-
Name
1,12-Dodecanediamine
- EINECS 220-489-6
- CAS No. 2783-17-7
- Article Data30
- CAS DataBase
- Density 0.856 g/cm3
- Solubility 520mg/L at 19.5℃
- Melting Point 67-69 °C(lit.)
- Formula C12H28N2
- Boiling Point 274.9 °C at 760 mmHg
- Molecular Weight 200.368
- Flash Point 155 °C
- Transport Information
- Appearance White crystalline solid
- Safety 26-36/37/39-45
- Risk Codes 22-34-43
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms 1,12-Diamino-n-dodecane;1,12-Diaminododecane;1,12-Dodecamethylenediamine;1,12-Dodecylenediamine;1,12-n-Dodecanediamine;Dodecamethylenediamine;NSC 55050;NSC 59861;
- PSA 52.04000
- LogP 4.20540
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen; sodium methylate In methanol at 130℃; under 37503.8 Torr; for 5h; | 91.7% |
| With lithium aluminium tetrahydride | |
| With triisobutylaluminum |
-
-
113665-32-0
1,12-diazido-dodecane

-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| With cerium(III) chloride; sodium iodide In acetonitrile for 24h; Heating; | 55% |
| With trimethylphosphane In water-d2 at 20℃; for 25h; Staudinger Azide Reduction; |

| Conditions | Yield |
|---|---|
| In water-d2 at 24.85℃; pH=7.3; Equilibrium constant; Decomposition; |

| Conditions | Yield |
|---|---|
| In water-d2 at 24.85℃; pH=7.3; Equilibrium constant; Decomposition; |

-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| With ethanol; sodium |

-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With tris-(2-chloro-ethyl)-amine; sulfuric acid at 35 - 45℃; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: dimethylformamide 2: H2, NH3 / Raney-Ni / methanol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Na / methanol / (electrolysis) 2: dimethylformamide 3: H2, NH3 / Raney-Ni / methanol View Scheme |
-
-
38279-34-4
dodecanedial

-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: ozone / acetic acid methyl ester / 1.5 h / 5 °C 1.2: 15 - 20 °C 2.1: 5%-palladium/activated carbon; hydrogen / acetic acid methyl ester; water / 5.5 h / 15 - 25 °C / 11103.3 Torr / Autoclave 3.1: ammonium hydroxide / water; isopropyl alcohol / 10 - 60 °C / 31789.8 Torr / Autoclave View Scheme | |
| Multi-step reaction with 3 steps 1.1: ozone / acetic acid methyl ester / 1.67 h / 4.9 °C 1.2: 15 - 20 °C 2.1: 5%-palladium/activated carbon; hydrogen / acetic acid methyl ester; water / 5.5 h / 15 - 25 °C / 11103.3 Torr / Autoclave 3.1: ammonium hydroxide / water; isopropyl alcohol / 10 - 60 °C / 31789.8 Torr / Autoclave View Scheme | |
| Multi-step reaction with 3 steps 1.1: ozone / 3.5 h / 5 °C 1.2: 5 - 20 °C 2.1: 5%-palladium/activated carbon; hydrogen / acetic acid methyl ester; water / 5.5 h / 15 - 25 °C / 11103.3 Torr / Autoclave 3.1: ammonium hydroxide / water; isopropyl alcohol / 10 - 60 °C / 31789.8 Torr / Autoclave View Scheme |
-
-
55303-96-3
dodeca-4,8-diene-1,12-dialdehyde

-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 5%-palladium/activated carbon; hydrogen / acetic acid methyl ester; water / 5.5 h / 15 - 25 °C / 11103.3 Torr / Autoclave 2: ammonium hydroxide / water; isopropyl alcohol / 10 - 60 °C / 31789.8 Torr / Autoclave View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: acetic acid; ozone / 2.5 h / 5 °C / Inert atmosphere 1.2: 2 h / 10 - 20 °C 2.1: ammonium hydroxide / water; isopropyl alcohol / 60 °C / 11103.3 - 31789.8 Torr / Inert atmosphere; Autoclave View Scheme | |
| Multi-step reaction with 2 steps 1.1: ozone / acetic acid methyl ester / 4.4 °C / Inert atmosphere 1.2: 20 °C 2.1: ammonium hydroxide / water; isopropyl alcohol / 60 °C / 11103.3 - 31789.8 Torr / Inert atmosphere; Autoclave View Scheme |

-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: trifluoroacetic acid / 5 h / 20 °C 2.1: trimethylphosphane / water-d2; deuteromethanol / 25 h 2.2: 24 h View Scheme |
-
-
1416710-80-9
12-azidododecan-1-amine

