Dayang Chem (Hangzhou) Co.,Ltd.
As a leading manufacturer and supplier of chemicals in China, DayangChem not only supply popular chemicals, but also DayangChem's R&D center offer custom synthesis services. DayangChem can provide different quantities of custom synthesis ch
Cas:75-24-1
Min.Order:1 Kilogram
FOB Price: $3.0
Type:Lab/Research institutions
inquiryHebei yanxi chemical co.,LTD.
Hebei yanxi chemical co. LTD. has expanded a compositive entity from initially only as a small manufacturer. The company dedicated to the development, production and marketing of chemicals. After many years of efforts, we have established stable
Cas:75-24-1
Min.Order:1 Metric Ton
FOB Price: $1.0 / 2.0
Type:Manufacturers
inquiryHenan Allgreen Chemical Co.,Ltd
high quality Appearance:White or off-white Solid Storage:Sealed, dry, microtherm , avoid light and smell. Package:According to the demand of customer Application:Organic synthesis Transportation:by air or by sea Port:shanghai
Cas:75-24-1
Min.Order:1 Gram
Negotiable
Type:Manufacturers
inquiryAlity Chemical Corporation
The above product is Ality Chemical's strong item with best price, good quality and fast supply. Ality Chemical has been focusing on the research and production of this field for over 14 years. At the same time, we are always committed to providi
Chemwill Asia Co., Ltd.
Our main production base is located in Xuzhou industry park. We are certified both to the ISO 9001 and ISO 14001 Standards, have a safety management system in place.Our R&D team masters core technology for process-design of target building block
Cas:75-24-1
Min.Order:5 Kiloliter
FOB Price: $1.2 / 5.0
Type:Manufacturers
inquiryBaoji Guokang Healthchem co.,ltd
Our company has been in existence for 10 years since its establishment. We have our own unique team. The company integrates independent research and development, production and sales. We have established famous brands at home and abroad. At present
Cas:75-24-1
Min.Order:1 Kilogram
FOB Price: $6.0 / 10.0
Type:Trading Company
inquiryHenan Wentao Chemical Product Co., Ltd.
Henan Wentao Chemical Product Co.,Ltd is Located in Zhengzhou High-tech Development Zone with import and export license, We passed ISO 9001:2008 as well, Henan Wentao has developed more than 1000 compounds, which are widely used in the fields of prod
Hangzhou Keyingchem Co.,Ltd
Hangzhou KeyingChem Co., Ltd. exported this product to many countries and regions at best price. If you are looking for the material’s manufacturer or supplier in China, KeyingChem is your best choice. Pls contact with us freely for getting det
Cas:75-24-1
Min.Order:0 Metric Ton
Negotiable
Type:Lab/Research institutions
inquiryHangzhou J&H Chemical Co., Ltd.
J&H CHEM R&D center can offer custom synthesis according to the contract research and development services for the fine chemicals, pharmaceutical, biotechnique and some of the other chemicals. J&H CHEM has some Manufacturing base in Jia
Shanghai Massive Chemical Technology Co., Ltd.
Massive Chemical is certified with ISO9001 and ISO14001 manufacturer for this product. We will offer all documents as requirement for the materials which includes, Certificate of Analysis, Material Safety Data Sheet, and Method of Analysis and
Cas:75-24-1
Min.Order:1 Gram
FOB Price: $1.0
Type:Lab/Research institutions
inquiryZibo Hangyu Biotechnology Development Co., Ltd
Zibo Hangyu Biotechnology Development Co., Ltd is a leading manufacturer and supplier of chemicals in China. We develop produce and distribute high quality pharmaceuticals, intermediates, special chemicals and OLED intermediates and other fine chemi
Cas:75-24-1
Min.Order:10 Gram
FOB Price: $100.0
Type:Lab/Research institutions
inquiryHANGZHOU YUNUO CHEMICAL CO.,LTD
Superior quality Appearance:clear colorless solution Storage:Stored in cool, dry and ventilation place; Away from fire and heat Package:1kg/bag, 1kg/drum or 25kg/drum or as per your request. Application:Used as catalysts for olefin polymerization,
