Sinoway Industrial Co., Ltd.
Why is SINOWAY: 1) Specialized in pharmaceutical and healthcare industrial for 34 years. 2) ISO 9001:2015 & SGS audited supplier . 3) Accept various payment terms : T.T 30-60 days. 4) We have warehouse in USA with quickly shipment . 5) We c
Cas:84449-90-1
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryKono Chem Co.,Ltd
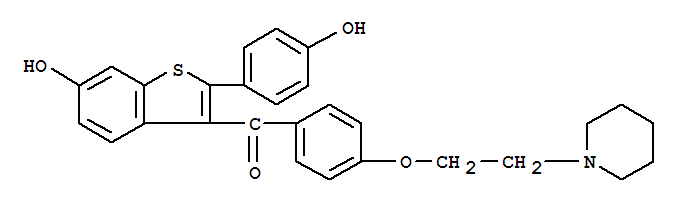
Specifications CAS No.: 84449-90-1 Other Names: Raloxifene hydrochloride MF: C28H27NO4S EINECS No.:
Wuhan Fortuna Chemical Co.,Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; ...... Appearance:Light yellow powder Storage:Desiccate at +4°C Package:1kg/foi
Cas:84449-90-1
Min.Order:500 Gram
FOB Price: $10.0
Type:Trading Company
inquiryXi'an Quanao Biotech Co., Ltd.
Xi’an Quanao Biotech Co., Ltd. with 18 years experience in plant extract filed. Product grade: Medicine, Health care product, Cosmetics, Food, Beverage, Feed. Hot selling market: Europe, North America, South America, Middle East, Asia Pacifi
Cas:84449-90-1
Min.Order:1 Kilogram
FOB Price: $259.0 / 269.0
Type:Manufacturers
inquiryXi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
Cas:84449-90-1
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryShanghai Seasonsgreen Chemical Co.,Ltd
Shanghai Seasonsgreen Chemical is a high-tech research and development, production, sale and custom synthesis set in one high-tech chemical products enterprises. Our sales and marketing division is located in Shanghai, serving international pharmaceu
Cas:84449-90-1
Min.Order:1 Kilogram
FOB Price: $1.0
Type:Manufacturers
inquiryChemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Ality Chemical Corporation
Raloxifene Basic information Application in Particular Diseases Product Name: Raloxifene Synonyms: [6-Hydroxy-2-(4-hydroxyphenyl)benzo[b]thien-3-y1][4-[2-(1-piperidinyl)ethoxy]phenyl]methamone;Evista;[4-[2-(1-Piperidinyl) Ethoxy] Phenyl]methano
Hebei Nengqian Chemical Import and Export Co., LTD
Our advantages: 1, High quality with competitive price: 1) Standard:BP/USP/EP/Enterprise standard 2) All Purity≥99% 3) We are manufacturer and can provide high quality products with factory price. 2, Fast and safe delivery 1) Parcel can be sent
Henan Sinotech Import&Export Corporation
Name: Raloxifene CAS No.: 84449-90-1 Formula: C28H27NO4S Molecular Weight: 473.58 Synonyms: Keoxifene;LY 139481;[2-(4-Hydroxyphenyl)-6-hydroxybenzo[b]thien-3-yl][4-(2-(1-piperidinyl)ethoxy)phenyl]methanone; Density: 1.289 g/cm3 Solubility: DM
Hubei Langyou International Trading Co., Ltd
1. Guaranteed purity; 2. Large quantity in stock; 3. Largest manufacturer; 4. Best service after shipment with email; 5. High quality & competitive price; Appearance: yellow powder Storage:Store in sealed containers at cool &
Cas:84449-90-1
Min.Order:100 Gram
Negotiable
Type:Other
inquiryShanghai Upbio Tech Co.,Ltd
1.No Less 8 years exporting experience. Clients can 100% received goods 2.Lower Price with higher quality 3,Free sample 4,We are sincerely responsible for the "product quality" and "After Service" Upbio is Specialized
Cas:84449-90-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryQingdao Beluga Import and Export Co., LTD
Raloxifene CAS:84449-90-1 Qingdao Belugas Import and Export Co., Ltd. is a scientific and technological company integrating research and development, production and trade of chemical intermediates, specializing in high quality organic intermediates,
Cas:84449-90-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryShandong Hanjiang Chemical Co., Ltd.
Hello, dear friend! I'm Hansen and Allen from China. Welcome to my lookchem mall! The following is a brief introduction of our company's products and services. If you are interested in our products, please contact us by emai
Cas:84449-90-1
Min.Order:1 Kilogram
Negotiable
Type:Lab/Research institutions
inquiryTriumph International Development Limilted
Triumph has the complete production of G- KG - MT service chain,we can make the new technology into productivity quickly in the research and development of new products. Main Business Custom Synthesis: Trading(Raw M
Cas:84449-90-1
Min.Order:0 Metric Ton
FOB Price: $18.0 / 20.0
Type:Lab/Research institutions
inquiryShanghai Terui OP New Material Technology Co., Ltd.
