Lonwin Chemical Group Limited
Thionyl chloride CAS:7719-09-7 Specification Welcome to contact us to get complete COA. Shanghai Lonwin Chemical Co.,Ltd is a leading manufacturer and supplier of chemicals in China.We develop,produce and distribute high quality pharmaceutic
Hunan Russell Chemicals Technology Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:Foil bag; Drum; Plastic bottle Application:Pharma;Industry;Agricultural Transportation:by sea or air Port:any port in China
Shang Hai Grade Biochemical Co,. Limited
low price and good quality Application:It is an important raw material and intermediate used in Organic Synthesis, Pharmaceuticals, Agrochemicals and D
Synthetic route
-
-
10545-99-0
sulfur dichloride

-
-
7446-11-9
sulfur trioxide

-
A
-
7719-09-7
thionyl chloride

-
B
-
7446-09-5
sulfur dioxide

| Conditions | Yield |
|---|---|
| In neat (no solvent) below -10°C or under pressure;; | A 100% B n/a |

-
-
7647-01-0
hydrogenchloride

-
-
222851-56-1
N-phenylsulfinylamine

-
A
-
7719-09-7
thionyl chloride

-
B
-
142-04-1
aniline hydrochloride

| Conditions | Yield |
|---|---|
| In Petroleum ether decompn. of thioaniline dissolved in dild. petroleum (b.p. 200-250°C) on introduction of dry HCl (shaking) at -10°C;; filtration (anilinium chloride); fractionation;; | A 94% B n/a |

-
-
10545-99-0
sulfur dichloride

-
-
7446-11-9
sulfur trioxide

-
A
-
7719-09-7
thionyl chloride

-
B
-
7791-27-7
pyrosulfuryl chloride

-
C
-
7446-09-5
sulfur dioxide

| Conditions | Yield |
|---|---|
| In neat (no solvent) SO3 is distilled (from 65 % oleum) into cooled (ice/water) vessel containing SCl2; evolution of SO2;; multiple fractionation (at last in presence of sulfur);; | A 80% B n/a C n/a |


| Conditions | Yield |
|---|---|
| With sulfur dioxide In neat (no solvent) reaction of CCl4 with SO2 on heating to 200°C;; | A n/a B 72% |
-
-
594-44-5
Ethanesulfonyl chloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
75-00-3
chloroethane

-
C
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
D
-
10025-87-3, 12599-09-6, 63736-95-8
trichlorophosphate

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
98-09-9
benzenesulfonyl chloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
108-90-7
chlorobenzene

-
C
-
10025-87-3, 12599-09-6, 63736-95-8
trichlorophosphate

| Conditions | Yield |
|---|---|
| at 200 - 210℃; |


| Conditions | Yield |
|---|---|
| at 200℃; unter Druck; |


| Conditions | Yield |
|---|---|
| at 200 - 400℃; |

-
-
2547-61-7
Trichloromethanesulfonyl chloride

-
A
-
56-23-5
tetrachloromethane

-
B
-
75-44-5
phosgene

-
C
-
7719-09-7
thionyl chloride

| Conditions | Yield |
|---|---|
| at 200℃; |


| Conditions | Yield |
|---|---|
| at 150℃; |

-
-
623-81-4
diethyl sulphite

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
6378-11-6
ethyl chlorosulfite

-
C
-
75-00-3
chloroethane

| Conditions | Yield |
|---|---|
| bei niedrigerer Temperatur; |

-
-
623-98-3
dipropyl sulfite

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
540-54-5
1-Chloropropane

| Conditions | Yield |
|---|---|
| beim Erwaermen; |

-
-
626-85-7
sulfurous acid dibutyl ester

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
109-69-3
n-Butyl chloride

| Conditions | Yield |
|---|---|
| bei schwachem Erwaermen; |

-
-
7446-70-0
aluminium trichloride

-
-
7791-25-5
sulfuryl dichloride

-
-
95-94-3
1,2,4,5-tetrachlorobenzene

-
A
-
56-23-5
tetrachloromethane

-
B
-
7719-09-7
thionyl chloride

-
C
-
608-93-5
pentachlorobenzene

-
D
-
118-74-1
hexachlorobenzene

-
-
6378-11-6
ethyl chlorosulfite

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
75-00-3
chloroethane

-
C
-
10025-87-3, 12599-09-6, 63736-95-8
trichlorophosphate

| Conditions | Yield |
|---|---|
| at 25℃; |

-
-
98-67-9
p-hydoroxybenzenesulfonic acid

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
106-46-7
para-dichlorobenzene