Henan Tianfu Chemical Co., Ltd.
Trimethylaluminium Chemical Properties Melting point 15 °C Boiling point 126 °C density 0.81 g/mL at 25 °C Fp 40 °F storage temp. 0-6°C solubility Soluble in arom
Cas:75-24-1
Min.Order:1 Metric Ton
FOB Price: $20.0
Type:Lab/Research institutions
inquiryHunan chemfish Pharmaceutical co.,Ltd
Appearance:95%+ Package:R&D,Pilot run Transportation:per client require Port:Express ,Air, Sea
SAGECHEM LIMITED
SAGECHEM is a chemical R&D, manufacturing and distribution company in China since 2009, including pharmaceutical intermediates, agrochemical, dyestuff intermediates, organosilicone, API and etc. We also offer a full range of services in custom synthe
Kono Chem Co.,Ltd
high purity lowest priceAppearance:solid or liquid Storage:in sealed air resistant place Package:drum and bag Application:for pharma use Transportation:by sea or air Port:Beijing or Guangzhou
Hangzhou Dingyan Chem Co., Ltd
R & D enterprises have their own stock in stockAppearance:To be subject to the object Package:Customized Application:pharmaceutical intermediates Transportation:Air Port:Shanghai;Guangzhou
JINHUA HUAYI CHEMICAL CO., LTD.
Jinhua huayi chemical co., ltd. is dedicated to the development, production and marketing of chemicals. On the basis of equality and mutual benefit, and under the principle of customer first, credit first, quality first, we are ready to join hands
Hunan Russell Chemicals Technology Co.,Ltd
low price and high purity Package:as customer required Application:ALD related industries,medicine and electronic materials
Zhuozhou Wenxi import and Export Co., Ltd
Product Description Description & Specification Category Pharmaceutical Raw Materials, Fine Chemicals, Bulk drug Standard Medical standard
Cas:75-24-1
Min.Order:1 Kilogram
FOB Price: $112.0
Type:Trading Company
inquiryAntimex Chemical Limied
Ansciep Chemical is a professional enterprise manufacturing and distributing fine chemicals and speciality chemicals. We have been dedicated to heterocycle compounds and phenyl rings for tens of years. This is our mature product for export. Our quali
Xiamen AmoyChem Co.,Ltd
Amoychem is committed to providing the top-quality chemical products and services Internationally. We offer our customers with friendly, professional service and reliable, high performance products that have been manufactured according to the accredi
Hubei Langyou International Trading Co., Ltd
75-24-1 Trimethylaluminium Application:75-24-1 Trimethylaluminium
Hangzhou Fandachem Co.,Ltd
Trimethylaluminium cas 75-24-1Appearance:white crystalline powder Storage:Store in dry, dark and ventilated place Package:25KG drum Application:intermediate Transportation:by air, by sea, by express
Hangzhou ZeErRui Chemical Co., Ltd.
Hangzhou ZeErRui Chemical Co., Ltd. located in Lingang industrial areas, our plant covers an area of 6000 square meters.ZeErRui dedicated to the development, production and marketing of chemicals. We have earned ourselves a good reputation at home an
Cas:75-24-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryHunan Longxianng Runhui Trading Co.,Ltd
75-24-1 TrimethylaluminiumAppearance:powder Storage:room tempurature Package:As required Application:medical Transportation:By express (Door to door) such as FEDEX, DHL, EMS for small amount. By air(airport to airport) or by sea LCL/FCL for large amo
Cas:75-24-1
Min.Order:0
Negotiable
Type:Lab/Research institutions
inquiryBeantown Chemical
in stock Application:75-24-1
Cas:75-24-1
Min.Order:0
Negotiable
Type:Trading Company
inquiryAmadis Chemical Co., Ltd.
1.Professional synthesis laboratory and production base. 2.Strong synthesis team and service team. 3.Professional data management system. 4.We provide the professional test date and product information ,ex. HNMR ,CNMR,FNMR, HPLC/G
Cas:75-24-1
Min.Order:10 Milligram
Negotiable
Type:Lab/Research institutions
inquirySynthetic route

