Synthetic route

-
-
7446-11-9
sulfur trioxide

| Conditions | Yield |
|---|---|
| With chlorine In neat (no solvent) at 1100 - 1150°C;; | 100% |
| With chlorine In neat (no solvent) addn. of SiO2, NaCl or Na2SO4 increase reaction temp.; react. accelerated;; | |
| With chlorine In neat (no solvent) in presence of charcoal strong react.; addn. of NiSO4, CuSO4, MgSO4, Fe2O3 or KCl reduced yield; no influence of overheated water vapor;; react. detected at 700 - 1150°C;; |

| Conditions | Yield |
|---|---|
| manganese(IV) oxide 450°C; | 98% |
| 2Na2O*3MoO3*V2O5 440°C; | 98.4% |
| two-stage catalyst contact cooled by SO2 gas, external heat exchangers, temp. in contact bed:400-411°C; | 98% |

| Conditions | Yield |
|---|---|
| vanadia Kinetics; 437°C; | 98% |
| platinum Kinetics; 415°C; | 98% |
| vanadia Kinetics; 452°C; | 97% |

-
-
7446-11-9
sulfur trioxide

| Conditions | Yield |
|---|---|
| 3-stage contact bed; | 98% |
| no C content in pyrite; | 96% |
| no C content in pyrite; | 96% |

| Conditions | Yield |
|---|---|
| With catalyst: Fe compd. High Pressure; higher yield with increasing pressure, 600-630°C at 100 at; | 97% |
| With catalyst: Fe compd. High Pressure; higher yield with increasing pressure, 600-630°C at 100 at; | 97% |
| Kinetics; 420°C; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; copper dichloride In neat (no solvent) Kinetics; (450°C, 1.5 s); | 95% |
| With hydrogenchloride SO3 stabilized as Lewis-addukt(HCl.SO3); O2: 30l/h, HCl: 8l/h; 60min; | 40% |
| Kinetics; 420°C; |

| Conditions | Yield |
|---|---|
| With oxygen In neat (no solvent) heating in a stream of O2 at 550 - 600°C (formation of SO2 from 82 % of S and of Ag2SO4), later at 980 - 1070°C (decomposition of Ag2SO4);; | A 95% B 5% |
| With O2 In neat (no solvent) heating in a stream of O2 at 550 - 600°C (formation of SO2 from 82 % of S and of Ag2SO4), later at 980 - 1070°C (decomposition of Ag2SO4);; | A 95% B 5% |
-
-
25362-97-4
SbCl5*SO3

-
A
-
7791-27-7
pyrosulfuryl chloride

-
C
-
7647-18-9
antimonypentachloride

-
D
-
7446-11-9
sulfur trioxide

| Conditions | Yield |
|---|---|
| 70°C in vac.; | A n/a B n/a C n/a D 95% |


-
A
-
608-93-5
pentachlorobenzene

-
B
-
7446-11-9
sulfur trioxide

-
C
-
1043-49-8
Bis-pentachlor-phenyl-quecksilber

| Conditions | Yield |
|---|---|
| 130-220°C; | A <1 B 80% C 64% |


| Conditions | Yield |
|---|---|
| 130-220°C; | A 78% B 80% |

| Conditions | Yield |
|---|---|
| With pyrographite 700°C; | A n/a B 80% C n/a |
| With C 700°C; | A n/a B 80% C n/a |
| 700°C; | A n/a B 50% C n/a |

-
-
126885-21-0
disulfur dioxide

-
A
-
7446-09-5
sulfur dioxide

-
B
-
7446-11-9
sulfur trioxide

-
C
-
7704-34-9
sulfur

| Conditions | Yield |
|---|---|
| With oxygen In neat (no solvent) oxidn. of S2O2 with an excess of O2, min. ignition pressure at 32°C 54 Torr, at 150°C 2 Torr, mechanism discussed, sometimes react. with lightening, inhibition by SO2;; | A 10-25 B 15-30 C 80% |