-
C
-
772-79-2
4-chlorophenylphosphorodichloridate

-
D
-
10025-87-3, 12599-09-6, 63736-95-8
trichlorophosphate

| Conditions | Yield |
|---|---|
| Reaktion des Kaliumsalzes; |

-
-
623-81-4
diethyl sulphite

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
75-00-3
chloroethane

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
32337-45-4
4,4'-sulfonyl-bis-benzenesulfonyl chloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
106-46-7
para-dichlorobenzene

-
C
-
10025-87-3, 12599-09-6, 63736-95-8
trichlorophosphate

-
D
-
7719-12-2, 52843-90-0
phosphorus trichloride

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
-
85-46-1
1-Naphthalenesulfonyl chloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
10025-87-3, 12599-09-6, 63736-95-8
trichlorophosphate

-
C
-
90-13-1
1-Chloronaphthalene

| Conditions | Yield |
|---|---|
| at 150 - 160℃; |

-
-
4895-65-2
phenyltetrachlorophosphorane

-
A
-
824-72-6
P,P-dichlorophenylphosphine oxide

-
B
-
7719-09-7
thionyl chloride


| Conditions | Yield |
|---|---|
| at 70℃; Einleiten von HCl; |
-
-
13165-73-6
phenyl chlorosulphinate

-
-
10025-87-3, 12599-09-6, 63736-95-8
trichlorophosphate

-
A
-
7719-09-7
thionyl chloride

-
B
-
115-86-6
phosphoric acid triphenyl ester

-
-
861087-97-0
2-benzoylamino-[1]naphthoic acid

-
A
-
7719-09-7
thionyl chloride
-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
95-50-1
1,2-dichloro-benzene

| Conditions | Yield |
|---|---|
| at 200 - 210℃; |


-
A
-
7719-09-7
thionyl chloride

-
B
-
40881-98-9
N,N-dipropylphosphoramidic dichloride

-
C
-
10025-87-3, 12599-09-6, 63736-95-8
trichlorophosphate

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
7719-09-7
thionyl chloride

-
B
-
100-44-7
benzyl chloride

-
C
-
10025-87-3, 12599-09-6, 63736-95-8
trichlorophosphate


-
A
-
7719-09-7
thionyl chloride

| Conditions | Yield |
|---|---|
| In acetone |
-
-
75-15-0
carbon disulfide

-
-
7791-21-1
hypochlorous anhydride

-
A
-
75-44-5
phosgene

-
B
-
7719-09-7
thionyl chloride

| Conditions | Yield |
|---|---|
| In tetrachloromethane CS2 dild. in CCl4 (to diminuish vehemence of reaction);; | |
| In tetrachloromethane |

-
-
7719-09-7
thionyl chloride

-
-
114786-39-9
methyl (S)-4,5,6,7-tetrahydro-3H-imidazo[4,5-c]pyridine-6-carboxylate hydrochloric acid salt

| Conditions | Yield |
|---|---|
| In methanol for 3.5h; Heating / reflux; | 100% |
-
-
7719-09-7
thionyl chloride

| Conditions | Yield |
|---|---|
| In thionyl chloride (argon); stirring (ca. 16 h); pptn. with Et2O, washing (Et2O), drying (vac.); | 100% |
-
-
7719-09-7
thionyl chloride

| Conditions | Yield |
|---|---|
| In thionyl chloride (argon); stirring (ca. 16 h); pptn. with Et2O, washing (Et2O), drying (vac.); | 100% |
-
-
7719-09-7
thionyl chloride

-
-
173105-66-3
(5,7,12,14-tetramethyldibenzo[b,i][1,4,8,11]tetraazacyclotetradecinate(2-))VCl2

| Conditions | Yield |
|---|---|
| In dichloromethane (Ar); react. (0°C), warming (room temp.), stirring (6 h), solventevapn.; washing (diethyl ether, pentane), drying (high vacuum, 50°C, 8 h); | 100% |

| Conditions | Yield |
|---|---|
| In thionyl chloride vac., 150°C, 3 d; | 100% |
-
-
7719-09-7
thionyl chloride

-
-
6575-24-2
2-(2,6-dichlorophenyl)acetic acid

-
-
880089-70-3
1-bromo-3-(2,6-dichlorophenyl)propan-2-one

| Conditions | Yield |
|---|---|
| Stage #1: thionyl chloride; 2-(2,6-dichlorophenyl)acetic acid With diazomethyl-trimethyl-silane Stage #2: With hydrogen bromide In water | 100% |

| Conditions | Yield |
|---|---|
| 100% |
-
-
7719-09-7
thionyl chloride

-
-
1376437-09-0
4-[1-(trifluoroacetyl)piperidin-4-yl]benzoic acid

-
-
1331747-36-4
4-(1-(2,2,2-trifluoroacetyl)piperidin-4-yl)benzoyl chloride

| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 70℃; for 5h; Sealed tube; | 100% |

| Conditions | Yield |
|---|---|
| With triethylamine In ethyl acetate at 35℃; for 4h; Inert atmosphere; | 100% |
-
-
7719-09-7
thionyl chloride

| Conditions | Yield |
|---|---|
| at 80℃; for 12h; | 100% |
-
-
67-56-1
methanol