| Conditions | Yield |
|---|---|
| With methylmagnesium bromide In not given react. of AlCl3 with MeMgBr; | 99% |

| Conditions | Yield |
|---|---|
| With methylene chloride; sodium at 40 - 50℃; under 375.038 Torr; Inert atmosphere; Flow reactor; | A 93.7% B 96.8% |

-
-
97-93-8
triethylaluminum

-
-
7429-90-5
aluminium

-
A
-
75-24-1
trimethylaluminum

-
B
-
2040-00-8
diethylaluminum iodide

| Conditions | Yield |
|---|---|
| With iodine; methyl iodide In ethanol Sonication; N2; CH3I, I2 and Al introduced in a condenser with C2H5OH at -20°C, ultrasonic acceleration at room temp. for 2 h, Et3Al (ratio MeI/Et3Al = 1.50) dropped into soln. within 10 min, sonication for 30 min; distilled (vac.); | A 86% B 95% |


-
-
66415-27-8
2,4,6-tri-tert-butylbenzoic acid

-
B
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In hexane; toluene byproducts: CH4; in a glovebox, Nd-contg. compd. (0.50 mmol) was dissolved in hexane, anda toluene suspn. of carboxylic acid (0.50 mmol) was slowly added; stirr ing at ambient temp. for 18 h; the solvent was removed in vac.; extn. in toluene; the complex was washed with hexane and dried for several hours; elem. anal.; | A 94% B n/a |

-
-
97-93-8
triethylaluminum

-
-
7429-90-5
aluminium

-
A
-
75-24-1
trimethylaluminum

-
B
-
2938-72-9
dimethyl aluminum iodide

-
C
-
2938-73-0
ethyl aluminium iodide

| Conditions | Yield |
|---|---|
| With iodine; methyl iodide In ethanol Sonication; N2, CH3I, I2 and Al introduced in a condenser with C2H5OH at -20°C, ultrasonic acceleration at room temp. for 2 h, Et3Al (ratio MeI/Et3Al = 12.00) dropped into soln. within 10 min, sonicated for 30 min; distilled (vac.); | A 62.5% B 22.5% C 93% |


-
-
66415-27-8
2,4,6-tri-tert-butylbenzoic acid

-
B
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In hexane; toluene byproducts: CH4; in a glovebox, La-contg. compd. (0.50 mmol) was dissolved in hexane, anda toluene suspn. of carboxylic acid (0.50 mmol) was slowly added; stirr ing at ambient temp. for 18 h; the solvent was removed in vac.; extn. in toluene; the complex was washed with hexane and dried for several hours; elem. anal.; | A 93% B n/a |


| Conditions | Yield |
|---|---|
| In hexane byproducts: CH4; Schlenk technique; diene (1 equiv.) in hexane was added with stirring tosoln. of La complex in hexane; mixt. was stirred for 5 min; solvent removed (vac.); dried (vac.); elem. anal.; | A 93% B n/a |


| Conditions | Yield |
|---|---|
| In hexane byproducts: CH4; Schlenk technique; diene (1 equiv.) in hexane was added with stirring tosoln. of Nd complex in hexane; mixt. was stirred for 5 min; solvent removed (vac.); dried (vac.); elem. anal.; | A 91% B n/a |


| Conditions | Yield |
|---|---|
| In hexane byproducts: CH4; Schlenk technique; diene (1 equiv.) in hexane was added with stirring tosoln. of Lu complex in hexane; mixt. was stirred for 5 min; solvent removed (vac.); dried (vac.); elem. anal.; | A 91% B n/a |