-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| Stage #1: 12-azidododecan-1-amine With trimethylphosphane In deuteromethanol; water-d2 for 25h; Staudinger Azide Reduction; Stage #2: for 24h; |
-
-
887353-35-7
N-(tent-butoxycarbonyl)-12-bromododecylamine

-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: sodium azide / N,N-dimethyl-formamide / 20 h / 85 °C 2.1: trifluoroacetic acid / 5 h / 20 °C 3.1: trimethylphosphane / water-d2; deuteromethanol / 25 h 3.2: 24 h View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: lithium aluminium tetrahydride / tetrahydrofuran / 0 - 20 °C 2: tris(2,4-pentanedionato)ruthenium(III); [2-((diphenylphospino)methyl)-2-methyl-1,3-propanediyl]bis[diphenylphosphine]; hydrogen; ammonia / 1,4-dioxane; water / 20 h / 220 °C / 7500.75 Torr / Autoclave; Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1: ammonium / 0.5 h / Heating 2: phosphorus pentoxide / 1 h / 15 - 37.5 Torr / Heating 3: hydrogen / ethanol / 1 h / 100 °C / 60006 Torr View Scheme | |
| Multi-step reaction with 3 steps 1: ammonium / 0.5 h / Heating 2: 1 h / 15 - 37.5 Torr / Heating 3: hydrogen / ethanol / 1 h / 100 °C / 60006 Torr View Scheme |

| Conditions | Yield |
|---|---|
| With tris(2,4-pentanedionato)ruthenium(III); ammonia; hydrogen; [2-((diphenylphospino)methyl)-2-methyl-1,3-propanediyl]bis[diphenylphosphine] In 1,4-dioxane; water at 220℃; under 7500.75 Torr; for 20h; Temperature; Autoclave; Inert atmosphere; | 87 %Spectr. |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: phosphorus pentoxide / 1 h / 315 °C / 15001.5 - 37503.8 Torr 2: hydrogen / ethanol / 1 h / 100 °C / 60006 Torr View Scheme | |
| Multi-step reaction with 2 steps 1: phosphorus pentoxide / 1 h / 15 - 37.5 Torr / Heating 2: hydrogen / ethanol / 1 h / 100 °C / 60006 Torr View Scheme | |
| Multi-step reaction with 2 steps 1: 1 h / 15 - 37.5 Torr / Heating 2: hydrogen / ethanol / 1 h / 100 °C / 60006 Torr View Scheme |

| Conditions | Yield |
|---|---|
| With pyridoxal 5'-phosphate; MATITNHMPTAELQALDAAHHLHPFSANNALGEEGTRVITRARGVWLNDSEGEEILDAMAGLWCVNIGYGRDELAEVAARQMRELPYYNTFFKTTHVPAIALAQKLAELAPGDLNHVFFAGGGSEANDTNIRMVRTYWQNKGQPEKTVIISRKNAYHGSTVASSALGGMAGMHAQSGLIPDVHHINQPNWWAEGGDMDPEEFGLARARELEEAILELGENRVAAFIAEPVQGAGGVIVAPDSYWPEIQRICDKYDILLIADEVICGFGRTGNWFGTQTMGIRPHIMTIAKGLSSGYAPIGGSIVCDEVAHVIGKDEFNHGYTYSGHPVAAAVALENLRILEEENILDHVRNVAAPYLKEKWEALTDHPLVGEAKIVGMMASIALTPNKASRAKFASEPGTIGYICRERCFANNLIMRHVGDRMIISPPLVITPAEIDEMFVRIRKSLDEAQAEIEKQGLMKSA; MIKAYAALEANGKLQPFEYDPGALGANEVEIEVQYCGVCHSDLSMINNEWGISNYPLVPGHEVVGTVAAMGEGVNHVEVGDLVGLGWHSGYCMTCHSCLSGYHNLCATAESTIVGHYGGFGDRVRAKGVSVVKLPKGIDLASAGPLFCGGITVFSPMVELSLKPTAKVAVIGIGGLGHLAVQFLRAWGCEVTAFTSSARKQTEVLELGAHHILDSTNPEAIASAEGKFDYIISTVNLKLDWNLYISTLAPQGHFHFVGVVLEPLDLNLFPLLMGQRSVSASPVGSPATIATMLDFAVRHDIKPVVEQFSFDQINEAIAHLESGKAHYRVVLSHSKN; nicotinamide adenine dinucleotide phosphate; benzylamine In dimethyl sulfoxide at 37℃; for 24h; pH=8; Solvent; Microbiological reaction; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With hydrogen chloride In dimethylsulfoxide-d6 |


| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 2h; Inert atmosphere; | 100% |
| With potassium hydroxide In 1,2-dichloro-ethane | |
| With potassium hydroxide |
-
-
96-82-2, 534-41-8, 534-42-9, 51267-45-9, 58459-39-5, 69728-97-8
lactobionic acid

