-
Name
Trimethylaluminium
- EINECS 200-853-0
- CAS No. 75-24-1
- Article Data55
- CAS DataBase
- Density 0.81 g/mL at 25 °C
- Solubility reacts with water
- Melting Point 15 °C
- Formula C3H9Al
- Boiling Point 126 °C
- Molecular Weight 72.086
- Flash Point 40 °F
- Transport Information UN 3399 4.3/PG 1
- Appearance clear colorless solution
- Safety 26-36/37/39-45-61-62-6A-43A-24/25-16-43-60-33-25-24
- Risk Codes 11-14-17-34-50/53-65-67-14/15-63-48/20-51/53-20-62
-
Molecular Structure
-
Hazard Symbols
 F,
F, C,
C, N
N
- Synonyms Alane,trimethyl-;Trimethylalane;Trimethylaluminum (Al(CH3)3);
- PSA 0.00000
- LogP 1.75140
Synthetic route

| Conditions | Yield |
|---|---|
| With methylmagnesium bromide In not given react. of AlCl3 with MeMgBr; | 99% |

| Conditions | Yield |
|---|---|
| With methylene chloride; sodium at 40 - 50℃; under 375.038 Torr; Inert atmosphere; Flow reactor; | A 93.7% B 96.8% |

-
-
97-93-8
triethylaluminum

-
-
7429-90-5
aluminium

-
A
-
75-24-1
trimethylaluminum

-
B
-
2040-00-8
diethylaluminum iodide

| Conditions | Yield |
|---|---|
| With iodine; methyl iodide In ethanol Sonication; N2; CH3I, I2 and Al introduced in a condenser with C2H5OH at -20°C, ultrasonic acceleration at room temp. for 2 h, Et3Al (ratio MeI/Et3Al = 1.50) dropped into soln. within 10 min, sonication for 30 min; distilled (vac.); | A 86% B 95% |


-
-
66415-27-8
2,4,6-tri-tert-butylbenzoic acid

-
B
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In hexane; toluene byproducts: CH4; in a glovebox, Nd-contg. compd. (0.50 mmol) was dissolved in hexane, anda toluene suspn. of carboxylic acid (0.50 mmol) was slowly added; stirr ing at ambient temp. for 18 h; the solvent was removed in vac.; extn. in toluene; the complex was washed with hexane and dried for several hours; elem. anal.; | A 94% B n/a |

-
-
97-93-8
triethylaluminum

-
-
7429-90-5
aluminium

-
A
-
75-24-1
trimethylaluminum

-
B
-
2938-72-9
dimethyl aluminum iodide

-
C
-
2938-73-0
ethyl aluminium iodide

| Conditions | Yield |
|---|---|
| With iodine; methyl iodide In ethanol Sonication; N2, CH3I, I2 and Al introduced in a condenser with C2H5OH at -20°C, ultrasonic acceleration at room temp. for 2 h, Et3Al (ratio MeI/Et3Al = 12.00) dropped into soln. within 10 min, sonicated for 30 min; distilled (vac.); | A 62.5% B 22.5% C 93% |


-
-
66415-27-8
2,4,6-tri-tert-butylbenzoic acid

-
B
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In hexane; toluene byproducts: CH4; in a glovebox, La-contg. compd. (0.50 mmol) was dissolved in hexane, anda toluene suspn. of carboxylic acid (0.50 mmol) was slowly added; stirr ing at ambient temp. for 18 h; the solvent was removed in vac.; extn. in toluene; the complex was washed with hexane and dried for several hours; elem. anal.; | A 93% B n/a |


| Conditions | Yield |
|---|---|
| In hexane byproducts: CH4; Schlenk technique; diene (1 equiv.) in hexane was added with stirring tosoln. of La complex in hexane; mixt. was stirred for 5 min; solvent removed (vac.); dried (vac.); elem. anal.; | A 93% B n/a |


| Conditions | Yield |
|---|---|
| In hexane byproducts: CH4; Schlenk technique; diene (1 equiv.) in hexane was added with stirring tosoln. of Nd complex in hexane; mixt. was stirred for 5 min; solvent removed (vac.); dried (vac.); elem. anal.; | A 91% B n/a |


| Conditions | Yield |
|---|---|
| In hexane byproducts: CH4; Schlenk technique; diene (1 equiv.) in hexane was added with stirring tosoln. of Lu complex in hexane; mixt. was stirred for 5 min; solvent removed (vac.); dried (vac.); elem. anal.; | A 91% B n/a |