-
-
7719-09-7
thionyl chloride

-
-
5334-40-7
4-nitro-1H-pyrazole-3-carboxylic acid

-
-
138786-86-4
methyl 4-nitro-1H-pyrazole-3-carboxylate

| Conditions | Yield |
|---|---|
| at 0 - 25℃; for 16 - 48h; Product distribution / selectivity; | 99.5% |


| Conditions | Yield |
|---|---|
| In diethyl ether byproducts: SO2; to finely ground dry Ag salt in anhyd. Et2O cooled to 0°C added dropwise with vigorous stirring equimolar amt. of thionyl chloride; suspn. stirred for 15 min; soln. was decanted from AgCl; AgCl dried; soln. washed (Et2O); all extracts combined; cooled to 0°C; | A n/a B 99% |


| Conditions | Yield |
|---|---|
| In diethyl ether byproducts: SO2, SO3; to finely ground dry Ag salt in anhyd. Et2O cooled to 0°C added dropwise with vigorous stirring equimolar amt. of thionyl chloride; suspn. stirred for 15 min; soln. was decanted from AgCl; AgCl dried; soln. washed (Et2O); all extracts combined; cooled to 0°C; | A n/a B n/a C 99% |

-
-
7719-09-7
thionyl chloride

| Conditions | Yield |
|---|---|
| In dichloromethane (N2); stirring (4 h, reflux); solvent removal (reduced pressure), drying (vac., 60-70 degree.C, 1 h); elem. anal.; | 99% |
-
-
7719-09-7
thionyl chloride

| Conditions | Yield |
|---|---|
| In dichloromethane SOCl2 added to CH2Cl2 soln. of B compd. (molar ratio 1:3, 1:4); concd. slowly; ppt. purified by column chromy. (silica gel, CH2Cl2); | 99% |
| In neat (no solvent) excess of SOCl2 added to B compd.; purified by column chromy. (silica gel, CH2Cl2); | 99% |
| In dichloromethane SOCl2 added to CH2Cl2 soln. of B compd. (molar ratio 2:1, 1:1, 1:2); concd. slowly; mixt. of the starting B compd. and title compd. pptd.; purified by column chromy. (silica gel, CH2Cl2); | 15% |
-
-
7719-09-7
thionyl chloride

-
-
864767-27-1
[(C5(CH3)5)TiCl(C5H4C(CH3)2CH2CO2)]

-
-
864767-39-5
[(C5(CH3)5)TiCl2(C5H4C(CH3)2CH2COCl)]

| Conditions | Yield |
|---|---|
| In not given | 99% |
-
-
7719-09-7
thionyl chloride

-
-
864766-99-4
chloro(cyclopentadienyl)(cyclopentadienyl-3-methyl-3-butanoate)titanium(IV)

-
-
864767-00-0
Cp(CpCMe2CH2COCl)titanium(IV)dichloride

| Conditions | Yield |
|---|---|
| In not given | 99% |
| In neat (no solvent) at 20℃; for 1h; Inert atmosphere; Schlenk technique; Glovebox; |
-
-
7719-09-7
thionyl chloride

-
-
864767-24-8
[(C5H5)TiCl(C5H4C(CH2)5CH2CO2)]

-
-
864767-36-2
[(C5H5)TiCl2(C5H4C(CH2)5CH2COCl)]

| Conditions | Yield |
|---|---|
| In not given | 99% |
-
-
7719-09-7
thionyl chloride

-
-
864766-99-4
chloro(cyclopentadienyl)(cyclopentadienyl-3-methyl-3-butanoate)titanium(IV)

-
-
57-88-5
cholesterol

| Conditions | Yield |
|---|---|
| With sodium hydride In dichloromethane mixt. of Ti complex and SOCl2 was stirred for 3 h; evapd. (vac., 80°C); dissolved in CH2Cl2; added to mixt. of NaH and cholesterol; stirred for 16 h; filtered; solvent removed (vac.); crystd. (CH2Cl2); | 99% |


| Conditions | Yield |
|---|---|
| With sodium hydride In dichloromethane mixt. of Ti complex and SOCl2 was stirred for 3 h; evapd. (vac., 80°C); dissolved in CH2Cl2; added to mixt. of NaH and cholesterol; stirred for 16 h; filtered; solvent removed (vac.); crystd. (CH2Cl2); | 99% |

