-
Name
1,2,3-Trifluoro-4-nitrobenzene
- EINECS 212-238-4
- CAS No. 771-69-7
- Article Data12
- CAS DataBase
- Density 1.554 g/cm3
- Solubility
- Melting Point
- Formula C6H2F3NO2
- Boiling Point 201.8 °C at 760 mmHg
- Molecular Weight 177.083
- Flash Point 87 °C
- Transport Information UN 2810
- Appearance clear yellow liquid after melting
- Safety 26-36/37/39-24/25
- Risk Codes 20/21/22-36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms 2,3,4-Trifluoro-1-nitrobenzene;2,3,4-Trifluoronitrobenzene;4-Nitro-1,2,3-trifluorobenzene;2.3.4-Dichloronitrobenzeme;
- PSA 45.82000
- LogP 2.53530
Synthetic route
-
-
17700-09-3
4-nitro-1,2,3-trichlorobenzene

-
-
771-69-7
2,3,4-trifluoronitrobenzene

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitro-1,2,3-trichlorobenzene With potassium fluoride; tetrabutylammomium bromide In dimethyl sulfoxide at 75 - 80℃; for 4h; Stage #2: With potassium fluoride; tetrabutyl ammonium fluoride In dimethyl sulfoxide at 75 - 180℃; for 12h; Time; | 99.7% |

| Conditions | Yield |
|---|---|
| Stage #1: 1,3-dichloro-2-fluorobenzene With sulfuric acid; nitric acid at 20 - 25℃; for 2.5h; Stage #2: With sulfolane; potassium fluoride for 5h; | 85.5% |
| Multi-step reaction with 2 steps 1: sulfuric acid; aqueous nitric acid / 40 °C 2: dimethylformamide; potassium fluoride / 150 °C View Scheme | |
| Multi-step reaction with 2 steps 1: sulfuric acid; nitric acid / 2 h / Heating 2: sulfolane; potassium fluoride / 8 h / 200 - 210 °C View Scheme |

| Conditions | Yield |
|---|---|
| With potassium fluoride at 160 - 165℃; for 7h; Time; | 85% |
| With potassium fluoride; immobilized pyridinium salt In sulfolane at 180℃; for 2h; | 68% |
| With potassium fluoride; N,N-dimethyl-formamide at 150℃; | |
| With sulfolane; potassium fluoride at 200 - 210℃; for 8h; Temperature; | 124 g |
-
-
163733-96-8
3,4,5-trifluoro aniline

-
-
771-69-7
2,3,4-trifluoronitrobenzene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tetrahydrofuran View Scheme |


-
-
393-79-3
1,3-dichloro-2-fluoro-4-nitrobenzene

-
-
3115-68-2
tetrabutyl phosphonium bromide

-
-
771-69-7
2,3,4-trifluoronitrobenzene

| Conditions | Yield |
|---|---|
| With potassium fluoride In 5,5-dimethyl-1,3-cyclohexadiene |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: nitric acid; sulfuric acid / 2 h / 95 - 100 °C 2.1: potassium fluoride / 3 h / 140 - 150 °C 3.1: chlorine / 180 - 190 °C 4.1: nitric acid; sulfuric acid / 2.5 h / 20 - 25 °C 4.2: 5 h View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: potassium fluoride / 3 h / 140 - 150 °C 2.1: chlorine / 180 - 190 °C 3.1: nitric acid; sulfuric acid / 2.5 h / 20 - 25 °C 3.2: 5 h View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: chlorine / 180 - 190 °C 2.1: nitric acid; sulfuric acid / 2.5 h / 20 - 25 °C 2.2: 5 h View Scheme |
-
-
20098-48-0
3,4,5-trichloronitrobenzen