We have overseas warehouses in California, Nuevo Laredo, Mexico, Vancouver, Canada, Amsterdam, The Netherlands and Melbourne, Australia. Overseas warehouses can provide some of the best selling products. We look forward to cooperating with strong loc
Cas:84449-90-1
Min.Order:1 Kilogram
Negotiable
Type:Trading Company
inquiryWuhan Han Sheng New Material Technology Co.,Ltd
Our Advantage: high quality with competitive price High quality standard: BP/USP/EP Enterprise standard All purity customized Fast and safe delivery We have reliable forwarder who can help us deliver our goods more fast and safe. We
Cas:84449-90-1
Min.Order:10 Kilogram
Negotiable
Type:Trading Company
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Shaanxi New nine Material Technology Co., Ltd.
Our Advantage & Service 1.Top quality: Using high quality material and establishing a strict quality control system,assigning specific persons in charge of each part of production,from raw material purchase to assembly. 2 Professional R&D
Cas:84449-90-1
Min.Order:10 Gram
FOB Price: $8.0 / 10.0
Type:Trading Company
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:84449-90-1
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality, moderate price & quick delivery. Appearance:white powder Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:10g/bag,or as your request Application:Used as Pharmaceutical Intermediates Transporta
Cas:84449-90-1
Min.Order:1 Gram
Negotiable
Type:Trading Company
inquiryTaiChem Taizhou Limited
Established in May 2015, TaiChem Ltd. is initially invested by a British research and development company and started by PhDs back from aboard. The company is registered in China Medical City (CMC), Taizhou, Jiangsu Province, and the production site
Henan Tianfu Chemical Co., Ltd.
Our company was built in 2009 with an ISO certificate.In the past 6 years, we have grown up as a famous fine chemicals supplier in China and we had established stable business relationships with Samsung,LG,Merck,Thermo Fisher Scientific and so on.O
Cas:84449-90-1
Min.Order:1 Kilogram
FOB Price: $1000.0
Type:Lab/Research institutions
inquiryZibo Dorne chemical technology co. LTD
Product Details Grade: pharmaceutical grade Purity:99%+ ProductionCapacity: 1000 Kilogram/Month Scope of use: For scientific research only(The product must be used legally) Our Advantage 1. Best quality with competitive price. 2. Quick shipping,
Jiangsu Qianyu Molecular Technology Co., LTD.
Our Advantages A. International Top level TechnologyOur company owned biomedicine experts are famous at home and abroad with rich experience in research and development in the field of efficient chiral functional molecules research and development an
Xiamen Jenny Chemical Technology Co., Ltd.
GMP standard, high purity, competitive price, in stock 1. Quick Response: within 6 hours after receiving your email. 2. Quality Guarantee: All products are strictly tested by our QC, confirmed by QA, and approved by a third-party lab in China, USA,
EAST CHEMSOURCES LIMITED
factory?direct?saleAppearance:White Powder Storage:Store In Dry, Cool And Ventilated Place Package:25kg/drum, also according to the clients requirement Application:It is widely used as a thickener, emulsifier and stabilizer Transportation:By Sea Or B
Cas:84449-90-1
Min.Order:1 Kilogram
FOB Price: $18.0 / 20.0
Type:Trading Company
inquiryTAIZHOU ZHENYU BIOTECHNOLOGY CO., LTD
Zhenyu biotech exported this product to many countries and regions at best price. if you are looking for the material's manufacturer or supplier in china, zhenyu biotech is your best choice. pls contact with us freely for getting detailed
Cas:84449-90-1
Min.Order:1 Kilogram
FOB Price: $2.0
Type:Lab/Research institutions
inquiryShandong Mopai Biotechnology Co., LTD
Shandong Mopai Biotechnology Co., LTD is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemicals. W
Cas:84449-90-1
Min.Order:1 Gram
Negotiable
Type:Lab/Research institutions
inquiryKAISA GROUP INC
1.Applied in food field.it can improve the immune system and prolong life. 2.Appliedin cosmetic field.it can improve the skin care. 3.Applied in pharmaceutical field.it can treat various dieases. 4.Our product quality assurance will make our customer
Cas:84449-90-1
Min.Order:1 Metric Ton
FOB Price: $1.5
Type:Trading Company
inquirySynthetic route
-
-
82640-04-8
raloxifene hydrochloride

