-
Name
1,1-Diphenylethane
- EINECS
- CAS No. 612-00-0
- Article Data357
- CAS DataBase
- Density 0.979 g/cm3
- Solubility 5040 mg/L at 25ºC
- Melting Point -18 ºC
- Formula C14H14
- Boiling Point 272.6 °C at 760 mmHg
- Molecular Weight 182.265
- Flash Point 109.6 °C
- Transport Information
- Appearance clear low viscosity liquid
- Safety
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms Ethane,1,1-diphenyl- (6CI,8CI); Ethane, as-diphenyl- (3CI); 1,1-Diphenylethane;Methyldiphenylmethane; NSC 33534; Pabsol 300
- PSA 0.00000
- LogP 3.83840
Synthetic route

| Conditions | Yield |
|---|---|
| With C28H18Co(1-)*K(1+)*2C4H10O2; hydrogen In toluene at 60℃; under 7500.75 Torr; for 24h; Solvent; chemoselective reaction; | 100% |
| With water for 6h; Milling; Green chemistry; | 100% |
| With iPr-amboxCoCl2; hydrogen; sodium triethylborohydride In toluene at 0 - 20℃; Reagent/catalyst; Sealed tube; | 100% |

| Conditions | Yield |
|---|---|
| With iodine; hypophosphorous acid In acetic acid at 60℃; for 24h; | 100% |
| With phosphonic Acid; methanesulfonic acid; sodium iodide In water at 95℃; for 24h; Inert atmosphere; | 99.6% |
| With formic acid; 5%-palladium/activated carbon; ammonium formate In methanol; water at 80℃; for 0.666667h; | 95% |

| Conditions | Yield |
|---|---|
| With hydrogen; triethylamine; palladium on activated charcoal In methanol at 20℃; for 0.5h; | 100% |
| With Pd(0)/HAP; hydrogen; caesium carbonate In isopropyl alcohol at 60℃; under 7600.51 Torr; for 1h; | > 99 %Chromat. |
-
-
5433-78-3
diphenylmethyl p-tolyl sulfone

-
-
75-24-1
trimethylaluminum

-
-
612-00-0
1,1'-ethylidenebis-benzene

| Conditions | Yield |
|---|---|
| In dichloromethane; toluene at 25℃; for 2h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With silica-supported sulfuric acid at 60℃; for 1h; Friedel-Crafts type alkylation; | 99% |
| With silica sulfuric acid at 60℃; for 1h; |

| Conditions | Yield |
|---|---|
| With palladium dichloride In methanol at 40℃; for 12h; Inert atmosphere; Green chemistry; chemoselective reaction; | 99% |
-
-
60702-29-6
(1-(phenylsulfonyl)ethane-1,1-diyl)dibenzene

-
-
612-00-0
1,1'-ethylidenebis-benzene

| Conditions | Yield |
|---|---|
| With N1,N1,N12,N12-tetramethyl-7,8-dihydro-6H-dipyrido[1,2-a:2,1'-c][1,4]diazepine-2,12-diamine In N,N-dimethyl-formamide at 100℃; Inert atmosphere; | 98% |
| With tetraazaethylene-type neutral organic super-electron donor In N,N-dimethyl-formamide at 110℃; for 18h; | 97% |
| Stage #1: (1-(phenylsulfonyl)ethane-1,1-diyl)dibenzene With C3H6C9H10N4 In N,N-dimethyl-formamide at 110℃; Inert atmosphere; Stage #2: In water Inert atmosphere; | 97% |
| With C24H32N8Ni(2+)*2I(1-); sodium amalgam In N,N-dimethyl-formamide at 20℃; for 18h; Inert atmosphere; | 70% |
| With 4-(dimethylamino)-1-methylpyridinium-2-carboxylate In N,N-dimethyl-formamide at 150℃; for 24h; Schlenk technique; Inert atmosphere; | 15% |

| Conditions | Yield |
|---|---|
| With sodium hydride; lithium iodide In tetrahydrofuran; mineral oil at 85℃; for 7h; Reagent/catalyst; Inert atmosphere; Sealed tube; | 98% |
-
-
585-71-7, 38661-81-3
(1-bromoethyl)benzne

-
-
100-58-3
phenylmagnesium bromide

-
-
612-00-0
1,1'-ethylidenebis-benzene

| Conditions | Yield |
|---|---|
| With dichloro bis(acetonitrile) palladium(II); 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In tetrahydrofuran at 20℃; for 14h; Kumada-Corriu reaction; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With titanium tetrachloride In dichloromethane at 0℃; for 2h; | 95% |
-
-
292638-84-7
styrene