-
-
771-69-7
2,3,4-trifluoronitrobenzene

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1.1: potassium fluoride; N-benzyl-N,N,N-triethylammonium chloride / 11 h / 140 - 175 °C 2.1: hydrogen / methanol / Inert atmosphere 3.1: nitrosylsulfuric acid / 6 h / 10 - 15 °C 3.2: 1 h 4.1: sulfuric acid; nitric acid / 2 h / Heating 5.1: sulfolane; potassium fluoride / 8 h / 200 - 210 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: hydrogen / methanol / Inert atmosphere 2.1: nitrosylsulfuric acid / 6 h / 10 - 15 °C 2.2: 1 h 3.1: sulfuric acid; nitric acid / 2 h / Heating 4.1: sulfolane; potassium fluoride / 8 h / 200 - 210 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: nitrosylsulfuric acid / 6 h / 10 - 15 °C 1.2: 1 h 2.1: sulfuric acid; nitric acid / 2 h / Heating 3.1: sulfolane; potassium fluoride / 8 h / 200 - 210 °C View Scheme |

| Conditions | Yield |
|---|---|
| With sodium hexamethyldisilazane In tetrahydrofuran at -78℃; | 100% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
100-79-8
(R,S)-2,2-dimethyl-1,3-dioxolane-4-methanol

-
-
129798-33-0
(R)-3,4-difluoro-2-(2,3-isopropylidenedioxypropoxy)nitrobenzene

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; toluene | 100% |

| Conditions | Yield |
|---|---|
| With sodium t-butanolate In tetrahydrofuran at 0 - 20℃; | 100% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
62-53-3
aniline

-
-
1393178-31-8
(2,3-Difluoro-6-nitro-phenyl)-phenylamine

| Conditions | Yield |
|---|---|
| Stage #1: aniline With lithium hexamethyldisilazane In tetrahydrofuran at -78℃; for 0.166667h; Inert atmosphere; Stage #2: 2,3,4-trifluoronitrobenzene In tetrahydrofuran at -78℃; for 0.5h; Stage #3: With water In tetrahydrofuran | 100% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
124-41-4
sodium methylate

-
-
155020-44-3
2-fluoro-1,3-dimethoxy-4-nitrobenzene

| Conditions | Yield |
|---|---|
| In methanol Ambient temperature; | 99% |
| In methanol at 4 - 20℃; for 0.5h; | 99% |
| In methanol at 0 - 20℃; | 98% |
| In methanol at 0 - 20℃; | 80% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
74-89-5
methylamine

-
-
170432-54-9
2,3-difluoro-N-methyl-6-nitroaniline

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 40℃; for 18h; | 99% |

| Conditions | Yield |
|---|---|
| With sodium hypophosphite monohydrate; 1% platinum on charcoal; hydrogen at 65 - 80℃; under 6750.68 Torr; for 2.75h; Reagent/catalyst; Temperature; Autoclave; | 98.52% |
| With hydrogen; nickel Autoclave; | 91.8% |
| With iron; ammonium chloride |
-
-
1658-27-1
1,5-dioxaspiro[5.5]undecane-2,4-dione

-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

| Conditions | Yield |
|---|---|
| In acetonitrile at 45℃; for 20h; | 98% |

-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
55346-93-5
1,2,3-Trifluoro-4,6-dinitrobenzene

| Conditions | Yield |
|---|---|
| With trifluorormethanesulfonic acid; tris(trifluoromethanesulfonyloxy)boron; nitric acid at 65℃; for 6h; | 96% |
| With sulfuric acid; potassium nitrate | 91% |
| With sulfuric acid; nitric acid at 80℃; for 2h; | 84.2% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
251940-05-3
1-(4-fluoro-phenyl)-2-(tetrahydro-pyrimidin-2-ylidene)-ethanone

| Conditions | Yield |
|---|---|
| Stage #1: 2,3,4-trifluoronitrobenzene; 1-(4-fluoro-phenyl)-2-(tetrahydro-pyrimidin-2-ylidene)-ethanone With sodium carbonate In 1,4-dioxane at 80℃; for 4h; Stage #2: With caesium carbonate In 1,4-dioxane for 3h; Reflux; | 93% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
82100-30-9
1-phenyl-2-(tetrahydropyrimidin-2(1H)-ylidene)ethan-1-one

