Xi'an Xszo Chem Co., Ltd.
1. Factory price and high quality must be guaranteed, base on 8 years of production and R&D experience2. Free samples will be provided,ensure specifications and quality are right for customer3. Customers will receive the most professional technical s
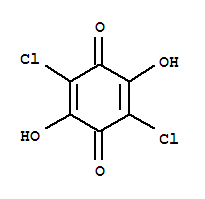
Cas:87-86-5
Min.Order:1 Gram
FOB Price: $0.1
Type:Manufacturers
inquiryAfine Chemicals Limited
excellent quality and reliable supplierAppearance:COA mentioned Storage:COA mentioned Package:Standard or custom package Application:pharmaceutical raw materails Transportation:sea/air /courier Port:China main port
Shanghai Chinqesen Biotechnology Co., Ltd.
Good Quality Package:1kg/bag Application:Medical or chemical Transportation:Air/Train/Sea Port:Shenzhen
Amadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Cas:87-86-5
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquirySynthetic route

| Conditions | Yield |
|---|---|
| With chlorine; aluminum (III) chloride at 100 - 180℃; for 10.8333h; Product distribution / selectivity; | A 83% B 10% |
| With chlorine; nickel; aluminum (III) chloride at 90 - 180℃; for 10h; Product distribution / selectivity; | A 79% B 20% |

| Conditions | Yield |
|---|---|
| With chlorine; nickel; aluminum (III) chloride at 90 - 180℃; for 4.16667 - 4.66667h; Product distribution / selectivity; | A 80% B 10% |
| Stage #1: 2,4,6-Trichlorophenol With chlorine; aluminum (III) chloride at 105 - 183℃; Stage #2: With phenol at 180 - 190℃; Product distribution / selectivity; | A 80% B 10% |
| With chlorine; aluminum (III) chloride at 105 - 190℃; for 3.66667 - 4.83333h; Product distribution / selectivity; | A 77% B 10% |
-
-
62269-80-1
pentachlorophenol-TMS

-
-
87-86-5
Pentachlorophenol

| Conditions | Yield |
|---|---|
| With nano magnetic sulfated zirconia (Fe3O4 at ZrO2/SO42-) In neat (no solvent) at 20℃; for 0.833333h; Green chemistry; | 80% |
-
-
17540-48-6
2,3,4,5,6-pentachloro-4-(pentachlorophenoxy)-2,5-cyclohexadienone

-
A
-
87-86-5
Pentachlorophenol

-
B
-
21567-21-5
2,3,5,6-tetrachloro-4-(2,3,4,5,6-pentachlorophenoxy)phenol

-
C
-
35245-80-8
3,4,5,6-tetrachloro-2-(2,3,4,5,6-pentachlorophenoxy)phenol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In methanol; benzene for 1h; Ambient temperature; | A 4 % Chromat. B 52.6% C 26 % Chromat. |


| Conditions | Yield |
|---|---|
| With sodium hydroxide In water for 3h; Heating; | 50% |
-
-
56-23-5
tetrachloromethane

-
-
626-02-8
3-Iodophenol

-
A
-
87-86-5
Pentachlorophenol

-
B
-
846042-42-0
2,4,6-trichloro-3-iodo-phenol

| Conditions | Yield |
|---|---|
| at 60 - 70℃; Einleiten von Chlor; |


| Conditions | Yield |
|---|---|
| With chlorosulfonic acid at 150 - 160℃; |

| Conditions | Yield |
|---|---|
| With tetrachloromethane; chlorine at 50℃; |

| Conditions | Yield |
|---|---|
| With chlorine |
-
-
118-74-1
hexachlorobenzene