-
-
530-48-3
1,1-Diphenylethylene

-
A
-
612-00-0
1,1'-ethylidenebis-benzene

-
B
-
100-41-4
ethylbenzene

| Conditions | Yield |
|---|---|
| With C28H18Co(1-)*K(1+)*2C4H10O2; hydrogen at 20℃; under 1500.15 Torr; for 3h; | A 18% B 95% |

-
-
87057-91-8
5-benzhydryl-2,2,5-trimethyl-1,3-dioxane-4,6-dione

-
-
75-24-1
trimethylaluminum

-
-
612-00-0
1,1'-ethylidenebis-benzene

| Conditions | Yield |
|---|---|
| In n-heptane; dichloromethane at 20℃; for 1h; | 92% |

| Conditions | Yield |
|---|---|
| With nickel(II) iodide; potassium fluoride; 4,4'-dimethyl-2,2'-bipyridines; zinc In N,N-dimethyl acetamide at 25℃; for 16h; Inert atmosphere; Glovebox; Schlenk technique; | 92% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate; oxygen; (-)-sparteine; imidazole-carbene-derived Pd complex In isopropyl alcohol at 55℃; for 24h; | 91% |
| With potassium tert-butylate; oxygen; isopropyl alcohol; [Pd(SiPr)Cl2]2; (-)-sparteine at 55℃; for 24h; | 91% |
| With di-μ-chlorobis[chloro(N,N'-bis-(2,6-(diisopropyl)phenyl)imidazolidine-2-ylidene)palladium]; potassium tert-butylate; oxygen; isopropyl alcohol; (-)-sparteine at 55℃; for 24h; regioselective reaction; | 91% |

| Conditions | Yield |
|---|---|
| With methanol at 40℃; for 24h; Schlenk technique; Irradiation; Inert atmosphere; | 91% |
| With 10-phenyl-9-(2,4,6-trimethylphenyl)acridinium tetrafluoroborate; N-ethyl-N,N-diisopropylamine; diphenyldisulfane In 2,2,2-trifluoroethanol at 20℃; for 24h; Glovebox; Irradiation; Sealed tube; | 83% |
-
-
100865-93-8
O-diphenylmethyl 2,2,2-trichloroacetimidate

-
-
75-24-1
trimethylaluminum

-
A
-
612-00-0
1,1'-ethylidenebis-benzene

-
B
-
100865-92-7
N-benzhydryl-trichloroacetamide

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In dichloromethane at 20℃; for 0.5h; Solvent; Temperature; Reagent/catalyst; | A 91% B 8% |

-
-
100865-93-8
O-diphenylmethyl 2,2,2-trichloroacetimidate

-
-
75-24-1
trimethylaluminum

-
-
612-00-0
1,1'-ethylidenebis-benzene

| Conditions | Yield |
|---|---|
| Stage #1: trimethylaluminum With aluminum (III) chloride In hexane; dichloromethane for 0.0833333h; Inert atmosphere; Stage #2: O-diphenylmethyl 2,2,2-trichloroacetimidate In hexane; dichloromethane at 0 - 20℃; for 0.25h; Reagent/catalyst; Inert atmosphere; | 91% |

| Conditions | Yield |
|---|---|
| Stage #1: benzophenone; methyllithium In tetrahydrofuran at -50 - 0℃; for 2h; Inert atmosphere; Stage #2: With hexylsilane; tris(pentafluorophenyl)borate In dichloromethane at 20℃; for 1h; Inert atmosphere; chemoselective reaction; | 91% |
-
-
530-48-3
1,1-Diphenylethylene

-
A
-
612-00-0
1,1'-ethylidenebis-benzene

-
B
-
10496-82-9
2,2,3,3-tetraphenylbutane

| Conditions | Yield |
|---|---|
| With titanium(III) citrate; Tris buffer; tetra(n-butyl)ammonium hydroxide; vitamin B-12 In ethanol pH=8; | A 5% B 90% |
| With hydrogen In acetone at -78℃; under 2 Torr; for 0.5h; |
-
-
119-61-9
benzophenone