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Stage #1: raloxifene hydrochloride With sodium hydroxide In water; acetone at 20℃; Stage #2: With acetic acid In water; acetone at 16 - 20℃; for 1.75h; pH=8; | 100% |
| Stage #1: raloxifene hydrochloride With sodium hydroxide In water; acetone at 20℃; Stage #2: With acetic acid In water; acetone at 16 - 18℃; for 0.75h; pH=8; | 100% |
| With sodium hydroxide; water; isopropyl alcohol at 20℃; for 5h; pH=9; | |
| With sodium hydroxide In methanol; water at 15 - 30℃; for 12.5h; pH=8 - 8.5; | 180 g |
-
-
1293408-87-3
1-(2-(4-[6-Hydroxy-2-(4-hydroxy-phenyl)-benzo[b]thiophene-3-carbonyl]-phenoxy)-ethyl)-piperidinium bromide

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Stage #1: 1-(2-(4-[6-Hydroxy-2-(4-hydroxy-phenyl)-benzo[b]thiophene-3-carbonyl]-phenoxy)-ethyl)-piperidinium bromide With sodium hydroxide In water; acetone at 20℃; Stage #2: With acetic acid In water; acetone at 12℃; for 1h; pH=8 - 9; | 100% |
| Stage #1: 1-(2-(4-[6-Hydroxy-2-(4-hydroxy-phenyl)-benzo[b]thiophene-3-carbonyl]-phenoxy)-ethyl)-piperidinium bromide With sodium hydroxide In 1,4-dioxane; water at 16 - 20℃; Stage #2: With acetic acid In 1,4-dioxane; water for 0.166667h; pH=8; Product distribution / selectivity; | 85.8% |
-
-
84541-38-8
[6-Methoxy-2-(4-Methoxyphenyl)benzo[b]thien-3-yl]-[4-[2-(1-Piperidinyl)ethoxy]phenyl] Methanone

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In dichloromethane at 50℃; | 90% |
| With aluminium trichloride; ethanethiol demethylation; | 78% |
| In dichloromethane for 6h; Ionic liquid; Reflux; | 61% |
-
-
3040-44-6
1-piperidinoethanol

-
-
202336-25-2
[2-(4-hydroxyphenyl)-6-hydroxybenzo[b]thien-3-yl] [4-fluorophenyl]methanone

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 50℃; for 5h; | 86% |
-
-
84449-85-4
<6-<(methylsulfonyl)oxy>-2-<4-<(methylsulfonyl)oxy>phenyl>benzothien-3-yl><4-<2-(1-pyrrolidinyl)ethoxy>phenyl>methanone hydrochloride

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Stage #1: <6-<(methylsulfonyl)oxy>-2-<4-<(methylsulfonyl)oxy>phenyl>benzothien-3-yl><4-<2-(1-pyrrolidinyl)ethoxy>phenyl>methanone hydrochloride With potassium hydroxide; water In dimethyl sulfoxide at 60 - 62℃; for 4h; Stage #2: With acetic acid In water; dimethyl sulfoxide at 20℃; Stage #3: With ammonia In water; dimethyl sulfoxide at 20℃; Product distribution / selectivity; | 70.5% |
| Stage #1: <6-<(methylsulfonyl)oxy>-2-<4-<(methylsulfonyl)oxy>phenyl>benzothien-3-yl><4-<2-(1-pyrrolidinyl)ethoxy>phenyl>methanone hydrochloride With potassium hydroxide; water In dimethyl sulfoxide at 60 - 62℃; for 4h; Stage #2: With hydrogenchloride In water; dimethyl sulfoxide at 20℃; for 1h; Stage #3: With ammonia In water; dimethyl sulfoxide at 20℃; Product distribution / selectivity; | 66.33% |
| With sodium hydroxide In ethanol for 1.5h; Heating; | 42% |

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| With potassium hydroxide In dimethyl sulfoxide Inert atmosphere; | 67% |
| Stage #1: 6-methanesulfonyloxy-2-(4-methanesulfonyloxyphenyl)-3-[4-(2-piperidinoethoxy)-benzoyl]benzo[b]thiophene With sodium hydroxide; ethanol; water for 1.5h; Stage #2: In water Acidic aqueous solution; Stage #3: With sodium hydrogencarbonate In water Product distribution / selectivity; | 42.2% |
| With sodium hydroxide In tetrahydrofuran; methanol; chloroform | |
| With sodium hydroxide In tetrahydrofuran; methanol; chloroform | |
| With sodium hydroxide In tetrahydrofuran; methanol; chloroform |
-
-
3040-44-6
1-piperidinoethanol

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 90 percent / NaH / dimethylformamide / 0.5 h / 20 °C 2: 78 percent / AlCl3; ethanethiol View Scheme | |
| Multi-step reaction with 3 steps 1: 97 percent / NaH / dimethylformamide / 0.25 h / 20 °C 2: 90 percent / tetrahydrofuran / 2 h / 5 - 23 °C 3: 78 percent / AlCl3; ethanethiol View Scheme |
-
-
403-43-0
4-fluorobenzoyl chloride