-
-
124-41-4
sodium methylate

-
A
-
87-86-5
Pentachlorophenol

-
B
-
1825-21-4
Pentachloroanisole

| Conditions | Yield |
|---|---|
| at 180℃; im Rohr; | |
| at 180℃; im Rohr; |


| Conditions | Yield |
|---|---|
| With sodium hydroxide; glycerol at 250 - 280℃; | |
| With sodium hydroxide at 250℃; | |
| With sodium hydroxide; tert-butyl alcohol at 135℃; | |
| With sodium hydroxide; ethylene glycol at 200℃; | |
| With sodium hydroxide at 135℃; |

| Conditions | Yield |
|---|---|
| With sodium methylate at 180℃; im Rohr; |

| Conditions | Yield |
|---|---|
| With tetrachloromethane at 60 - 70℃; Einleiten von Chlor; |

| Conditions | Yield |
|---|---|
| With aluminium trichloride; chlorine; iron at 120 - 195℃; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid; tin(ll) chloride | |
| With carbon disulfide; aluminium trichloride; acetyl chloride Verseifung des Reaktionsprodukts; | |
| With potassium iodide |
-
-
599-52-0
2,3,4,4,5,6-hexachlorocyclohexa-2,5-dien-1-one

-
A
-
87-86-5
Pentachlorophenol

-
B
-
118-75-2
chloranil

| Conditions | Yield |
|---|---|
| With hydrogenchloride | |
| With aluminium trichloride at 160℃; im Druckrohr; |

| Conditions | Yield |
|---|---|
| durch Reduktion; |

| Conditions | Yield |
|---|---|
| With acetone | |
| With potassium acetate; acetic acid | |
| With sodium acetate; acetic acid |

| Conditions | Yield |
|---|---|
| With sulfuric acid; sodium nitrite at 180℃; |
-
-
846042-42-0
2,4,6-trichloro-3-iodo-phenol

-
A
-
87-86-5
Pentachlorophenol

| Conditions | Yield |
|---|---|
| With tetrachloromethane at 60 - 70℃; Einleiten von Chlor; |
-
-
38983-95-8
4,5,6-trihydroxy-benzene-1,3-disulfonic acid

-
-
87-86-5
Pentachlorophenol

| Conditions | Yield |
|---|---|
| With thionyl chloride at 100℃; im Rohr; |
-
-
7462-04-6
2,3,4,4,5,6,6-heptachloro-cyclohex-2-enone

-
-
127-08-2
potassium acetate

-
-
64-19-7
acetic acid

-
-
87-86-5
Pentachlorophenol


| Conditions | Yield |
|---|---|
| With chlorosulfonic acid at 150 - 160℃; |

| Conditions | Yield |
|---|---|
| With antimony(III) chloride; chlorine at 200 - 220℃; | |
| With aluminium trichloride; chlorine; 1,2-dichloro-ethane at 70 - 85℃; | |
| With aluminium trichloride; chlorine; 1,1,2,2-tetrachloroethane at 70 - 85℃; | |
| With tetrachloromethane; chlorine; iron(III) chloride at 20 - 25℃; | |
| With aluminium trichloride; chlorine at 100 - 190℃; |

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid at 150℃; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid Electrolysis; |
-
-
87-86-5
Pentachlorophenol

-
-
102386-32-3
Succinic acid mono-[(2R,3R,4R,5R)-2-[bis-(4-methoxy-phenyl)-phenyl-methoxymethyl]-5-(4-hydroxy-2-oxo-2H-pyrimidin-1-yl)-4-(tetrahydro-furan-2-yloxy)-tetrahydro-furan-3-yl] ester

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In N,N-dimethyl-formamide for 16h; Ambient temperature; | 100% |
-
-
680981-19-5
2-methoxy-succinic acid 1-(6-tert-butoxycarbonylamino-hexyl) ester

-
-
87-86-5
Pentachlorophenol

-
-
680981-21-9
pentachlorophenyl N-tert-butoxycarbonyl-3-methoxy-4-oxo-5-oxy-11-aminoundecanoate

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In ethyl acetate at 20℃; for 120h; | 100% |
-
-
865623-19-4
(E)-[2-(phenylazo)phenyl]diphenylphosphine

-
-
87-86-5
Pentachlorophenol

-
-
1374254-19-9
C6Cl5O(1-)*C24H20N2P(1+)

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at -60℃; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: Pentachlorophenol With potassium carbonate In butanone at 20℃; for 0.25h; Stage #2: α-bromoacetophenone In butanone at 80℃; | 100% |

| Conditions | Yield |
|---|---|
| With silver trifluoromethanesulfonate at 60℃; for 0.0833333h; neat (no solvent); | 99% |
| With dicyclohexyl-carbodiimide In acetonitrile at 25℃; for 1h; | 91% |
| With sodium acetate |
-
-
87-86-5
Pentachlorophenol