-
-
917-54-4
methyllithium

-
A
-
530-48-3
1,1-Diphenylethylene

-
B
-
612-00-0
1,1'-ethylidenebis-benzene

| Conditions | Yield |
|---|---|
| With <<(n-PrO)3WCl2>2> In tetrahydrofuran -78 deg C -> RT, 1 h, then reflux, 3 h; | A 90% B 90% |


| Conditions | Yield |
|---|---|
| With nickel(II) bromide dimethoxyethane; [Ir(dF(CF3)ppy)2(phen)]PF6; Bathocuproine; diisopropylamine; magnesium bromide In N,N-dimethyl-formamide at 20℃; for 24h; Inert atmosphere; Glovebox; Sealed tube; Irradiation; regioselective reaction; | 90% |
| With nickel(II) iodide; Bathocuproine; tetrabutylammomium bromide; zinc at 20℃; regioselective reaction; | 85% |

-
-
612-00-0
1,1'-ethylidenebis-benzene

| Conditions | Yield |
|---|---|
| With triethylsilane; C19H3BF14 In dichloromethane at 20℃; Inert atmosphere; Schlenk technique; | 89% |
-
-
599-67-7
1,1-diphenylethanol

-
A
-
530-48-3
1,1-Diphenylethylene

-
B
-
612-00-0
1,1'-ethylidenebis-benzene

-
C
-
947-40-0
1-chloro-1,1-diphenyl-ethane

| Conditions | Yield |
|---|---|
| With acetyl chloride In dichloromethane 1.) 0 deg C, 2.) room temperature, 1 h; Yield given; | A 88% B 43% C n/a |


| Conditions | Yield |
|---|---|
| With Pd(0)/HAP; hydrogen; caesium carbonate In isopropyl alcohol at 60℃; under 7600.51 Torr; for 28h; | 87% |
| Multi-step reaction with 2 steps 1: 12 percent / H2; Et3N / Pd/C / methanol / 0.5 h / 20 °C 2: 27 percent / H2; Et3N / Pd/C / methanol / 24 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 34 percent / H2; Et3N / Pd/C / methanol / 0.17 h / 20 °C 2: 27 percent / H2; Et3N / Pd/C / methanol / 24 h / 20 °C View Scheme |
-
-
530-48-3
1,1-Diphenylethylene

-
A
-
612-00-0
1,1'-ethylidenebis-benzene

-
B
-
19303-32-3
1-methyl-1,3,3-triphenylindane

| Conditions | Yield |
|---|---|
| With bromobenzene-d5; (di-p-tolylmethyl)triethylsilane; C24H20B(1-)*C20H29NSi*H(1+) at 100℃; under 3040.2 Torr; for 24h; Schlenk technique; Inert atmosphere; | A 14% B 86% |
| With diethyl ether; tris(pentafluorophenyl)borate; hydrogen In dichloromethane-d2 at 50℃; under 3040.2 Torr; for 48h; Catalytic behavior; Concentration; Pressure; Reagent/catalyst; Solvent; Temperature; Time; Glovebox; |
-
-
110-89-4
piperidine

-
-
530-48-3
1,1-Diphenylethylene

-
-
201230-82-2
carbon monoxide

-
A
-
2591-86-8
N-Formylpiperidine

-
B
-
612-00-0
1,1'-ethylidenebis-benzene

-
C
-
3540-95-2
fenpiprane

| Conditions | Yield |
|---|---|
| With [(bicyclo[2.2.1]hepta-2,5-diene)(1,4-bis(diphenylphosphino)butane)rhodium(I)] tetrafluoroborate; 1,1'-Binaphthalin-2,2'-diylbis(methylen)bis(diphenylphosphan); hydrogen In toluene at 125℃; for 60h; Reagent/catalyst; Autoclave; | A 5.6%Chromat. B 13.8%Chromat. C 85% |
| With [(bicyclo[2.2.1]hepta-2,5-diene)(1,4-bis(diphenylphosphino)butane)rhodium(I)] tetrafluoroborate; 1,1'-Binaphthalin-2,2'-diylbis(methylen)bis(diphenylphosphan); hydrogen In methanol at 125℃; for 60h; Reagent/catalyst; Autoclave; | A 22.1%Chromat. B 40%Chromat. C 48% |

-
-
530-48-3
1,1-Diphenylethylene

-
-
4559-70-0
Diphenylphosphine oxide

-
A
-
612-00-0
1,1'-ethylidenebis-benzene

-
B
-
13685-66-0
(2,2-Diphenylethyl)diphenylphosphine oxide

-
C
-
78375-70-9
1,1-diphenyl-2-diphenylphosphinylethene oxide

| Conditions | Yield |
|---|---|
| With pyridine; (difluoroboryl)dimethylglyoximatocobalt(II) bis(acetonitrile) In dichloromethane at 20℃; for 16h; Catalytic behavior; Reagent/catalyst; Solvent; Inert atmosphere; Irradiation; | A n/a B 9% C 85% |