-
-
66415-27-8
2,4,6-tri-tert-butylbenzoic acid

-
B
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In hexane; toluene byproducts: CH4; in a glovebox, Y-contg. compd. (0.50 mmol) was dissolved in hexane, and a toluene suspn. of carboxylic acid (0.50 mmol) was slowly added; stirring at ambient temp. for 18 h; the solvent was removed in vac.; extn. in toluene; the complex was washed with hexane and dried for several hours; elem. anal.; | A 87% B n/a |


| Conditions | Yield |
|---|---|
| With sodium; copper In Hexadecane at 20 - 130℃; under 825.083 - 862.586 Torr; for 2.2h; Inert atmosphere; Flow reactor; | 87% |
| With sodium In melt; 1,3,5-trimethyl-benzene at 100 - 120℃; under 7.50075 Torr; for 6h; Inert atmosphere; | 69.5 g |

| Conditions | Yield |
|---|---|
| With sodium; gold In Hexadecane at 20 - 130℃; under 825.083 - 862.586 Torr; for 2.2h; Inert atmosphere; Flow reactor; | 87% |
| With sodium In melt; 1,3,5-trimethyl-benzene at 80 - 120℃; under 7.50075 Torr; for 8h; Inert atmosphere; | 68.4 g |

| Conditions | Yield |
|---|---|
| Stage #1: methyl iodide With magnesium In diethyl ether at 15℃; for 1h; Inert atmosphere; Stage #2: methylaluminum sesquichloride In pentane at 20℃; for 4h; Inert atmosphere; | 87% |

| Conditions | Yield |
|---|---|
| With silver; sodium In Hexadecane at 20 - 130℃; under 825.083 - 862.586 Torr; for 2.2h; Temperature; Inert atmosphere; Flow reactor; | 87% |

| Conditions | Yield |
|---|---|
| In hexane byproducts: CH4; Schlenk technique; diene (1 equiv.) in hexane was added with stirring tosoln. of Y complex in hexane; mixt. was stirred for 5 min; solvent removed (vac.); dried (vac.); elem. anal.; | A 86% B n/a |


-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In neat (no solvent) metal complex decomposed at 120-140°C for 2-3 h; | 86% |

| Conditions | Yield |
|---|---|
| With iodine; methyl iodide In ethanol Sonication; N2, CH3I, I2 and Al introduced in a condenser with C2H5OH at -20°C, ultrasonic acceleration at room temp. for 2 h, Et3Al (ratio MeI/Et3Al = 0.75) dropped into soln. within 10 min, sonicated for 30 min; distilled (vac.); | 85% |

| Conditions | Yield |
|---|---|
| Stage #1: methyl bromide With magnesium In tetrahydrofuran at 60℃; for 4h; Inert atmosphere; Stage #2: methylaluminum sesquichloride In hexane at 40℃; for 6h; Inert atmosphere; | 85% |

-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In neat (no solvent) metal complex decomposed at 120-130°C for 2-3 h; | 84% |

| Conditions | Yield |
|---|---|
| Stage #1: methyl iodide With magnesium In diethyl ether at 15℃; for 1h; Inert atmosphere; Stage #2: aluminium(III) iodide In pentane at 30℃; for 4h; Inert atmosphere; | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: methylene chloride With magnesium at 80℃; for 6h; Inert atmosphere; Stage #2: methylaluminum sesquichloride at 60℃; for 8h; Inert atmosphere; | 81% |

-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In neat (no solvent) metal complex decomposed at 120-140°C for 2-3 h; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: methyl bromide With magnesium In tetrahydrofuran at 40℃; for 3h; Inert atmosphere; Stage #2: aluminum tri-bromide In hexane at 40℃; for 7h; Inert atmosphere; | 79% |

-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| With sodium In mineral oil at 120 - 140℃; under 19.502 Torr; for 5.5h; Inert atmosphere; | 76.7% |
-
-
97-93-8
triethylaluminum