-
-
66415-27-8
2,4,6-tri-tert-butylbenzoic acid

-
B
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In hexane; toluene byproducts: CH4; in a glovebox, Y-contg. compd. (0.50 mmol) was dissolved in hexane, and a toluene suspn. of carboxylic acid (0.50 mmol) was slowly added; stirring at ambient temp. for 18 h; the solvent was removed in vac.; extn. in toluene; the complex was washed with hexane and dried for several hours; elem. anal.; | A 87% B n/a |


| Conditions | Yield |
|---|---|
| With sodium; copper In Hexadecane at 20 - 130℃; under 825.083 - 862.586 Torr; for 2.2h; Inert atmosphere; Flow reactor; | 87% |
| With sodium In melt; 1,3,5-trimethyl-benzene at 100 - 120℃; under 7.50075 Torr; for 6h; Inert atmosphere; | 69.5 g |

| Conditions | Yield |
|---|---|
| With sodium; gold In Hexadecane at 20 - 130℃; under 825.083 - 862.586 Torr; for 2.2h; Inert atmosphere; Flow reactor; | 87% |
| With sodium In melt; 1,3,5-trimethyl-benzene at 80 - 120℃; under 7.50075 Torr; for 8h; Inert atmosphere; | 68.4 g |

| Conditions | Yield |
|---|---|
| Stage #1: methyl iodide With magnesium In diethyl ether at 15℃; for 1h; Inert atmosphere; Stage #2: methylaluminum sesquichloride In pentane at 20℃; for 4h; Inert atmosphere; | 87% |

| Conditions | Yield |
|---|---|
| With silver; sodium In Hexadecane at 20 - 130℃; under 825.083 - 862.586 Torr; for 2.2h; Temperature; Inert atmosphere; Flow reactor; | 87% |

| Conditions | Yield |
|---|---|
| In hexane byproducts: CH4; Schlenk technique; diene (1 equiv.) in hexane was added with stirring tosoln. of Y complex in hexane; mixt. was stirred for 5 min; solvent removed (vac.); dried (vac.); elem. anal.; | A 86% B n/a |


-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In neat (no solvent) metal complex decomposed at 120-140°C for 2-3 h; | 86% |

| Conditions | Yield |
|---|---|
| With iodine; methyl iodide In ethanol Sonication; N2, CH3I, I2 and Al introduced in a condenser with C2H5OH at -20°C, ultrasonic acceleration at room temp. for 2 h, Et3Al (ratio MeI/Et3Al = 0.75) dropped into soln. within 10 min, sonicated for 30 min; distilled (vac.); | 85% |

| Conditions | Yield |
|---|---|
| Stage #1: methyl bromide With magnesium In tetrahydrofuran at 60℃; for 4h; Inert atmosphere; Stage #2: methylaluminum sesquichloride In hexane at 40℃; for 6h; Inert atmosphere; | 85% |

-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In neat (no solvent) metal complex decomposed at 120-130°C for 2-3 h; | 84% |

| Conditions | Yield |
|---|---|
| Stage #1: methyl iodide With magnesium In diethyl ether at 15℃; for 1h; Inert atmosphere; Stage #2: aluminium(III) iodide In pentane at 30℃; for 4h; Inert atmosphere; | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: methylene chloride With magnesium at 80℃; for 6h; Inert atmosphere; Stage #2: methylaluminum sesquichloride at 60℃; for 8h; Inert atmosphere; | 81% |