-
-
7719-09-7
thionyl chloride

-
-
1202357-35-4
(4R,5S)-5-phenylthio-4-octanol

-
-
1202357-36-5
((4S,5R)-5-chlorooctan-4-yl)(phenyl)sulfane

| Conditions | Yield |
|---|---|
| With N,N-dimethyl-formamide In dichloromethane at 0℃; for 0.5h; Inert atmosphere; optical yield given as %ee; | 99% |
-
-
7719-09-7
thionyl chloride

-
-
50571-15-8
1,7-dihydroxycarbonyl-1,7-dicarba-closo-dodecaborane

-
-
23810-52-8
m-carborane-1,7-dicarboxylic acid dichloride

| Conditions | Yield |
|---|---|
| In thionyl chloride B compd. and SOCl2 placed into a flask under N2, refluxed for 24 h; evapd. (vac.); obtained as an oil; | 99% |
-
-
7719-09-7
thionyl chloride

-
-
372515-55-4
tris(8-carboxyl-2,2,6,6-tetramethylbenzo[1,2-d;4,5-d′]bis[1,3]dithiol-4-yl)methyl radical sodium salt

-
-
1079975-19-1
C40H36Cl3O3S12

| Conditions | Yield |
|---|---|
| Stage #1: tris(8-carboxyl-2,2,6,6-tetramethylbenzo[1,2-d;4,5-d′]bis[1,3]dithiol-4-yl)methyl radical sodium salt With triethylamine In chloroform at 20℃; for 0.5h; Stage #2: thionyl chloride In chloroform at 20 - 50℃; | 99% |

| Conditions | Yield |
|---|---|
| In acetonitrile TlCl suspd. in CH3CN; Cl2 added under vigorous stirring; stirred for 30 min; soln. filtered; solvent evapd. in vacuum; SOCl2 added; evapd. after 10 min; | 98.5% |

-
-
7719-09-7
thionyl chloride

-
-
10113-29-8
tetrabenzyltin

-
A
-
41719-05-5
phenylmethanesulfinyl chloride

-
B
-
3002-01-5
dibenzyltin(IV) dichloride

| Conditions | Yield |
|---|---|
| In not given dropwise addn. of thionyl chloride to tetraalkyltin(IV) compd. under dry N2 atmosphere under ice cooling, warming to room temp., refluxing for 30 min; distn. (removal of excess of SOCl2), cooling resulting liquids, sepn., elem. anal.; | A n/a B 98% |

-
-
7719-09-7
thionyl chloride

-
-
69934-86-7
(29H,31H-2,9,16,23-tetracarboxyphthalocyaninato-N(29),N(30),N(31),N(32))cobalt(II)

-
-
73533-44-5
cobalt tetra-4-(chlorocarbonyl)phthalocyanine

| Conditions | Yield |
|---|---|
| In benzene a mixt. in dry benzene was refluxed for 7 h; filtered, washed with dry benzene, dried in a vac. at room temp.; | 98% |
| With pyridine In benzene at 80℃; | 96% |
-
-
7719-09-7
thionyl chloride

-
-
72850-61-4
2-chloro-4-(trifluoromethyl)-5-thiazolecarboxylic acid

-
-
72850-62-5
2-chloro-4-(trifluoromethyl)-5-thiazolecarbonyl chloride

| Conditions | Yield |
|---|---|
| 6 h refluxing with excess SOCl2; | 98% |
-
-
7719-09-7
thionyl chloride

-
-
683816-30-0
(m-carborane-9,10-diyl)diacetic acids

-
-
683816-32-2
(m-carboranyl-9,10-diyl)di(acetyl chlorides)

| Conditions | Yield |
|---|---|
| In benzene byproducts: SO2, HCl; addn. of SOCl2 to soln. of (CH2COOH)2C2B10H10 in dry benzene at 20°C; heating for 2 h; cooling, removal of solvent and excess SOCl2 in vac.; | 98% |
-
-
7719-09-7
thionyl chloride

-
-
1489175-55-4
C6H3F2N2(1+)*Cl(1-)

-
-
210532-24-4
2,3-difluorobenzenesulphonyl chloride

| Conditions | Yield |
|---|---|
| Stage #1: thionyl chloride With copper(l) chloride In water at -5 - 20℃; Stage #2: C6H3F2N2(1+)*Cl(1-) In water at -5℃; for 0.5h; | 98% |
Related products
Raw Materials
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View




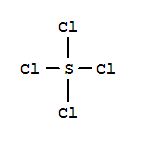










 C
C