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 70 percent / chlorobenzene / 20 °C 2: 97 percent / NaH / dimethylformamide / 0.25 h / 20 °C 3: 90 percent / tetrahydrofuran / 2 h / 5 - 23 °C 4: 78 percent / AlCl3; ethanethiol View Scheme | |
| Multi-step reaction with 4 steps 1: 70 percent / chlorobenzene / 20 °C 2: 83 percent / tetrahydrofuran / 1 h / 5 - 23 °C 3: 90 percent / NaH / dimethylformamide / 0.5 h / 20 °C 4: 78 percent / AlCl3; ethanethiol View Scheme | |
| Multi-step reaction with 4 steps 1: 70 percent / chlorobenzene / 20 °C 2: 97 percent / dimethylformamide / 0.25 h / 20 °C 3: 90 percent / tetrahydrofuran / 2 h / 5 - 23 °C 4: 78 percent / AlCl3; ethanethiol View Scheme | |
| Multi-step reaction with 3 steps 1: 74 percent / AlCl3 / CH2Cl2 2: 81 percent / BBr3 / CH2Cl2 / 3 h / 5 °C 3: 86 percent / NaH / dimethylformamide / 5 h / 50 °C View Scheme |
-
-
111359-29-6
6-methoxy-N,N-dimethylbenzo[b]thiophen-2-amine

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 70 percent / chlorobenzene / 20 °C 2: 97 percent / NaH / dimethylformamide / 0.25 h / 20 °C 3: 90 percent / tetrahydrofuran / 2 h / 5 - 23 °C 4: 78 percent / AlCl3; ethanethiol View Scheme | |
| Multi-step reaction with 4 steps 1: 70 percent / chlorobenzene / 20 °C 2: 83 percent / tetrahydrofuran / 1 h / 5 - 23 °C 3: 90 percent / NaH / dimethylformamide / 0.5 h / 20 °C 4: 78 percent / AlCl3; ethanethiol View Scheme | |
| Multi-step reaction with 4 steps 1: 70 percent / chlorobenzene / 20 °C 2: 97 percent / dimethylformamide / 0.25 h / 20 °C 3: 90 percent / tetrahydrofuran / 2 h / 5 - 23 °C 4: 78 percent / AlCl3; ethanethiol View Scheme |
-
-
165742-76-7
[2-dimethylamino-6-methoxybenzothien-3-yl] [4-[2-(1-piperidinyl)ethoxy]phenyl]-methanone

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 90 percent / tetrahydrofuran / 2 h / 5 - 23 °C 2: 78 percent / AlCl3; ethanethiol View Scheme |
-
-
206434-77-7
6-methoxy-2-(4-methoxyphenyl)-3-(4-fluorobenzoyl)benzo[b]thiophene

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 90 percent / NaH / dimethylformamide / 0.5 h / 20 °C 2: 78 percent / AlCl3; ethanethiol View Scheme | |
| Multi-step reaction with 2 steps 1: 81 percent / BBr3 / CH2Cl2 / 3 h / 5 °C 2: 86 percent / NaH / dimethylformamide / 5 h / 50 °C View Scheme |
-
-
243845-88-7
(2-dimethylamino-6-methoxy-benzo[b]thiophen-3-yl)-(4-fluoro-phenyl)-methanone

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 97 percent / NaH / dimethylformamide / 0.25 h / 20 °C 2: 90 percent / tetrahydrofuran / 2 h / 5 - 23 °C 3: 78 percent / AlCl3; ethanethiol View Scheme | |
| Multi-step reaction with 3 steps 1: 83 percent / tetrahydrofuran / 1 h / 5 - 23 °C 2: 90 percent / NaH / dimethylformamide / 0.5 h / 20 °C 3: 78 percent / AlCl3; ethanethiol View Scheme | |
| Multi-step reaction with 3 steps 1: 97 percent / dimethylformamide / 0.25 h / 20 °C 2: 90 percent / tetrahydrofuran / 2 h / 5 - 23 °C 3: 78 percent / AlCl3; ethanethiol View Scheme |
-
-
63675-74-1
2-(4'-methoxyphenyl)-6-methoxybenzothiophene

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 74 percent / AlCl3 / CH2Cl2 2: 81 percent / BBr3 / CH2Cl2 / 3 h / 5 °C 3: 86 percent / NaH / dimethylformamide / 5 h / 50 °C View Scheme | |
| Multi-step reaction with 4 steps 1: 71 percent / pyridine hydrochloride / 6 h / 220 °C 2: 99 percent / 4-(dimethylamino)pyridine, pyridine / Ambient temperature 3: 93 percent / trifluoromethanesulfonic acid / 1,2-dichloro-ethane / Heating 4: 42 percent / 5 N aq. NaOH / ethanol / 1.5 h / Heating View Scheme |
-
-
15570-12-4
3-methoxybenzenethiol