| Conditions | Yield |
|---|---|
| Stage #1: 2,3,4-trifluoronitrobenzene; 1-phenyl-2-(tetrahydropyrimidin-2(1H)-ylidene)ethan-1-one With potassium carbonate In 1,4-dioxane at 80℃; for 3h; Stage #2: With caesium carbonate In 1,4-dioxane for 3h; Reflux; | 93% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
211693-73-1
2-amino-3,4-difluoronitro-benzene

| Conditions | Yield |
|---|---|
| With ammonium hydroxide In ethanol at 20℃; for 8h; | 91.2% |
| With ammonia In methanol at 70℃; for 1.5h; Microwave irradiation; | 73% |
| With ammonia In methanol at 70℃; for 1.5h; Microwave irradiation; | 35.6% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
89401-28-5
(2R)-1-(benzyloxy)propan-2-ol

| Conditions | Yield |
|---|---|
| With potassium hydroxide; potassium carbonate In water; toluene | 91% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
141-97-9
ethyl acetoacetate

-
-
121247-16-3
1-(2,3-difluoro-6-nitrophenyl)propan-2-one

| Conditions | Yield |
|---|---|
| With hydrogenchloride; acetic acid In tetrahydrofuran; water | 90% |
| Stage #1: ethyl acetoacetate With potassium tert-butylate In tetrahydrofuran at 11 - 25℃; for 1h; Stage #2: 2,3,4-trifluoronitrobenzene In tetrahydrofuran at 10 - 21℃; for 3.08333h; Stage #3: With sulfuric acid In acetic acid at 70℃; for 3h; Heating / reflux; | 82% |
| With acetic acid In tetrahydrofuran; hydrogenchloride | 72% |
| With acetic acid In tetrahydrofuran; hydrogenchloride | 72% |
| With sodium hydride Yield given. Multistep reaction; |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
770719-09-0
3-chloro-5-[(2,3-difluoro-6-nitrophenyl)oxy]benzonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 3-chloro-5-hydroxybenzonitrile With sodium t-butanolate In tetrahydrofuran at 0℃; for 0.25h; Stage #2: 2,3,4-trifluoronitrobenzene In tetrahydrofuran at 0℃; for 3.5h; | 89% |
| Stage #1: 3-bromo-5-chlorophenol With sodium t-butanolate In tetrahydrofuran at 0℃; for 1h; Stage #2: 2,3,4-trifluoronitrobenzene In tetrahydrofuran at 0℃; Stage #3: With hydrogenchloride In tetrahydrofuran; water for 1h; | 82% |
| Stage #1: 3-chloro-5-hydroxybenzonitrile With sodium t-butanolate In tetrahydrofuran at 0℃; for 1h; Stage #2: 2,3,4-trifluoronitrobenzene In tetrahydrofuran at 0℃; | 82% |
| Stage #1: 3-chloro-5-hydroxybenzonitrile With potassium tert-butylate In tetrahydrofuran at 0℃; for 0.333333h; Stage #2: 2,3,4-trifluoronitrobenzene In tetrahydrofuran at 20℃; for 2h; |
-
-
473923-97-6
3-chloro-5-hydroxybenzonitrile

-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
770719-09-0
3-chloro-5-[(2,3-difluoro-6-nitrophenyl)oxy]benzonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 3-chloro-5-hydroxybenzonitrile With sodium t-butanolate In tetrahydrofuran at 0℃; Stage #2: 2,3,4-trifluoronitrobenzene In tetrahydrofuran at 0 - 20℃; | 89% |
| With sodium t-butanolate In tetrahydrofuran at 0℃; for 1h; | 82% |
| Stage #1: 3-chloro-5-hydroxybenzonitrile With sodium t-butanolate In tetrahydrofuran at 0℃; for 1h; Stage #2: 2,3,4-trifluoronitrobenzene In tetrahydrofuran at 0℃; Stage #3: With hydrogenchloride; water In tetrahydrofuran for 1h; | 82% |
-
-
67-56-1
methanol

-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
155020-44-3
2-fluoro-1,3-dimethoxy-4-nitrobenzene

| Conditions | Yield |
|---|---|
| With sodium methylate for 48h; Reflux; | 89% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
141-97-9
ethyl acetoacetate