-
A
-
363-72-4
Pentafluorobenzene

-
B
-
313-50-8
pentafluorobenzenesulfonic acid

-
C
-
7446-11-9
sulfur trioxide

-
D
-
973-17-1
bis(pentafluorophenyl)mercury(II)

| Conditions | Yield |
|---|---|
| 130-220°C; | A 12% B 9% C 70% D 53% |
| 130-240°C; | |
| 130-240°C; |


-
A
-
2262-05-7
(p-HC6F4)2Hg

-
B
-
327-54-8
1,2,4,5-Tetrafluorobenzene

-
C
-
40707-55-9
2,3,5,6-tetrafluorobenzenesulfonic acid

-
D
-
7446-11-9
sulfur trioxide

| Conditions | Yield |
|---|---|
| 130-230°C; | A 17% B 17% C 17% D 60% |


| Conditions | Yield |
|---|---|
| 130-220°C; | A 44% B 60% |
-
-
12395-41-4
bis(pentafluorosulfur) peroxide

-
-
7446-09-5
sulfur dioxide

-
A
-
173009-97-7, 118492-84-5, 13709-54-1
thionyl tetrafluoride

-
B
-
81439-35-2
pentafluorosulfur fluorosulfonate

-
C
-
7446-11-9
sulfur trioxide

-
D
-
7783-42-8
thionyl fluoride

-
E
-
2551-62-4
sulfur(VI) hexafluoride

| Conditions | Yield |
|---|---|
| byproducts: SiF4, sulfur; other Radiation; photochemical reaction with 253.7 nm radiation, 48 h; | A n/a B 45% C n/a D n/a E n/a |


| Conditions | Yield |
|---|---|
| In gas mixt. of N2 and sulfur vapor; O2: 100l/h; 15min, tube system, products cooled; | A n/a B 41% |
| 1000-1370°C, rapid cooling to minimize SO3 formation; | |
| very low SO3 content; |


| Conditions | Yield |
|---|---|
| 130-250°C; | A 15% B 10% C 40% |


| Conditions | Yield |
|---|---|
| 995 °C in N2; part of a Mg-S-I water splitting cycle; | A 30% B 30% |
| In neat (no solvent) heating at 950-1050 K (effusion orifices with A/a ratios of 10-30); | |
| 1120-1200°C; 29,1mol% in 15min; 90mol% in 2h; | |
| 900°C, in air stream; |

-
-
7446-11-9
sulfur trioxide

| Conditions | Yield |
|---|---|
| In neat (no solvent) evolution of SO3 by heating FeSO4 in a glass tube by little admission of air; beginning at 590 °C, only 3% after heating 2h at 625 to 635 °C;; | 3% |
-
-
75-15-0
carbon disulfide

-
A
-
7446-11-9
sulfur trioxide

-
B
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

| Conditions | Yield |
|---|---|
| Bei der Explosion mit ueberschuessigem Sauerstoff; |

-
-
63549-29-1
disulfuric acid carbonylamide chloride

-
A
-
1189-71-5
isocyanate de chlorosulfonyle

-
B
-
7446-11-9
sulfur trioxide

| Conditions | Yield |
|---|---|
| at 160℃; |
-
-
54060-35-4
2,3-disulfosuccinic acid

-
A
-
7446-11-9
sulfur trioxide

-
B
-
7732-18-5
water

-
C
-
15719-64-9, 15719-76-3, 97762-63-5
methylammonium carbonate

-
-
7647-01-0
hydrogenchloride

-
A
-
50-00-0
formaldehyd

-
B
-
7664-93-9
sulfuric acid

-
C
-
7446-11-9
sulfur trioxide

-
D
-
302-01-2
hydrazine


| Conditions | Yield |
|---|---|
| at 155℃; beim Schmelzen; |

-
-
7791-27-7
pyrosulfuryl chloride

-
-
10034-85-2
hydrogen iodide

-
A
-
7647-01-0
hydrogenchloride

-
B
-
7446-09-5
sulfur dioxide

-
C
-
7446-11-9
sulfur trioxide

-
D
-
7553-56-2
iodine

| Conditions | Yield |
|---|---|
| In neat (no solvent) violent reaction of dry HI even at temp. of a cold mixture of ice and sodium chloride;; |