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 80.4 percent / KOH / ethanol; H2O; ethyl acetate / Ambient temperature 2: 69 percent / PPA / 1 h / 85 - 90 °C 3: 71 percent / pyridine hydrochloride / 6 h / 220 °C 4: 99 percent / 4-(dimethylamino)pyridine, pyridine / Ambient temperature 5: 93 percent / trifluoromethanesulfonic acid / 1,2-dichloro-ethane / Heating 6: 42 percent / 5 N aq. NaOH / ethanol / 1.5 h / Heating View Scheme |
-
-
63676-22-2
6-hydroxy-2-(4-hydroxyphenyl)benzo[B]-thiophene

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 99 percent / 4-(dimethylamino)pyridine, pyridine / Ambient temperature 2: 93 percent / trifluoromethanesulfonic acid / 1,2-dichloro-ethane / Heating 3: 42 percent / 5 N aq. NaOH / ethanol / 1.5 h / Heating View Scheme |
-
-
166975-76-4
4-(2-(piperidin-1-yl)ethoxy)benzoyl chloride

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 93 percent / trifluoromethanesulfonic acid / 1,2-dichloro-ethane / Heating 2: 42 percent / 5 N aq. NaOH / ethanol / 1.5 h / Heating View Scheme |
-
-
63675-73-0
α-(3-methoxyphenyl-thio)-4-methoxyacetophenone

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 69 percent / PPA / 1 h / 85 - 90 °C 2: 71 percent / pyridine hydrochloride / 6 h / 220 °C 3: 99 percent / 4-(dimethylamino)pyridine, pyridine / Ambient temperature 4: 93 percent / trifluoromethanesulfonic acid / 1,2-dichloro-ethane / Heating 5: 42 percent / 5 N aq. NaOH / ethanol / 1.5 h / Heating View Scheme |
-
-
84449-65-0
6-<(methylsulfonyl)oxy>-2-<4-<(methylsulfonyl)oxy>phenyl>benzothiophene

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 93 percent / trifluoromethanesulfonic acid / 1,2-dichloro-ethane / Heating 2: 42 percent / 5 N aq. NaOH / ethanol / 1.5 h / Heating View Scheme | |
| Multi-step reaction with 3 steps 1: aluminum (III) chloride / 1,2-dichloro-ethane / 20.5 h / 20 °C / Inert atmosphere 2: sodium t-butanolate / acetonitrile / 16 h / 80 °C / Inert atmosphere 3: potassium hydroxide / dimethyl sulfoxide / Inert atmosphere View Scheme |
-
-
84449-80-9
4-(2-piperidin-1-ylethoxy)benzoic acid hydrochloride

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: SOCl2, DMF / toluene 2: 93 percent / trifluoromethanesulfonic acid / 1,2-dichloro-ethane / Heating 3: 42 percent / 5 N aq. NaOH / ethanol / 1.5 h / Heating View Scheme |
-
-
2632-13-5
2-Bromo-4'-methoxyacetophenone

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 80.4 percent / KOH / ethanol; H2O; ethyl acetate / Ambient temperature 2: 69 percent / PPA / 1 h / 85 - 90 °C 3: 71 percent / pyridine hydrochloride / 6 h / 220 °C 4: 99 percent / 4-(dimethylamino)pyridine, pyridine / Ambient temperature 5: 93 percent / trifluoromethanesulfonic acid / 1,2-dichloro-ethane / Heating 6: 42 percent / 5 N aq. NaOH / ethanol / 1.5 h / Heating View Scheme |
-
-
84449-82-1
6-acetoxy-2-(4-acetoxyphenyl)-3-[4-(2-piperidinoethoxy)benzoyl]benzo[b]thiophene, hydrochloride

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; chloroform; water | |
| With sodium hydroxide In methanol; chloroform; water |
-
-
84449-83-2
6-benzoyloxy-2-(4-benzoyloxyphenyl)-3-[4-(2-piperidinoethoxy)benzoyl]benzo[b]thiophene, hydrochloride