-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| In methanol for 24h; Heating; | 100% |
-
-
86-98-6
4,7-dichloroquinoline

-
-
2783-17-7
1,12-Diaminododecane

-
-
1026678-59-0
N1-(7-Chloro-quinolin-4-yl)-dodecane-1,12-diamine

| Conditions | Yield |
|---|---|
| Inert atmosphere; | 100% |
| 1.) 80 deg C, 1 h, 2.) 135-145 deg C, 3 h; |
-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide; toluene at 145 - 150℃; for 24h; | 100% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 0.25h; | 100% |
| In tetrahydrofuran at 20℃; for 0.25h; | 100% |

| Conditions | Yield |
|---|---|
| In water for 13h; Reflux; | 100% |
| In water at 20℃; Reflux; | |
| In water at 20℃; Reflux; |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 20℃; for 0.25h; | 100% |
| In tetrahydrofuran at 20℃; for 0.25h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 2,3,4-tris(benzyloxy)benzoic acid With benzotriazol-1-yloxyl-tris-(pyrrolidino)-phosphonium hexafluorophosphate In dichloromethane for 0.0833333h; Stage #2: 1,12-Diaminododecane With triethylamine In dichloromethane at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| In methanol at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 2h; Inert atmosphere; | 100% |
-
-
2783-17-7
1,12-Diaminododecane

-
-
3943-56-4, 15536-18-2
1,12-Dodecanediamine, dihydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride In 1,4-dioxane; methanol for 0.166667h; | 100% |
-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| With trihydroxy amine In water | 100% |
-
-
2783-17-7
1,12-Diaminododecane

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
109792-60-1
tert-butyl (12-aminododecyl)carbamate

| Conditions | Yield |
|---|---|
| With sodium hydroxide In dichloromethane; water for 1.08333h; | 99% |
| In chloroform at 0 - 20℃; | 90% |
| In chloroform | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: 1,12-Diaminododecane; pentaerythritol tetraacrylate In methanol at 25℃; for 18h; Michael addition; Stage #2: morpholine In methanol at 20℃; for 18h; Further stages.; | 99% |


| Conditions | Yield |
|---|---|
| In N,N-dimethyl-formamide at 20℃; for 14h; | 98% |

| Conditions | Yield |
|---|---|
| lipase from Candida antarctica In toluene at 70℃; for 19h; Product distribution / selectivity; | 98% |

| Conditions | Yield |
|---|---|
| for 3h; Milling; Green chemistry; | 98% |
-
-
2783-17-7
1,12-Diaminododecane

| Conditions | Yield |
|---|---|
| Stage #1: 1,12-Diaminododecane; bis(calix[4]imidazolylidene[2]pyrazolato)octakissilver(I) tetrakis(trifluoromethanesulfonate) In N,N-dimethyl-formamide at 20℃; for 0.166667h; Schlenk technique; Inert atmosphere; Stage #2: 3,5-di-tert-butylbenzoic anhydride With N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at -50 - 20℃; for 16h; Schlenk technique; Inert atmosphere; | 98% |

-
-
2783-17-7
1,12-Diaminododecane

-
-
108-24-7
acetic anhydride

-
-
31991-77-2
N,N'-Diacetyl-1,12-dodecandiamin

| Conditions | Yield |
|---|---|
| With pyridine at 20℃; | 97% |

| Conditions | Yield |
|---|---|
| Stage #1: N-Cbz-L-Phe With 1,1'-carbonyldiimidazole In chloroform at 20℃; for 1.5h; Stage #2: 1,12-Diaminododecane In chloroform at 20℃; | 97% |
| With dmap; dicyclohexyl-carbodiimide In chloroform for 20h; Ambient temperature; | 74% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) at 20℃; for 24h; | 97% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 2h; Inert atmosphere; | 97% |
-
-
2783-17-7
1,12-Diaminododecane

-
-
51814-82-5, 51814-83-6, 58910-28-4
ethyl 2-(1-ethoxyethylidene)hydrazinecarboxylate

| Conditions | Yield |
|---|---|
| In water for 5.5h; Heating; | 96% |

| Conditions | Yield |
|---|---|
| In water for 5.5h; Heating; | 96% |

| Conditions | Yield |
|---|---|
| In tetrahydrofuran Heating; | 96% |

| Conditions | Yield |
|---|---|
| In neat (no solvent) for 1.5h; Milling; Green chemistry; | 96% |
-
-
2783-17-7
1,12-Diaminododecane

