl3 / acetone / 0.12 h View Scheme | |
| Multi-step reaction with 3 steps 1: N-ethyl-N,N-diisopropylamine / dichloromethane / 1.5 h / 0 - 25 °C / Inert atmosphere 2: 0.75 h / Reflux 3: palladium 10% on activated carbon; hydrogen / ethanol / 20 h / 25 °C / 760.05 Torr View Scheme | |
| Multi-step reaction with 3 steps 1: N-ethyl-N,N-diisopropylamine / dichloromethane / 1.5 h / 0 - 25 °C / Inert atmosphere 2: 0.75 h / Reflux 3: palladium 10% on activated carbon; hydrogen; acetic acid; methanesulfonic acid / 25 - 50 °C / 760.05 Torr / Flow reactor View Scheme | |
| Multi-step reaction with 2 steps 1.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 2 h / 0 - 20 °C 2.1: 5 h / Reflux; Inert atmosphere 2.2: 12 h / 60 °C View Scheme |
-
-
3740-52-1
2-(2-nitrophenyl)acetic acid

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: SOCl2 / 50 °C 2: i-Pr2NEt / CH2Cl2 / 1) 0 deg C, 1h, 2) RT, 1h 3: Heating 4: 75 percent / aq. ammonium acetate, aq. TiCl3 / acetone / 0.12 h View Scheme | |
| Multi-step reaction with 4 steps 1: oxalyl dichloride; N,N-dimethyl-formamide / dichloromethane / 1 h / 25 °C / Inert atmosphere 2: N-ethyl-N,N-diisopropylamine / dichloromethane / 1.5 h / 0 - 25 °C / Inert atmosphere 3: 0.75 h / Reflux 4: palladium 10% on activated carbon; hydrogen / ethanol / 20 h / 25 °C / 760.05 Torr View Scheme | |
| Multi-step reaction with 4 steps 1: oxalyl dichloride; N,N-dimethyl-formamide / dichloromethane / 1 h / 25 °C / Inert atmosphere 2: N-ethyl-N,N-diisopropylamine / dichloromethane / 1.5 h / 0 - 25 °C / Inert atmosphere 3: 0.75 h / Reflux 4: palladium 10% on activated carbon; hydrogen; acetic acid; methanesulfonic acid / 25 - 50 °C / 760.05 Torr / Flow reactor View Scheme | |
| Multi-step reaction with 3 steps 1.1: oxalyl dichloride; N,N-dimethyl-formamide / dichloromethane / 4 h / 20 °C / Inert atmosphere 2.1: N-ethyl-N,N-diisopropylamine / dichloromethane / 2 h / 0 - 20 °C 3.1: 5 h / Reflux; Inert atmosphere 3.2: 12 h / 60 °C View Scheme |
-
-
74965-87-0
2,2-dimethyl-5-(2-phenylacetyl)-1,3-dioxane-4,6-dione

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Heating 2: 75 percent / aq. ammonium acetate, aq. TiCl3 / acetone / 0.12 h View Scheme |
-
-
61417-33-2
ethyl 2-acetyl-3-oxo-4-(2-nitrophenyl)butanoate

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: ethanolic NH3 2: zinc; aqueous acetic acid View Scheme |
-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
172226-77-6
tert-butyl 2-((ethoxycarbonyl)methyl)-1H-indole-1-carboxylate

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane for 3h; Ambient temperature; | 100% |
| With dmap; triethylamine In dichloromethane at 0 - 20℃; for 16h; | 97% |
| With dmap; triethylamine In dichloromethane at 20℃; for 14h; | 93% |
| With dmap In dichloromethane at 20℃; for 2h; | 91% |
-
-
50-00-0
formaldehyd

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
124-40-3
dimethyl amine

-
-
252637-04-0
ethyl 3-[(dimethylamino)methyl]-1H-indole-2-acetate

| Conditions | Yield |
|---|---|
| Substitution; | 95% |
| With acetic acid In water at 20℃; for 24h; | 41% |

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
75-36-5
acetyl chloride

-
-
129410-13-5
ethyl 3-acetylindol-2-ylacetate

| Conditions | Yield |
|---|---|
| With tin(IV) chloride In dichloromethane for 60h; Ambient temperature; | 93% |

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In dichloromethane at 0℃; for 0.5h; | 93% |
-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
57197-26-9
5,6,6-trimethoxycyclohexa-2,4-dien-1-one

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In dichloromethane at 0℃; for 0.5h; | 92% |
-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
34619-03-9
tert-butyldicarbonate

-
-
172226-77-6
tert-butyl 2-((ethoxycarbonyl)methyl)-1H-indole-1-carboxylate

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 20℃; for 2h; | 91% |
| With dmap In hexane; dichloromethane; ethyl acetate | 91% |