-
-
7429-90-5
aluminium

-
A
-
75-24-1
trimethylaluminum

-
B
-
2938-72-9
dimethyl aluminum iodide

| Conditions | Yield |
|---|---|
| With iodine; methyl iodide In ethanol Sonication; N2, CH3I, I2 and Al introduced in a condenser with C2H5OH at -20°C, ultrasonic acceleration at room temp. for 2 h, Et3Al (ratio MeI/Et3Al = 3.00) dropped into soln. within 10 min, sonicated for 30 min; distilled (vac.); | A 76% B 11.2% |
| With iodine; methyl iodide In ethanol Sonication; N2, CH3I, I2 and Al introduced in a condenser with C2H5OH at -20°C, ultrasonic acceleration at room temp. for 2 h, Et3Al (ratio MeI/Et3Al = 6.00) dropped into soln. within 10 min, sonicated for 30 min; distilled (vac.); | A 65% B 15.7% |


| Conditions | Yield |
|---|---|
| Stage #1: methylene chloride With magnesium In tetrahydrofuran at 60℃; for 6h; Inert atmosphere; Stage #2: aluminum (III) chloride In hexane at 50℃; for 11h; Inert atmosphere; | 76% |
-
-
6130-98-9
2,2,7,7-tetramethyl-octa-3,5-diyne

-
-
865-37-2
dimethylaluminum hydride

-
B
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In neat (no solvent) under Ar atm. excess Me2AlH and di(tert-butyl)butadiyne were stirred at 100°C for 1.5 h; react. mixt. was cooled to room temp., volatiles were removed in vacuo, residue was dissolved in n-pentane and cooled to -15°C; | A 70% B n/a |


| Conditions | Yield |
|---|---|
| Stage #1: dimethylaluminum chloride With pyridine; iodine In dodecane at 40℃; for 1.5h; Autoclave; Stage #2: methylene chloride In dodecane at 130℃; for 24h; Temperature; Reagent/catalyst; | 70% |


| Conditions | Yield |
|---|---|
| In hexane; toluene byproducts: CH4; in a glovebox, Nd-contg. compd. (0.40 mmol) was dissolved in hexane, anda toluene suspn. of carboxylic acid (0.40 mmol) was slowly added; stirr ing at ambient temp. for 18 h; the solvent was removed in vac.; extn. in toluene; the complex was washed with hexane and dried for several hours; purifn. by crystn.; elem. anal.; | A 64% B n/a |

-
-
58435-00-0
1-chloro-4-trimethylsilyl but-3-yne

-
-
75-24-1
trimethylaluminum

-
-
86994-12-9
2-methyl-1-(trimethylsilyl)cyclobutene

| Conditions | Yield |
|---|---|
| With zirconocene dichloride In dichloromethane at 25℃; for 24h; | 100% |
| With zirconocene dichloride In dichloromethane at 25℃; for 24h; | 95 % Chromat. |
-
-
41423-29-4
4-iodo-1-butynyltrimethylsilane

-
-
75-24-1
trimethylaluminum

-
-
86994-12-9
2-methyl-1-(trimethylsilyl)cyclobutene

| Conditions | Yield |
|---|---|
| With zirconocene dichloride In dichloromethane at 25℃; for 24h; | 100% |
| With zirconocene dichloride In dichloromethane at 25℃; for 24h; | 85% |
-
-
75-24-1
trimethylaluminum

-
-
156479-95-7
(S)-2-(1,3-Dioxo-1,3-dihydro-isoindol-2-yl)-6-oxo-hexanoic acid methyl ester

| Conditions | Yield |
|---|---|
| In hexane; dichloromethane at 0℃; | 100% |
-
-
625-35-4, 3488-22-0, 10487-71-5
trans-chrotonyl chloride

-
-
75-24-1
trimethylaluminum

-
-
3102-33-8
trans-3-penten-2-one

| Conditions | Yield |
|---|---|
| With aluminium trichloride In dichloromethane for 2h; Ambient temperature; | 100% |
| With aluminium trichloride 1.) CH2Cl2, 1 h, 2.) CH2Cl2, room temperature, 2 h; other reagents: GaCl3, AlF3; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With zirconocene dichloride In hexane; 1,2-dichloro-ethane at 20℃; for 22h; Addition; | 100% |
-
-
75-24-1
trimethylaluminum

-
-
286958-06-3
(R)-1,4-Dioxa-spiro[4.5]decane-2-carboxylic acid (2S,3R)-2-(2-benzyloxy-ethyl)-tetrahydro-pyran-3-yl ester