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| With methanesulfonic acid In ethanol; water |

| Conditions | Yield |
|---|---|
| Stage #1: 6-methanesulfonyloxy-2-(4-methanesulfonyloxyphenyl)-3-[4-(2-piperidinoethoxy)-benzoyl]benzo[b]thiophene With sodium hydroxide; water In N,N-dimethyl-formamide at 80 - 90℃; for 3h; Stage #2: With hydrogenchloride In water; N,N-dimethyl-formamide at 20℃; for 1h; Stage #3: With ammonia In water; N,N-dimethyl-formamide Product distribution / selectivity; | |
| Stage #1: 6-methanesulfonyloxy-2-(4-methanesulfonyloxyphenyl)-3-[4-(2-piperidinoethoxy)-benzoyl]benzo[b]thiophene With sodium hydroxide; water In tetrahydrofuran at 60 - 62℃; for 14h; Stage #2: With acetic acid In tetrahydrofuran; water at 20℃; for 0.333333h; Stage #3: With ammonia In tetrahydrofuran; water Product distribution / selectivity; |

-
-
1390631-69-2
C24H20O10S4

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium t-butanolate / acetonitrile / 16 h / 80 °C / Inert atmosphere 2: potassium hydroxide / dimethyl sulfoxide / Inert atmosphere View Scheme |
-
-
89938-63-6
methanesulfonic acid 4-chlorocarbonyl-phenyl ester

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: aluminum (III) chloride / 1,2-dichloro-ethane / 20.5 h / 20 °C / Inert atmosphere 2: sodium t-butanolate / acetonitrile / 16 h / 80 °C / Inert atmosphere 3: potassium hydroxide / dimethyl sulfoxide / Inert atmosphere View Scheme |
-
-
99-76-3
methyl 4-hydroxylbenzoate

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: potassium carbonate / acetone / Reflux 2: hydrogenchloride; water / tetrahydrofuran 3: thionyl chloride / dichloromethane 4: aluminum (III) chloride / dichloromethane / 12 h / 50 °C / Inert atmosphere 5: neat liquid / 12 h / Inert atmosphere; Reflux 6: aluminum (III) chloride / dichloromethane / 50 °C View Scheme |
-
-
38567-00-9
4-(2-Chloroethoxy)benzoic acid methyl ester

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: hydrogenchloride; water / tetrahydrofuran 2: thionyl chloride / dichloromethane 3: aluminum (III) chloride / dichloromethane / 12 h / 50 °C / Inert atmosphere 4: neat liquid / 12 h / Inert atmosphere; Reflux 5: aluminum (III) chloride / dichloromethane / 50 °C View Scheme |
-
-
65136-51-8
4-(2-chloroethoxy)benzoic acid

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: thionyl chloride / dichloromethane 2: aluminum (III) chloride / dichloromethane / 12 h / 50 °C / Inert atmosphere 3: neat liquid / 12 h / Inert atmosphere; Reflux 4: aluminum (III) chloride / dichloromethane / 50 °C View Scheme |
-
-
65136-50-7
4-(2-chloro-ethoxy)-benzoyl chloride

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: aluminum (III) chloride / dichloromethane / 12 h / 50 °C / Inert atmosphere 2: neat liquid / 12 h / Inert atmosphere; Reflux 3: aluminum (III) chloride / dichloromethane / 50 °C View Scheme |
-
-
84449-90-1
Raloxifen

-
-
1326716-58-8
[6-hydroxy-2-(4-hydroxyphenyl)-benzothiophene-3-yl]-[4-[2-(1-piperidyl)ethoxy]phenyl]methanone sulfamate

| Conditions | Yield |
|---|---|
| With aminosulfonic acid In water; acetonitrile at 80℃; Product distribution / selectivity; | 96.4% |
-
-
84449-90-1
Raloxifen

-
-
174731-13-6
[2-(4-Hydroxyphenyl)-6-hydroxybenzo[b]thien-3-yl][4-[2-(1-piperidinyl)ethoxy]phenyl]methanol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 0℃; for 6h; | 91% |
-
-
84449-90-1
Raloxifen

-
-
1293408-87-3
1-(2-(4-[6-Hydroxy-2-(4-hydroxy-phenyl)-benzo[b]thiophene-3-carbonyl]-phenoxy)-ethyl)-piperidinium bromide

| Conditions | Yield |
|---|---|
| With hydrogen bromide In methanol; water at 1 - 35℃; pH=1.5; Reflux; | 87.4% |
| With hydrogen bromide In methanol; water at 35℃; pH=1.5; Reflux; | 87.4% |
-
-
358-23-6
trifluoromethylsulfonic anhydride

-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| With triethylamine In 1,2-dichloro-ethane | 51% |
| With pyridine In dichloromethane at 0 - 20℃; for 1h; | 0.16 g |
-
-
100-44-7
benzyl chloride

-
-
84449-90-1
Raloxifen

-
A
-
189940-64-5
[6-Benzyloxy-2-(4-hydroxy-phenyl)-benzo[b]thiophen-3-yl]-[4-(2-piperidin-1-yl-ethoxy)-phenyl]-methanone


| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide | A 20% B 20% C 20% |

-
-
84449-90-1
Raloxifen

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
A
-
174264-47-2
(6-((tert-butyldimethylsilyl)oxy)-2-(4-hydroxyphenyl)benzo[b]thiophen-3-yl)(4-(2-(piperidin-1-yl)ethoxy)phenyl)methanone