-
-
530-48-3
1,1-Diphenylethylene

-
-
201230-82-2
carbon monoxide

-
A
-
612-00-0
1,1'-ethylidenebis-benzene

-
B
-
4279-81-6
3,3-diphenylpropanal

-
C
-
20017-67-8
3,3-diphenylpropan-1-ol

| Conditions | Yield |
|---|---|
| With hydrogen; di(rhodium)tetracarbonyl dichloride In toluene at 150℃; under 103430 Torr; for 2h; Product distribution; Mechanism; other catalysts, times, temp., solvents; | A 11.3% B 84.7% C 3.9% |

-
-
110-89-4
piperidine

-
-
530-48-3
1,1-Diphenylethylene

-
-
201230-82-2
carbon monoxide

-
A
-
612-00-0
1,1'-ethylidenebis-benzene

-
B
-
4279-81-6
3,3-diphenylpropanal

-
C
-
3540-95-2
fenpiprane

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate; 1,1'-Binaphthalin-2,2'-diylbis(methylen)bis(diphenylphosphan); hydrogen In toluene at 125℃; for 60h; Autoclave; | A 11.7%Chromat. B 6.7%Chromat. C 81% |
| With bis(1,5-cyclooctadiene)rhodium(I) tetrafluoroborate; hydrogen; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In toluene at 125℃; for 60h; Autoclave; | A 40.2%Chromat. B 22.4%Chromat. C 29% |

-
-
612-00-0
1,1'-ethylidenebis-benzene

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; water-d2 In methanol at 30℃; for 12h; | 99% |

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile); oxygen; nickel dibromide In chloroform at 75℃; for 48h; Reagent/catalyst; | A 88% B 10% |
| With 2,2'-azobis(isobutyronitrile); oxygen In acetonitrile at 75℃; for 72h; Solvent; Reagent/catalyst; | A 20% B 44% |
1,1-DIPHENYLETHANE Chemical Properties
1,1-Diphenylethane (612-00-0) is called for (1-Phenylethyl)benzene ; 1,1’-Ethylidenebis-benzen ; 1,1’-Ethylidenebis-benzene ; 1,1-Diphenylethane, as- ; 1,1'-Ethylidenebisbenzene ;Benzene,1,1’-ethylidenebis- ; Ethane, 1,1-diphenyl- (8CI) ; CID11918 ,and so on.
IUPAC Name: 1-Phenylethylbenzene
CAS: 612-00-0
Molecular Formula: C14H14
Molecular Weight: 182.26
Molecular structure: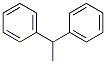
ACD/LogD (pH 5.5): 4.55
ACD/LogD (pH 7.4): 4.55
ACD/BCF (pH 5.5): 1703.58
ACD/BCF (pH 7.4): 1703.58
ACD/KOC (pH 5.5): 7154.96
ACD/KOC (pH 7.4): 7154.96
H bond acceptors: 0
H bond donors: 0
Freely Rotating Bonds: 2
Index of Refraction: 1.56
Molar Refractivity: 60.2 cm3
Molar Volume: 186 cm3
Polarizability: 23.86 10-24cm3
Surface Tension: 36.2 dyne/cm
Density: 0.979 g/cm3
Flash Point: 109.6 °C
Enthalpy of Vaporization: 49.04 kJ/mol
Boiling Point: 272.6 °C at 760 mmHg
Vapour Pressure: 0.01 mmHg at 25°C
1,1-DIPHENYLETHANE Uses
1,1-Diphenylethane (612-00-0) is commonly used as organic reagent.
1,1-DIPHENYLETHANE Specification
Removal in wastewater treatment of 1,1-Diphenylethane (612-00-0):
Total removal:48.27 percent
Total biodegradation:0.33 percent
Total sludge adsorption:34.19 percent
Total to Air:13.75 percent
(using 10000 hr Bio P,A,S)
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 6120-13-4
- 6120-31-6
- 61203-48-3
- 61203-71-2
- 61203-82-5
- 61203-83-6
- 61203-94-9
- 61203-99-4
- 61204-00-0
- 61204-01-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View