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In toluene at 130℃; for 18h; | 90% |


| Conditions | Yield |
|---|---|
| Stage #1: ethyl 2-(1H-indol-2-yl)acetate; 4-methoxy-benzaldehyde; edaravone With copper(II) sulfate In toluene at 130℃; for 0.3h; Stage #2: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile at 20℃; for 4h; diastereoselective reaction; | 90% |

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
536-74-3
phenylacetylene

| Conditions | Yield |
|---|---|
| With bis[(trifluoromethanesulfonyl)imidate]-2-(dicyclohexyl(2’,6’-dimethoxybiphenyl))phosphine gold(I) In acetonitrile at 50℃; for 24h; | 89% |

| Conditions | Yield |
|---|---|
| Stage #1: ethyl 2-(1H-indol-2-yl)acetate; m-Chlorobenzaldehyde; edaravone With copper(II) sulfate In toluene at 130℃; for 0.3h; Stage #2: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile at 20℃; for 4h; diastereoselective reaction; | 87% |

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; aluminium(III) triflate In 1,4-dioxane at 80℃; for 0.5h; Green chemistry; | 86% |

| Conditions | Yield |
|---|---|
| Stage #1: ethyl 2-(1H-indol-2-yl)acetate; 4-chlorobenzaldehyde; edaravone With copper(II) sulfate In toluene at 130℃; for 0.3h; Stage #2: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile at 20℃; for 4h; diastereoselective reaction; | 85% |


| Conditions | Yield |
|---|---|
| Stage #1: ethyl 2-(1H-indol-2-yl)acetate; 4-fluorobenzaldehyde; edaravone With copper(II) sulfate In toluene at 130℃; for 0.3h; Stage #2: With 2,3-dicyano-5,6-dichloro-p-benzoquinone In acetonitrile at 20℃; for 4h; diastereoselective reaction; | 85% |

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
104-87-0
4-methyl-benzaldehyde

-
-
504-02-9
1,3-cylohexanedione

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In toluene at 110℃; for 3h; Diels-Alder Cycloaddition; Green chemistry; diastereoselective reaction; | 84% |

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
64132-03-2
ethyl 2-(2,3-dihydro-1H-indole-2-yl)acetate

| Conditions | Yield |
|---|---|
| With borane; trimethylamine In trifluoroacetic acid | 82% |
| With trimethylamine-borane; trifluoroacetic acid for 0.25h; | 82% |
| With sodium cyanoborohydride; acetic acid | 82% |
-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
53316-56-6
4-(4-methylbenzylidene)-5-methyl-2-phenyl-2,4-dihydro-3H-pyrazol-3-one

-
-
555-16-8
4-nitrobenzaldehdye

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In toluene at 130℃; for 0.3h; diastereoselective reaction; | 82% |

-
-
827-46-3
2-phenyl-thiazol-4-one

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
555-16-8
4-nitrobenzaldehdye

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In toluene at 130℃; for 0.3h; diastereoselective reaction; | 82% |

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
504-02-9
1,3-cylohexanedione

-
-
123-11-5
4-methoxy-benzaldehyde

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In toluene at 110℃; for 3h; Diels-Alder Cycloaddition; Green chemistry; diastereoselective reaction; | 82% |

-
-
769-42-6
1,3-dimethylbarbituric acid

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
104-87-0
4-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In toluene at 110℃; for 3h; Diels-Alder Cycloaddition; Green chemistry; diastereoselective reaction; | 82% |

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
25105-60-6
2,3,6,7-tetrahydro-3-isopropyl-2-(4-nitrophenyl)cyclo-penta[e][1,3]oxazin-4(5H)-one

| Conditions | Yield |
|---|---|
| With acetic acid In toluene at 4℃; for 72h; | 81% |
-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; aluminium(III) triflate In 1,4-dioxane at 80℃; for 0.5h; Green chemistry; | 81% |
-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
80234-04-4
2-phenylacrylaldehyde diethyl acetal

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; aluminium(III) triflate In 1,4-dioxane at 80℃; for 0.5h; Green chemistry; | 80% |
-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
104-88-1
4-chlorobenzaldehyde

-
-
504-02-9
1,3-cylohexanedione

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In toluene at 110℃; for 3h; Diels-Alder Cycloaddition; Green chemistry; diastereoselective reaction; | 80% |

-
-
769-42-6
1,3-dimethylbarbituric acid

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
104-88-1
4-chlorobenzaldehyde

| Conditions | Yield |
|---|---|
| With copper(II) sulfate In toluene at 110℃; for 3h; Diels-Alder Cycloaddition; Green chemistry; diastereoselective reaction; | 80% |

-
-
769-42-6
1,3-dimethylbarbituric acid

-
-
33588-64-6
ethyl 2-(1H-indol-2-yl)acetate

-
-
620-23-5
m-tolyl aldehyde


| Conditions | Yield |
|---|---|
| With copper(II) sulfate In toluene at 110℃; for 3h; Diels-Alder Cycloaddition; Green chemistry; diastereoselective reaction; | A 77% B n/a |

Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View