-
-
13036-75-4
fluorosulfonyl anhydride

-
A
-
640723-20-2, 2699-79-8, 12769-73-2
fluorosulfonyl fluoride

-
B
-
7446-11-9
sulfur trioxide

| Conditions | Yield |
|---|---|
| 500°C; | |
| 500°C; |
-
-
10545-99-0
sulfur dichloride

-
-
7446-11-9
sulfur trioxide

-
A
-
7719-09-7
thionyl chloride

-
B
-
7446-09-5
sulfur dioxide

| Conditions | Yield |
|---|---|
| In neat (no solvent) below -10°C or under pressure;; | A 100% B n/a |


| Conditions | Yield |
|---|---|
| SO3 in excess, 10-15 min, closed vessel 300°C; | 100% |
| In neat (no solvent) formation on heating Li2SO4 in a SO3-atmosphere to .300°C;; | |
| In neat (no solvent) formation on heating Li2SO4 in a SO3-atmosphere to .300°C;; | |
| In neat (no solvent) reaction of SO3-vapour with Li2SO4 at 450°C; TGA; |

| Conditions | Yield |
|---|---|
| SO3 in excess, 10-15 min, closed vessel 450°C; | 100% |
| react. of Na2SO4 with SO3 at ambient temp.;; substance with Na2S2O7 and unchanged Na2SO4 obtained;; | |
| In neat (no solvent) react. of Na2SO4 with an excess of SO3 above 150°C;; |

| Conditions | Yield |
|---|---|
| In 1,2-dichloro-ethane dry CH2ClCH2Cl/3 h/5°C;; | 100% |
| In 1,2-dichloro-ethane dry CH2ClCH2Cl/3 h/5°C;; | 100% |

| Conditions | Yield |
|---|---|
| at 300℃; for 3h; | 100% |

-
-
1633-05-2
strontium(II) carbonate

-
-
7664-93-9
sulfuric acid

-
-
7446-11-9
sulfur trioxide

-
-
11113-50-1
boric acid

| Conditions | Yield |
|---|---|
| at 180℃; for 24h; | 100% |


| Conditions | Yield |
|---|---|
| 995 °C in N2; part of a Mg-S-I water splitting cycle; | A 99% B 99% |
| copper(II) oxide In gas equil. react.; | |
| platinum In gas equil. react.; |

| Conditions | Yield |
|---|---|
| In sulfuric acid aq. H2SO4; (NH42PbCl6 and oleum filled in glass tube; torch-sealed under vac.; heated up to 250°C; maintained for 24 h, slowly cooled (1.8 K/h); ppt. collected by decantation of mother liqour under inert condns.; | 99% |


| Conditions | Yield |
|---|---|
| In liquid sulphur dioxide Ar; 20°C;; condensation of SO2; residue washed with FCCl3; elem. anal.;; | 96% |
-
-
7446-11-9
sulfur trioxide

-
-
873376-62-6
triethoxyantimony

-
-
80398-49-8
ethoxyantimony bis(ethyl sulfate)

| Conditions | Yield |
|---|---|
| In dichloromethane SO3 soln. was added to Sb compd. at -50°C (N2 or Ar); evapn., drying in vac.; elem. anal.; | 96% |
-
-
116-15-4
perfluoropropylene