-
-
39206-48-9
pentachlorobenzenesulfonic anhydride

-
A
-
608-93-5
pentachlorobenzene

-
B
-
40707-29-7
pentachlorobenzenesulfonic acid

| Conditions | Yield |
|---|---|
| at 200 - 210℃; for 0.5h; | A 32% B 99% C 39% |

-
-
87-86-5
Pentachlorophenol

-
-
120188-25-2
Succinic acid mono-[3-[bis-(4-methoxy-phenyl)-phenyl-methoxy]-2-(5-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethoxy)-propyl] ester

-
-
120188-26-3
1-<<2-dimethoxytrityl-hydroxy-1-(pentachlorophenylsuccinyl-hydroxy-methyl)ethoxy>-methyl>thymine

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide | 99% |
-
-
87-86-5
Pentachlorophenol

-
-
66003-76-7
Diphenyliodonium triflate

-
-
22274-42-6
2,3,4,5,6-Pentachlorobiphenyl ether

| Conditions | Yield |
|---|---|
| Stage #1: Pentachlorophenol With potassium tert-butylate In tetrahydrofuran at 0℃; for 0.25h; Stage #2: Diphenyliodonium triflate In tetrahydrofuran at 40℃; for 2h; | 99% |
| Stage #1: Pentachlorophenol With potassium tert-butylate In tetrahydrofuran at 0℃; for 0.25h; Stage #2: Diphenyliodonium triflate In tetrahydrofuran at 40℃; for 2h; | 99% |
-
-
87-86-5
Pentachlorophenol

-
-
33756-42-2
tri-p-fluorophenylstibine

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide In diethyl ether; water at 20℃; for 24h; | 99% |
-
-
87-86-5
Pentachlorophenol

-
-
98015-53-3
1,2,2,2-tetrachloroethyl chloroformate

-
-
107960-04-3
pentachlorophenyl 1,2,2,2-tetrachloroethyl carbonate

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane for 3h; Ambient temperature; | 98% |
-
-
87-86-5
Pentachlorophenol

-
-
102386-34-5
Succinic acid mono-[(2R,3R,4R,5R)-2-[bis-(4-methoxy-phenyl)-phenyl-methoxymethyl]-5-(6-hydroxy-2-isobutyrylamino-purin-9-yl)-4-(tetrahydro-furan-2-yloxy)-tetrahydro-furan-3-yl] ester

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In N,N-dimethyl-formamide for 16h; Ambient temperature; | 98% |

| Conditions | Yield |
|---|---|
| With nickel dichloride; RedAl In tetrahydrofuran at 68℃; for 2h; Product distribution; other dehalogenating systems, other reaction times and temperatures; | A n/a B 98% |
-
-
87-86-5
Pentachlorophenol

-
-
309965-65-9
Ru(CH2C(CH3)CH2)2(((CH3)2CH)2PCH2P(C6H5)2)

-
-
309965-71-7
Ru(OC6Cl5)2(((CH3)2CH)2PCH2P(C6H5)2)

| Conditions | Yield |
|---|---|
| In toluene (Ar); stirring a soln. of ruthenium complex and pentachlorophenol in toluene for 30 min at room temp.; evapn., washing with EtOH and pentane, drying; elem. anal.; | 98% |

| Conditions | Yield |
|---|---|
| In diethyl ether at 20℃; for 24h; | 98% |

-
-
2158-02-3
morpholine-4-carboxylic acid amide

-
-
87-86-5
Pentachlorophenol

-
-
87448-70-2
2,3,4,5,6-Pentachloro-phenol; compound with morpholine-4-carboxylic acid amide

| Conditions | Yield |
|---|---|
| 96% |
-
-
87-86-5
Pentachlorophenol

-
-
16420-13-6
N,N-Dimethylthiocarbamoyl chloride

-
-
19387-18-9
O-pentachlorophenyl dimethylthiocarbamate

| Conditions | Yield |
|---|---|
| With potassium hydroxide In 1,4-dioxane 1.) 30 deg C, 1 h; 2.) 85 deg C, 3 h; | 96% |
-
-
87-86-5
Pentachlorophenol