-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In neat (no solvent) metal complex decomposed at 120-140°C for 2-3 h; | 80% |

| Conditions | Yield |
|---|---|
| Stage #1: methyl bromide With magnesium In tetrahydrofuran at 40℃; for 3h; Inert atmosphere; Stage #2: aluminum tri-bromide In hexane at 40℃; for 7h; Inert atmosphere; | 79% |

-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| With sodium In mineral oil at 120 - 140℃; under 19.502 Torr; for 5.5h; Inert atmosphere; | 76.7% |
-
-
97-93-8
triethylaluminum

-
-
7429-90-5
aluminium

-
A
-
75-24-1
trimethylaluminum

-
B
-
2938-72-9
dimethyl aluminum iodide

| Conditions | Yield |
|---|---|
| With iodine; methyl iodide In ethanol Sonication; N2, CH3I, I2 and Al introduced in a condenser with C2H5OH at -20°C, ultrasonic acceleration at room temp. for 2 h, Et3Al (ratio MeI/Et3Al = 3.00) dropped into soln. within 10 min, sonicated for 30 min; distilled (vac.); | A 76% B 11.2% |
| With iodine; methyl iodide In ethanol Sonication; N2, CH3I, I2 and Al introduced in a condenser with C2H5OH at -20°C, ultrasonic acceleration at room temp. for 2 h, Et3Al (ratio MeI/Et3Al = 6.00) dropped into soln. within 10 min, sonicated for 30 min; distilled (vac.); | A 65% B 15.7% |


| Conditions | Yield |
|---|---|
| Stage #1: methylene chloride With magnesium In tetrahydrofuran at 60℃; for 6h; Inert atmosphere; Stage #2: aluminum (III) chloride In hexane at 50℃; for 11h; Inert atmosphere; | 76% |
-
-
6130-98-9
2,2,7,7-tetramethyl-octa-3,5-diyne

-
-
865-37-2
dimethylaluminum hydride

-
B
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In neat (no solvent) under Ar atm. excess Me2AlH and di(tert-butyl)butadiyne were stirred at 100°C for 1.5 h; react. mixt. was cooled to room temp., volatiles were removed in vacuo, residue was dissolved in n-pentane and cooled to -15°C; | A 70% B n/a |


| Conditions | Yield |
|---|---|
| Stage #1: dimethylaluminum chloride With pyridine; iodine In dodecane at 40℃; for 1.5h; Autoclave; Stage #2: methylene chloride In dodecane at 130℃; for 24h; Temperature; Reagent/catalyst; | 70% |


| Conditions | Yield |
|---|---|
| In hexane; toluene byproducts: CH4; in a glovebox, Nd-contg. compd. (0.40 mmol) was dissolved in hexane, anda toluene suspn. of carboxylic acid (0.40 mmol) was slowly added; stirr ing at ambient temp. for 18 h; the solvent was removed in vac.; extn. in toluene; the complex was washed with hexane and dried for several hours; purifn. by crystn.; elem. anal.; | A 64% B n/a |

-
-
58435-00-0
1-chloro-4-trimethylsilyl but-3-yne

-
-
75-24-1
trimethylaluminum

-
-
86994-12-9
2-methyl-1-(trimethylsilyl)cyclobutene

| Conditions | Yield |
|---|---|
| With zirconocene dichloride In dichloromethane at 25℃; for 24h; | 100% |
| With zirconocene dichloride In dichloromethane at 25℃; for 24h; | 95 % Chromat. |
-
-
41423-29-4
4-iodo-1-butynyltrimethylsilane

-
-
75-24-1
trimethylaluminum

-
-
86994-12-9
2-methyl-1-(trimethylsilyl)cyclobutene

| Conditions | Yield |
|---|---|
| With zirconocene dichloride In dichloromethane at 25℃; for 24h; | 100% |
| With zirconocene dichloride In dichloromethane at 25℃; for 24h; | 85% |
-
-
75-24-1
trimethylaluminum

-
-
156479-95-7
(S)-2-(1,3-Dioxo-1,3-dihydro-isoindol-2-yl)-6-oxo-hexanoic acid methyl ester

| Conditions | Yield |
|---|---|
| In hexane; dichloromethane at 0℃; | 100% |
-
-
625-35-4, 3488-22-0, 10487-71-5
trans-chrotonyl chloride