-
B
-
174264-46-1
2-[4-[[(1,1-dimethylethyl)dimethylsilyl]oxy]phenyl]-6-hydroxybenzo[b]thiophen-3-yl 4-[2-(1-piperidinyl)ethoxy]phenyl ketone

| Conditions | Yield |
|---|---|
| With dmap In tetrahydrofuran; N,N-dimethyl-formamide | A 20% B 20% C 20% |

-
-
98-88-4
benzoyl chloride

-
-
84449-90-1
Raloxifen

-
-
1248544-66-2
4-(6-hydroxy-3-(4-(2-(piperidin-1-yl)ethoxy)phenylcarbonyl)benzo[b]thiophen-2-yl)phenyl benzoate

| Conditions | Yield |
|---|---|
| Stage #1: Raloxifen With sodium hydride In N,N-dimethyl-formamide; mineral oil at 20℃; for 0.0833333h; Stage #2: benzoyl chloride In N,N-dimethyl-formamide at 20℃; for 0.25h; | 7.8% |
-
-
84449-90-1
Raloxifen

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
A
-
174264-47-2
(6-((tert-butyldimethylsilyl)oxy)-2-(4-hydroxyphenyl)benzo[b]thiophen-3-yl)(4-(2-(piperidin-1-yl)ethoxy)phenyl)methanone

-
B
-
174264-46-1
2-[4-[[(1,1-dimethylethyl)dimethylsilyl]oxy]phenyl]-6-hydroxybenzo[b]thiophen-3-yl 4-[2-(1-piperidinyl)ethoxy]phenyl ketone

| Conditions | Yield |
|---|---|
| With dmap 1.) THF, DMF, RT, 1 h, 2.) THF, DMF, RT, 72 h; Yield given. Multistep reaction. Yields of byproduct given; | |
| With dmap In tetrahydrofuran; N,N-dimethyl-formamide |

-
-
84449-90-1
Raloxifen

-
-
166975-45-7
[2-(4-hydroxyphenyl)-6-hydroxybenzo[b]thien-3-yl][4-[2-(1-piperidinyl)ethoxy]phenyl]methane

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 91 percent / LiAlH4 / tetrahydrofuran / 6 h / 0 °C 2: 72 percent / Et3SiH, CF3CO2H / CH2Cl2 / 16 h / 0 °C View Scheme |
-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 20 percent / DMAP / tetrahydrofuran; dimethylformamide 2: 85 percent / Et3N / CH2Cl2 3: 1.) Pd(OAc)2, bis-diphenylphosphinopropane, Et3N, 2.) TBAF / 1) DMF; 2) CH2Cl2 View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) DMAP / 1.) THF, DMF, RT, 1 h, 2.) THF, DMF, RT, 72 h 2: 88 percent / Et3N / 1,2-dichloro-ethane / Ambient temperature 3: Et3N, Pd(OAc)2, 1,3-bis(diphenylphosphino)propane / dimethylformamide / 4 h / 100 °C 4: tetra-N-butylammoium fluoride / tetrahydrofuran / 3 h View Scheme |
-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 20 percent / DMAP / tetrahydrofuran; dimethylformamide 2: 85 percent / Et3N / CH2Cl2 3: 1.) Pd(OAc)2, bis-diphenylphosphinopropane, Et3N, 2.) TBAF / 1) DMF; 2) CH2Cl2 View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) DMAP / 1.) THF, DMF, RT, 1 h, 2.) THF, DMF, RT, 72 h 2: 88 percent / Et3N / 1,2-dichloro-ethane / Ambient temperature 3: Et3N, Pd(OAc)2, 1,3-bis(diphenylphosphino)propane / dimethylformamide / 10 h / 75 °C 4: tetra-N-butylammonium fluoride / tetrahydrofuran View Scheme |
-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 20 percent / DMAP / tetrahydrofuran; dimethylformamide 2: 85 percent / Et3N / CH2Cl2 3: 1.) Pd(OAc)2, bis-diphenylphosphinopropane, Et3N, 2.) TBAF / 1) DMF; 2) CH2Cl2 View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) DMAP / 1.) THF, DMF, RT, 1 h, 2.) THF, DMF, RT, 72 h 2: 88 percent / Et3N / 1,2-dichloro-ethane / Ambient temperature 3: Et3N, Pd(OAc)2, 1,3-bis(diphenylphosphino)propane / dimethylformamide / 19 h / 80 °C 4: tetra-N-butylammonium fluoride / tetrahydrofuran / 3 h View Scheme |
-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 20 percent / DMAP / tetrahydrofuran; dimethylformamide 2: 85 percent / Et3N / CH2Cl2 3: 1.) Pd(OAc)2, bis-diphenylphosphinopropane, Et3N, 2.) TBAF / 1) DMF; 2) CH2Cl2 4: NH3 / methanol View Scheme | |
| Multi-step reaction with 5 steps 1: 1.) DMAP / 1.) THF, DMF, RT, 1 h, 2.) THF, DMF, RT, 72 h 2: 88 percent / Et3N / 1,2-dichloro-ethane / Ambient temperature 3: 54 percent / Et3N, Pd(OAc)2, 1,3-bis(diphenylphosphino)propane / dimethylformamide / 4.5 h / 70 °C 4: MeAl(Cl)NH2 / toluene / Ambient temperature 5: 1N aq. HCl / tetrahydrofuran / 5 h / Heating View Scheme |
-
-
84449-90-1
Raloxifen