-
-
7446-11-9
sulfur trioxide

-
-
773-15-9
1,2,2-trifluoro-2-hydroxy-1-trifluoromethylethanesulfonic acid sultone

| Conditions | Yield |
|---|---|
| 150°C, 5 h; | 94% |
| 150°C, 5 h; | 94% |
| 60°C; | 92% |

| Conditions | Yield |
|---|---|
| With sulfuric acid In water; benzene | 93% |
| With phosphoric acid; sulfuric acid In water; benzene | 87% |

| Conditions | Yield |
|---|---|
| fresh distilled SO3, 2.7 atm, below 80°C, 1 h; | 93% |
| fresh distilled SO3, 2.7 atm, below 80°C, 1 h; | 93% |
| fresh distilled SO3, 2.7 atm, below 80°C, 1 h; | 93% |

| Conditions | Yield |
|---|---|
| In dichloromethane SO3 soln. was added to Sb compd. at -50°C (N2 or Ar); elem. anal.; | 93% |
-
-
180070-46-6, 180070-53-5
((CH3)3Sn)C(C6H9)CH(Sn(CH3)3)

-
-
7446-11-9
sulfur trioxide

-
-
180071-29-8
(CH3)3SnSO3C(C6H9)CHSO3Sn(CH3)3

| Conditions | Yield |
|---|---|
| In dichloromethane absence of air and moisture; 2 equiv. SO2, stirring at -78°C for 4 h; evapn., distn.; elem. anal.; | 93% |

| Conditions | Yield |
|---|---|
| A 91% B n/a |

-
-
685-63-2
hexafluoro-1,3-butadiene

-
-
7446-11-9
sulfur trioxide

-
-
106-99-0
buta-1,3-diene

-
-
69740-49-4
4-trifluorovinyltrifluoro-1,2-oxathietane 2,2-dioxide

| Conditions | Yield |
|---|---|
| In liquid sulphur dioxide dropping SO3 into a soln. of CF2=CFCF=CF2 in SO2 at -10°C followed by stripping SO2 and butadiene; distn.; | 91% |
| In sulfur dioxide dropping SO3 into a soln. of CF2=CFCF=CF2 in SO2 at -10°C followed by stripping SO2 and butadiene; distn.; | 91% |


-
-
7446-11-9
sulfur trioxide

-
-
174715-71-0
(5,7,12,14-tetramethyldibenzo[b,i][1,4,8,11]tetraazacyclotetradecinate(2-))V(O2SO2)

| Conditions | Yield |
|---|---|
| In dichloromethane Ar-atmosphere; slow addn. of SO3 to V-complex at 0°C, stirring atroom temp. for 2 h; ether addn., collection (filtration), washing (CH2Cl2, ether); elem. anal.; | 91% |
-
-
7664-93-9
sulfuric acid

-
-
14154-42-8, 62905-77-5
phthalocyaninealuminum chloride

-
-
7446-11-9
sulfur trioxide

| Conditions | Yield |
|---|---|
| In sulfuric acid mixt. Al complex and oleum was stirred at room temp. for 16 h.; react. mixt. was cooled to ambient temp., poured onto ice, ppt. was filtered off, washed with 1% aq. HCl, water, dried in vac. at 90°C; | 91% |

-
-
79-38-9
Chlorotrifluoroethylene

-
-
7446-11-9
sulfur trioxide

-
-
1004-47-3
3-chlorotrifluoro-1,2-oxathietane 2,2-dioxide

| Conditions | Yield |
|---|---|
| at 70°C; | 90% |
-
-
7446-11-9
sulfur trioxide

-
-
7790-89-8
chlorine monofluoride

-
A
-
640723-20-2, 2699-79-8, 12769-73-2
fluorosulfonyl fluoride

| Conditions | Yield |
|---|---|
| byproducts: Cl2; from -196 °C to room temp., excess ClF; | A n/a B 90% |


| Conditions | Yield |
|---|---|
| 220-280 °C, 30 atm NH3, 30 min; | 90% |
Related products
Raw Materials
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View



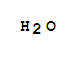

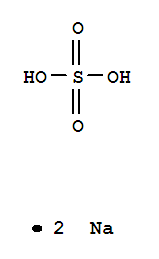

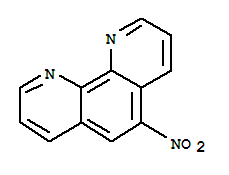




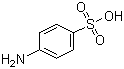


 O,
O, T+,
T+, C
C