-
-
75-24-1
trimethylaluminum

-
-
3102-33-8
trans-3-penten-2-one

| Conditions | Yield |
|---|---|
| With aluminium trichloride In dichloromethane for 2h; Ambient temperature; | 100% |
| With aluminium trichloride 1.) CH2Cl2, 1 h, 2.) CH2Cl2, room temperature, 2 h; other reagents: GaCl3, AlF3; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With zirconocene dichloride In hexane; 1,2-dichloro-ethane at 20℃; for 22h; Addition; | 100% |
-
-
75-24-1
trimethylaluminum

-
-
286958-06-3
(R)-1,4-Dioxa-spiro[4.5]decane-2-carboxylic acid (2S,3R)-2-(2-benzyloxy-ethyl)-tetrahydro-pyran-3-yl ester

-
-
286958-07-4
(R)-2-{1-[(2S,3R)-2-(2-Benzyloxy-ethyl)-tetrahydro-pyran-3-yloxy]-vinyl}-1,4-dioxa-spiro[4.5]decane

| Conditions | Yield |
|---|---|
| Stage #1: trimethylaluminum With bis(cyclopentadienyl)titanium dichloride In toluene at 23℃; for 72h; complex formation; Stage #2: (R)-1,4-Dioxa-spiro[4.5]decane-2-carboxylic acid (2S,3R)-2-(2-benzyloxy-ethyl)-tetrahydro-pyran-3-yl ester In tetrahydrofuran; toluene at 0 - 23℃; olefination; | 100% |

| Conditions | Yield |
|---|---|
| With titanium(IV) isopropylate; (R,S)-2-OH-3,5-Cl2-C6H2-SO2-NH-CH(CH2Ph)-CH(Ph)OH In tetrahydrofuran at 0℃; for 12h; | 100% |
| In tetrahydrofuran; hexane at -20℃; for 3h; Product distribution / selectivity; | 60% |
-
-
75-24-1
trimethylaluminum

-
-
498554-04-4
4,6-O-benzylidene-2,3-bis-O-(tert-butyldimethylsilyl)-D-glucono-1,5-lactone

-
-
498554-05-5
(1R,3R,6R,9R,10S)-9,10-bis(tert-butyldimethylsilyloxy)-8-methylene-3-phenyl-2,4,7-trioxabicyclo[4.4.0]decane

| Conditions | Yield |
|---|---|
| Stage #1: trimethylaluminum With titanocene dichloride In toluene at 23℃; for 72h; Stage #2: 4,6-O-benzylidene-2,3-bis-O-(tert-butyldimethylsilyl)-D-glucono-1,5-lactone In tetrahydrofuran; toluene at 0 - 24℃; Tebbe olefination; | 100% |
-
-
75-24-1
trimethylaluminum

-
-
107-19-7
propargyl alcohol

-
-
80434-59-9
(1R,2S,5R)-2-isopropyl-5-methylcyclohexanecarboxaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: trimethylaluminum With bis(cyclopentadienyl)dihydrozirconium In dichloromethane for 0.166667h; Stage #2: propargyl alcohol In dichloromethane at 20℃; for 12h; Stage #3: (1R,2S,5R)-2-isopropyl-5-methylcyclohexanecarbaldehyde In tetrahydrofuran; dichloromethane at -78 - 20℃; for 12h; | 100% |
| Stage #1: trimethylaluminum With zirconocene dichloride In dichloromethane at 20℃; for 0.166667h; Stage #2: propargyl alcohol In dichloromethane at 0 - 20℃; Stage #3: (1R,2S,5R)-2-isopropyl-5-methylcyclohexanecarbaldehyde In tetrahydrofuran; dichloromethane at -70 - 20℃; |