-
-
185416-94-8
4-{6-Hydroxy-3-[4-(2-piperidin-1-yl-ethoxy)-benzoyl]-benzo[b]thiophen-2-yl}-benzoic acid methyl ester

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 20 percent / DMAP / tetrahydrofuran; dimethylformamide 2: 85 percent / Et3N / CH2Cl2 3: 1.) Pd(OAc)2, bis-diphenylphosphinopropane, Et3N, 2.) TBAF / 1) DMF; 2) CH2Cl2 View Scheme | |
| Multi-step reaction with 3 steps 1: 1.) DMAP / 1.) THF, DMF, RT, 1 h, 2.) THF, DMF, RT, 72 h 2: 88 percent / Et3N / 1,2-dichloro-ethane / Ambient temperature 3: 27 percent / Et3N, Pd(OAc)2, 1,3-bis(diphenylphosphino)propane / dimethylformamide / 4.5 h / 70 °C View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) DMAP / 1.) THF, DMF, RT, 1 h, 2.) THF, DMF, RT, 72 h 2: 88 percent / Et3N / 1,2-dichloro-ethane / Ambient temperature 3: 54 percent / Et3N, Pd(OAc)2, 1,3-bis(diphenylphosphino)propane / dimethylformamide / 4.5 h / 70 °C 4: tetra-n-butylammonium fluoride / tetrahydrofuran View Scheme |
-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 20 percent / DMAP / tetrahydrofuran; dimethylformamide 2: 85 percent / Et3N / CH2Cl2 3: 1.) Pd(OAc)2, bis-diphenylphosphinopropane, Et3N, 2.) TBAF / 1) DMF; 2) CH2Cl2 4: Me3Al / toluene View Scheme | |
| Multi-step reaction with 5 steps 1: 1.) DMAP / 1.) THF, DMF, RT, 1 h, 2.) THF, DMF, RT, 72 h 2: 88 percent / Et3N / 1,2-dichloro-ethane / Ambient temperature 3: 54 percent / Et3N, Pd(OAc)2, 1,3-bis(diphenylphosphino)propane / dimethylformamide / 4.5 h / 70 °C 4: Me3Al / toluene / 21 h / 50 °C 5: tetra-N-butylammonium fluoride / tetrahydrofuran / 3 h View Scheme |
-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 20 percent / DMAP / tetrahydrofuran; dimethylformamide 2: 85 percent / Et3N / CH2Cl2 3: 1.) Pd(OAc)2, bis-diphenylphosphinopropane, Et3N, 2.) TBAF / 1) DMF; 2) CH2Cl2 View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) DMAP / 1.) THF, DMF, RT, 1 h, 2.) THF, DMF, RT, 72 h 2: 88 percent / Et3N / 1,2-dichloro-ethane / Ambient temperature 3: Et3N, Pd(OAc)2, 1,3-bis(diphenylphosphino)propane / dimethylformamide / 4.5 h / 70 °C 4: tetra-N-butylammonium fluoride / tetrahydrofuran / 3 h View Scheme |
-
-
84449-90-1
Raloxifen

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 20 percent / DMAP / tetrahydrofuran; dimethylformamide 2: 85 percent / Et3N / CH2Cl2 3: 1.) Pd(PPh3)4, 2.) TBAF / 1) MeCN; 2) CH2Cl2 View Scheme | |
| Multi-step reaction with 4 steps 1: 1.) DMAP / 1.) THF, DMF, RT, 1 h, 2.) THF, DMF, RT, 72 h 2: 88 percent / Et3N / 1,2-dichloro-ethane / Ambient temperature 3: Pd(PPh3)4, 2,6-lutidine / acetonitrile / 16 h / 70 °C 4: tetra-N-butylammonium fluoride / tetrahydrofuran / 3 h View Scheme |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View