-
-
286958-07-4
(R)-2-{1-[(2S,3R)-2-(2-Benzyloxy-ethyl)-tetrahydro-pyran-3-yloxy]-vinyl}-1,4-dioxa-spiro[4.5]decane

| Conditions | Yield |
|---|---|
| Stage #1: trimethylaluminum With bis(cyclopentadienyl)titanium dichloride In toluene at 23℃; for 72h; complex formation; Stage #2: (R)-1,4-Dioxa-spiro[4.5]decane-2-carboxylic acid (2S,3R)-2-(2-benzyloxy-ethyl)-tetrahydro-pyran-3-yl ester In tetrahydrofuran; toluene at 0 - 23℃; olefination; | 100% |

| Conditions | Yield |
|---|---|
| With titanium(IV) isopropylate; (R,S)-2-OH-3,5-Cl2-C6H2-SO2-NH-CH(CH2Ph)-CH(Ph)OH In tetrahydrofuran at 0℃; for 12h; | 100% |
| In tetrahydrofuran; hexane at -20℃; for 3h; Product distribution / selectivity; | 60% |
-
-
75-24-1
trimethylaluminum

-
-
498554-04-4
4,6-O-benzylidene-2,3-bis-O-(tert-butyldimethylsilyl)-D-glucono-1,5-lactone

-
-
498554-05-5
(1R,3R,6R,9R,10S)-9,10-bis(tert-butyldimethylsilyloxy)-8-methylene-3-phenyl-2,4,7-trioxabicyclo[4.4.0]decane

| Conditions | Yield |
|---|---|
| Stage #1: trimethylaluminum With titanocene dichloride In toluene at 23℃; for 72h; Stage #2: 4,6-O-benzylidene-2,3-bis-O-(tert-butyldimethylsilyl)-D-glucono-1,5-lactone In tetrahydrofuran; toluene at 0 - 24℃; Tebbe olefination; | 100% |
-
-
75-24-1
trimethylaluminum

-
-
107-19-7
propargyl alcohol

-
-
80434-59-9
(1R,2S,5R)-2-isopropyl-5-methylcyclohexanecarboxaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: trimethylaluminum With bis(cyclopentadienyl)dihydrozirconium In dichloromethane for 0.166667h; Stage #2: propargyl alcohol In dichloromethane at 20℃; for 12h; Stage #3: (1R,2S,5R)-2-isopropyl-5-methylcyclohexanecarbaldehyde In tetrahydrofuran; dichloromethane at -78 - 20℃; for 12h; | 100% |
| Stage #1: trimethylaluminum With zirconocene dichloride In dichloromethane at 20℃; for 0.166667h; Stage #2: propargyl alcohol In dichloromethane at 0 - 20℃; Stage #3: (1R,2S,5R)-2-isopropyl-5-methylcyclohexanecarbaldehyde In tetrahydrofuran; dichloromethane at -70 - 20℃; |

-
-
17122-96-2
1,8-dichloro-9-methylanthracene

-
-
75-24-1
trimethylaluminum

-
-
63335-10-4
1,8,9-trimethylanthracene

| Conditions | Yield |
|---|---|
| With 1,3-bis[(diphenylphosphino)propane]dichloronickel(II) In 1,2-dimethoxyethane; hexane for 7h; Heating; | 100% |
-
-
76527-25-8
3,3-diphenyloxirane-2-carboxylic acid methyl ester

-
-
75-24-1
trimethylaluminum

-
-
178306-50-8
(+/-)-methyl 2-hydroxy-3,3-diphenylbutanoate

| Conditions | Yield |
|---|---|
| In cyclohexane; toluene at 10 - 15℃; for 0.5h; Product distribution / selectivity; | 100% |
| In cyclohexane at 10℃; Product distribution / selectivity; | 96% |
| In n-heptane; toluene at -10℃; for 0.5h; Product distribution / selectivity; | 82% |
| In toluene at -10 - 10℃; Product distribution / selectivity; | 82% |
-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| With benzoic acid In toluene at 26 - 80℃; for 9.5h; | 100% |
-
-
25423-56-7
1,4,7,10-tetrathiacyclododecane