-
-
17122-96-2
1,8-dichloro-9-methylanthracene

-
-
75-24-1
trimethylaluminum

-
-
63335-10-4
1,8,9-trimethylanthracene

| Conditions | Yield |
|---|---|
| With 1,3-bis[(diphenylphosphino)propane]dichloronickel(II) In 1,2-dimethoxyethane; hexane for 7h; Heating; | 100% |
-
-
76527-25-8
3,3-diphenyloxirane-2-carboxylic acid methyl ester

-
-
75-24-1
trimethylaluminum

-
-
178306-50-8
(+/-)-methyl 2-hydroxy-3,3-diphenylbutanoate

| Conditions | Yield |
|---|---|
| In cyclohexane; toluene at 10 - 15℃; for 0.5h; Product distribution / selectivity; | 100% |
| In cyclohexane at 10℃; Product distribution / selectivity; | 96% |
| In n-heptane; toluene at -10℃; for 0.5h; Product distribution / selectivity; | 82% |
| In toluene at -10 - 10℃; Product distribution / selectivity; | 82% |
-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| With benzoic acid In toluene at 26 - 80℃; for 9.5h; | 100% |
-
-
25423-56-7
1,4,7,10-tetrathiacyclododecane

-
-
75-24-1
trimethylaluminum

-
-
112440-15-0
trimethylaluminum-thiacrown ether {12}aneS4

| Conditions | Yield |
|---|---|
| In toluene addn. of Me3Al to {12}aneS4 in abs. toluene , heated at 60°C for several h; solvent reduced in vacuo, cooling; | 100% |
-
-
1271-19-8
bis(cyclopentadienyl)titanium dichloride

-
-
75-24-1
trimethylaluminum

-
-
1278-83-7
bis(cyclopentadienyl)methyltitanium(IV) chloride

| Conditions | Yield |
|---|---|
| With tetrahydrofuran In benzene-d6; toluene N2 or Ar-atmosphere; stirring (22°C, 1 h), THF addn.; detd. by NMR spectroscopy; | 100% |
-
-
75-24-1
trimethylaluminum

| Conditions | Yield |
|---|---|
| In diethyl ether; toluene absence of air and moisture; dropwise addn. of AlMe3 in toluene to Mg(N(C3H7)2)2 in Et2O (molar ratio = 1 : 1, room temp., stirring); after 12h removal of solvent (vac.), recrystn. (hexane), sublimation (60°C); elem. anal.; | A <5 B 100% |


| Conditions | Yield |
|---|---|
| byproducts: CH4; not isolated, detected by NMR; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| byproducts: CH4; not isolated, detected by NMR; | A n/a B 100% |


| Conditions | Yield |
|---|---|
| In dichloromethane Ar-atmosphere; addn. of Al-compd. (0°C); gradual warming to room temp. (2 h); evapn.; | 100% |
-
-
912272-65-2
(CH3)3SiNC(C6H5)CH2C(C6H5)NSi(CH3)3

-
-
75-24-1
trimethylaluminum

-
-
226717-58-4
[AlMe2(CH(C(Ph)NSiMe3)2)]

| Conditions | Yield |
|---|---|
| In hexane; pentane addn. of Me3Al in hexane to ligand in pentane at -78°C; accordingto P. B. Hitchcock, M. F. Lappert and D.-S Liu, J. Chem. Soc., Chem. Co mmun., 1994, 1699; elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In hexane | 100% |
| In hexane; toluene B(C6F5)3 and AlMe3 in 1:3 toluene/hexanes mixt.; | 99% |
| In hexane; toluene (inert atm.); reaction of borane deriv. with trimethylaluminium in hexane/toluene (3:1); | 99% |


| Conditions | Yield |
|---|---|
| In hexane (inert atmosphere); -30°C slowly to room temp., stirring (2 h); evapn. to dryness, washing (hexane); | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) under N2 atm. react. Me3Al and Et4Sb2; elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
-
-
75-24-1
trimethylaluminum

-
-
32318-29-9
tris(diethylamino)difluorophosphorane

-
-
222190-83-2
[FP(N(C2H5)2)3](1+)*[(CH3)3AlF](1-)=[FP(N(C2H5)2)3][(CH3)3AlF]