-
-
13139-14-5
Boc-Trp-OH

| Conditions | Yield |
|---|---|
| With O-(1H-benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate; N-ethyl-N,N-diisopropylamine In N,N-dimethyl-formamide at 20℃; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: Z-Leu-OH With 1,1'-carbonyldiimidazole In chloroform at 20℃; for 1.5h; Stage #2: 1,12-Diaminododecane In chloroform at 20℃; | 95.7% |
-
-
2783-17-7
1,12-Diaminododecane

-
-
2381-23-9
9,10-dioxo-9,10-dihydro-2-anthracenesulfonyl chloride

-
-
912278-57-0
C40H40N2O8S2

| Conditions | Yield |
|---|---|
| With potassium carbonate In tetrahydrofuran; water at 60 - 65℃; for 2h; | 95% |
-
-
2783-17-7
1,12-Diaminododecane

-
-
111-27-3
hexan-1-ol

-
-
58982-05-1
Hexanoic acid (12-hexanoylamino-dodecyl)-amide

| Conditions | Yield |
|---|---|
| With C29H44Cl2N2Ru; potassium tert-butylate In toluene for 24h; Reflux; | 95% |
1,12-Dodecanediamine Chemical Properties
Molecular Structure of 1,12-Dodecamethylenediamine (2783-17-7):
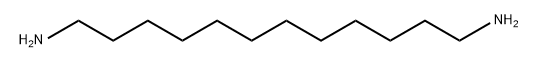
EINECS: 220-489-6
IUPAC Name: Dodecane-1,12-diamine
Molecular Formula: C12H28N2
Molecular Weight: 200.36412 g/mol
XLogP3-AA: 3
H-Bond Donor: 2
H-Bond Acceptor: 2
Canonical SMILES: C(CCCCCCN)CCCCCN
InChI: InChI=1S/C12H28N2/c13-11-9-7-5-3-1-2-4-6-8-10-12-14/h1-14H2
InChIKey: QFTYSVGGYOXFRQ-UHFFFAOYSA-N
Index of Refraction: 1.465
Molar Refractivity: 64.71 cm3
Molar Volume: 233.9 cm3
Surface Tension: 34.6 dyne/cm
Density: 0.856 g/cm3
Flash Point: 155 °C
Melting Point: 67-69 °C(lit.)
Boiling Point: 274.9 °C at 760 mmHg
Enthalpy of Vaporization: 51.33 kJ/mol
Vapour Pressure: 0.00526 mmHg at 25 °C
Water Solubility: 684.3 mg/L at 25 °C
BRN: 1742765
1,12-Dodecanediamine Toxicity Data With Reference
| 1. | eye-rbt 500 mg MLD | JACTDZ Journal of the American College of Toxicology. 1 (1990),92. | ||
| 2. | orl-rat LDLo:670 mg/kg | JACTDZ Journal of the American College of Toxicology. 1 (1990),92. | ||
| 3. | orl-mus LD50:1088 mg/kg | GISAAA Gigiena i Sanitariya. 43 (5)(1978),18. |
1,12-Dodecanediamine Consensus Reports
Reported in EPA TSCA Inventory.
1,12-Dodecanediamine Safety Profile
Safety Information of 1,12-Dodecamethylenediamine (2783-17-7):
Hazard Codes: C ![]()
Risk Statements: 22-34-43
22: Harmful if swallowed
34: Causes burns
43: May cause sensitization by skin contact
Safety Statements: 26-36/37/39-45
26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37/39: Wear suitable protective clothing, gloves and eye/face protection
45: In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
RIDADR: UN 3259 8/PG 3
WGK Germany: 1
RTECS: JR2200000
Moderately toxic by ingestion. An eye irritant. When heated to decomposition it emits toxic fumes of NOx. See also AMINES.
1,12-Dodecanediamine Specification
1,12-Dodecamethylenediamine (2783-17-7) is white crystalline solid, also known as 1,12'-Dodecamethylenediamine ; 1,12'-Dodecylenediamine ; 1,12-Diamindodecane ; 1,12-Diaminododecane ; 1,12-Dodecanediamine ; 4-04-00-01376 (Beilstein Handbook Reference) ; Dodecamethylenediamine ; Dodecyldiamine ; NSC 55050 ; NSC 59861 ; Dodecamethylenediamine . It is incompatible with strong oxidizing agents. It decompounds to yield nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, nitrogen. 1,12-Dodecamethylenediamine (2783-17-7) is one of the monomer to product nylon 1212 and also used as pharmaceutical intermediate.
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 27832-58-2
- 27832-84-4
- 27833-64-3
- 27834-99-7
- 27835-00-3
- 2783-94-0
- 27841-06-1
- 278-43-3
- 2784-50-1
- 27848-80-2
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View