-
-
75-24-1
trimethylaluminum

-
-
112440-15-0
trimethylaluminum-thiacrown ether {12}aneS4

| Conditions | Yield |
|---|---|
| In toluene addn. of Me3Al to {12}aneS4 in abs. toluene , heated at 60°C for several h; solvent reduced in vacuo, cooling; | 100% |
-
-
1271-19-8
bis(cyclopentadienyl)titanium dichloride

-
-
75-24-1
trimethylaluminum

-
-
1278-83-7
bis(cyclopentadienyl)methyltitanium(IV) chloride

| Conditions | Yield |
|---|---|
| With tetrahydrofuran In benzene-d6; toluene N2 or Ar-atmosphere; stirring (22°C, 1 h), THF addn.; detd. by NMR spectroscopy; | 100% |
-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In diethyl ether; toluene absence of air and moisture; dropwise addn. of AlMe3 in toluene to Mg(N(C3H7)2)2 in Et2O (molar ratio = 1 : 1, room temp., stirring); after 12h removal of solvent (vac.), recrystn. (hexane), sublimation (60°C); elem. anal.; | A <5 B 100% |


| Conditions | Yield |
|---|---|
| byproducts: CH4; not isolated, detected by NMR; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| byproducts: CH4; not isolated, detected by NMR; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| In dichloromethane Ar-atmosphere; addn. of Al-compd. (0°C); gradual warming to room temp. (2 h); evapn.; | 100% |
-
-
912272-65-2
(CH3)3SiNC(C6H5)CH2C(C6H5)NSi(CH3)3

-
-
75-24-1
trimethylaluminum

-
-
226717-58-4
[AlMe2(CH(C(Ph)NSiMe3)2)]

| Conditions | Yield |
|---|---|
| In hexane; pentane addn. of Me3Al in hexane to ligand in pentane at -78°C; accordingto P. B. Hitchcock, M. F. Lappert and D.-S Liu, J. Chem. Soc., Chem. Co mmun., 1994, 1699; elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In hexane | 100% |
| In hexane; toluene B(C6F5)3 and AlMe3 in 1:3 toluene/hexanes mixt.; | 99% |
| In hexane; toluene (inert atm.); reaction of borane deriv. with trimethylaluminium in hexane/toluene (3:1); | 99% |


| Conditions | Yield |
|---|---|
| In hexane (inert atmosphere); -30°C slowly to room temp., stirring (2 h); evapn. to dryness, washing (hexane); | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) under N2 atm. react. Me3Al and Et4Sb2; elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
-
-
75-24-1
trimethylaluminum

-
-
32318-29-9
tris(diethylamino)difluorophosphorane

-
-
222190-83-2
[FP(N(C2H5)2)3](1+)*[(CH3)3AlF](1-)=[FP(N(C2H5)2)3][(CH3)3AlF]

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Ar atm.; equimolar amts., stirring (20°C, 24 h); evapn. (vac.); elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
| In toluene dry Ar atm.; 3 equiv. of AlMe3, cooling (-15°C), heating (room temp., stirring), refluxing (1,5 h, 70-80°C); concn., standing (5 to -5 degree.C, several days), washing (toluene), drying (vac.); elem. anal.; | 54% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
| In toluene dry Ar atm.; 3 equiv. of AlMe3, cooling (-15°C), heating (room temp., stirring), refluxing (1,5 h, 70-80°C); concn., standing (5 to -5 degree.C, several days), washing (toluene), drying (vac.); elem. anal.; | 57% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
| In toluene dry Ar atm.; 3 equiv. of AlMe3, cooling (-15°C), heating (room temp., stirring), refluxing (1,5 h, 70-80°C); concn., standing (5 to -5 degree.C, several days), washing (toluene), drying (vac.); elem. anal.; | 58% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) substances mixted in glove box, N2; detd. by elem. anal., NMR; | 100% |
Related products
Downstream Products
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View



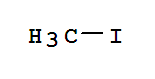


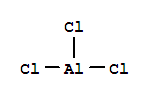







 F,
F, C,
C, N
N