-
-
115-77-5
Pentaerythritol

-
-
65284-06-2
3,9-bis(2',3',4',5',6'-pentachlorophenoxy)-2,4,8,10-tetraoxa-3,9-diphosphaspiro(5.5)undecane-3,9-dioxide

| Conditions | Yield |
|---|---|
| With potassium chloride; trichlorophosphate In toluene | 95.3% |

| Conditions | Yield |
|---|---|
| With bis-[(trifluoroacetoxy)iodo]benzene In various solvent(s) at 20℃; for 1h; pH=1.4; Oxidation; | 95% |
| With nitric acid | |
| With hydrogenchloride; chlorine |
-
-
87-86-5
Pentachlorophenol

-
-
309965-66-0
Ru(CH2C(CH3)CH2)2((C6H11)2PCH2P(C6H5)2)

-
-
309965-72-8
Ru(OC6Cl5)2((C6H11)2PCH2P(C6H5)2)

| Conditions | Yield |
|---|---|
| In toluene (Ar); stirring a soln. of ruthenium complex and pentachlorophenol in toluene for 30 min at room temp.; evapn., washing with EtOH and pentane, drying; elem. anal.; | 95% |
-
-
87-86-5
Pentachlorophenol

-
-
603-36-1
triphenylantimony

-
-
17363-17-6
di(pentachlorophenolato)triphenylantimony(V)

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide In diethyl ether at 20℃; for 24h; | 95% |

| Conditions | Yield |
|---|---|
| In toluene at 24℃; for 0.25h; | 95% |

-
-
87-86-5
Pentachlorophenol

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide In diethyl ether; water at 20℃; for 24h; | 95% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide In diethyl ether at 20℃; for 24h; | 94% |

| Conditions | Yield |
|---|---|
| With 3-(5-nitro-2-oxo-1,2-dihydro-1-pyridyl)-1,2-benzisothiazole 1,1,-dioxide; triethylamine In dichloromethane -10 deg C -> r.t., overnight; | 93% |
| With TEA; 3-(5-nitro-2-oxo-1,2-dihydro-1-pyridyl)-1,2-benzisothiazole 1,1,-dioxide In dichloromethane -10 deg C to room temperature; | 93% |
| With acetic anhydride |
-
-
87-86-5
Pentachlorophenol


-
-
89239-05-4
Succinic acid (3aR,4R,6R,6aR)-6-(2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-yl)-2-methoxy-tetrahydro-furo[3,4-d][1,3]dioxol-4-ylmethyl ester pentachlorophenyl ester

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In N,N-dimethyl-formamide | 93% |
-
-
87-86-5
Pentachlorophenol

-
-
13328-67-1, 51017-91-5
penta(p-tolyl)antimony(V)

| Conditions | Yield |
|---|---|
| In toluene at 24℃; for 0.25h; | 93% |
-
-
87-86-5
Pentachlorophenol

| Conditions | Yield |
|---|---|
| In hexane at -30 - 20℃; for 14.5h; Sealed tube; | 93% |

| Conditions | Yield |
|---|---|
| With dicyclohexyl-carbodiimide In acetonitrile at 25℃; for 1h; | 92% |
-
-
87-86-5
Pentachlorophenol

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
2613-89-0
phenylmalonic acid

-
-
59530-33-5
dipentachlorophenyl phenylmalonate

| Conditions | Yield |
|---|---|
| With pyridine In methanol; dichloromethane | 92% |

-
-
87-86-5
Pentachlorophenol

-
-
262359-92-2
[Ru(η3-2-MeC3H4)2(κ2-Ph2PCH2CH2PiPr2)]

| Conditions | Yield |
|---|---|
| In toluene (Ar); -30°C; warmed to room temp.; stirred (10 min); solvent evapd. (vac.); washed (pentane); dried; elem. anal.; | 92% |
Related products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View















 T+;
T+;  N;
N;  Xn
Xn