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Ar atm.; equimolar amts., stirring (20°C, 24 h); evapn. (vac.); elem. anal.; | 100% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
| In toluene dry Ar atm.; 3 equiv. of AlMe3, cooling (-15°C), heating (room temp., stirring), refluxing (1,5 h, 70-80°C); concn., standing (5 to -5 degree.C, several days), washing (toluene), drying (vac.); elem. anal.; | 54% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
| In toluene dry Ar atm.; 3 equiv. of AlMe3, cooling (-15°C), heating (room temp., stirring), refluxing (1,5 h, 70-80°C); concn., standing (5 to -5 degree.C, several days), washing (toluene), drying (vac.); elem. anal.; | 57% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |
| In toluene dry Ar atm.; 3 equiv. of AlMe3, cooling (-15°C), heating (room temp., stirring), refluxing (1,5 h, 70-80°C); concn., standing (5 to -5 degree.C, several days), washing (toluene), drying (vac.); elem. anal.; | 58% |

| Conditions | Yield |
|---|---|
| In toluene molar ratio 3:1; | 100% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) substances mixted in glove box, N2; detd. by elem. anal., NMR; | 100% |
Trimethylaluminium Specification
The IUPAC name of this chemical is Trimethylaluminum. With the CAS registry number 75-24-1 and EINECS registry number 200-853-0, it is also named as Aluminum, trimethyl-. In addition, the molecular formula is C3H9Al and the molecular weight is 72.09. It is a kind of clear colorless solution and belongs to the classes of Organometallics; Al (Alminum) Compounds; Alkyl Metals; Classes of Metal Compounds; Grignard Reagents & Alkyl Metals; Synthetic Organic Chemistry; Typical Metal Compounds; Chemical Synthesis; Organoaluminum; Organometallic Reagents.
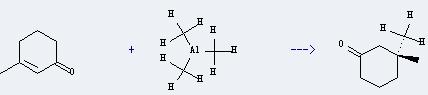
Preparation and uses of Trimethylaluminum: it can be prepared by dimethylmercury and aluminium with heating. Also it can be prepared by trihalidetrimethyldialuminium and aluminium. And it is mainly used for the production of methylaluminoxane, an activator for Ziegler-Natta catalysts for olefin polymerisation. In addition, it is used as the raw materials for MOCVD process and used in semiconductor fabrication to grow thin film. And it can react with 3-methyl-cyclohex-2-enone to get 3,3-dimethyl-cyclohexanone. This reaction will need reagents CuBr and Me3SiCl and solvent tetrahydrofuran. The reaction time is 1 hour at reaction temperature of 0 °C. The yield is about 97%.
When you are using this chemical, please be cautious about it as the following:
It is highly flammable and spontaneously flammable in air. And it can cause burns and react violently with water, liberating extremely flammable gases. In addition, it is very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. It may cause lung damage if swallowed. Besides, it may risk of impaired fertility and is harmful to the unborn child. During using it, wear suitable protective clothing, gloves and eye/face protection and avoid contact with skin and eyes. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. And in case of accident or if you feel unwell, seek medical advice immediately (show label where possible). If swallowed, do not induce vomiting: seek medical advice immediately and show this container or label. Moreover, keep away from sources of ignition-No smoking. This material and its container must be disposed of as hazardous waste. Take precautionary measures against static discharges. In case of fire, use ... (indicate in the space the precise type of fire-fighting equipment. If water increases the risk).
You can still convert the following datas into molecular structure:
(1)SMILES: C[Al](C)C
(2)InChI: InChI=1/3CH3.Al/h3*1H3;/rC3H9Al/c1-4(2)3/h1-3H3
(3)InChIKey: JLTRXTDYQLMHGR-MZZUXTGEA
Related Products
- Trimethylaluminium
- 7524-50-7
- 7524-52-9
- 75247-31-3
- 75251-98-8
- 75-25-2
- 75259-79-9
- 7526-21-8
- 7526-26-3
- 75-26-3
- 75266-40-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View