-
A
-
288385-96-6
1,2-difluoro-3-(2,2-dimethoxypropyl)-4-nitrobenzene

-
B
-
121247-16-3
1-(2,3-difluoro-6-nitrophenyl)propan-2-one

| Conditions | Yield |
|---|---|
| With acetic acid In tetrahydrofuran; hydrogenchloride | A 88% B 72% |

-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
866959-42-4
3,4-difluoro-2-(3,5-bis-tert-butylcarbamoyl-phenoxy)nitrobenzene

| Conditions | Yield |
|---|---|
| Stage #1: N,N'-di-tert-butyl-5-hydroxy-isophthalamide With potassium tert-butylate In tetrahydrofuran at 0℃; for 0.5h; Stage #2: 2,3,4-trifluoronitrobenzene In tetrahydrofuran at -35 - 30℃; for 21h; | 88% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
107165-84-4
1-(4-methoxyphenyl)-2-(tetrahydropyrimidin-2(1H)-ylidene)ethan-1-one

| Conditions | Yield |
|---|---|
| Stage #1: 2,3,4-trifluoronitrobenzene; 1-(4-methoxyphenyl)-2-(tetrahydropyrimidin-2(1H)-ylidene)ethan-1-one With triethylamine In 1,4-dioxane at 60℃; for 5h; Stage #2: With caesium carbonate In 1,4-dioxane for 3h; Reflux; | 87% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
82419-26-9
2,3-difluoro-6-nitrophenol

| Conditions | Yield |
|---|---|
| With potassium hydroxide; sulfuric acid In water | 86% |
| With potassium hydroxide In water; dimethyl sulfoxide at 18 - 20℃; for 2h; | 29% |
| With potassium hydroxide In water; dimethyl sulfoxide | |
| With potassium hydroxide In dimethyl sulfoxide at 15 - 17℃; for 3h; | 38.8g |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 0℃; for 1h; | 86% |

| Conditions | Yield |
|---|---|
| With sodium t-butanolate In tetrahydrofuran at 0℃; for 2h; | 84% |
| Stage #1: 7-Indolol With sodium t-butanolate In tetrahydrofuran at 0℃; for 0.166667h; Stage #2: 2,3,4-trifluoronitrobenzene In tetrahydrofuran | 84% |

-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
211693-73-1
2-amino-3,4-difluoronitro-benzene

| Conditions | Yield |
|---|---|
| In ethyl acetate | 84% |
-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
184110-41-6
(R)-(-)-2-(1-ethoxyethoxy)-1-propanol

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; diethyl ether; water | 82% |
| With potassium hydroxide; potassium carbonate In water; toluene | 6.7% |
-
-
874974-85-3
3-(difluoromethyl)-5-hydroxybenzonitrile

-
-
771-69-7
2,3,4-trifluoronitrobenzene

-
-
1068603-76-8
3-difluoromethyl-5-(2,3-difluoro-6-nitro-phenoxy)-benzonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 3-(difluoromethyl)-5-hydroxybenzonitrile; 2,3,4-trifluoronitrobenzene With potassium carbonate In tetrahydrofuran at 0℃; Stage #2: With acetic acid In tetrahydrofuran at 0 - 5℃; Product distribution / selectivity; | 80% |
1,2,3-Trifluoro-4-nitrobenzene Chemical Properties
The molecular formula of 1,2,3-Trifluoro-4-nitrobenzene(771-69-7) is C6H2F3NO2 and its formula weight is 177.08.
The density of 1,2,3-Trifluoro-4-nitrobenzene(771-69-7) is 1.541 g/mL at 25 °C(lit.) and it has a boiling point of 92 °C 20 mm Hg(lit.). The refractive index is about 1.492(lit.). Its flash point is 200 °F.
The chemical synonyms of 1,2,3-Trifluoro-4-nitrobenzene(771-69-7) are 4-NITRO-1,2,3-Trifluorobenzene;1,2,3-TRIFLUORO-4-NITROBENZENE;1,2,3-Trifluoro-4-nitrobenzene;2,3,4-TRIFLUORO-1-NITROBENZENE;3,4,5 TRIFLUORONITROBENZEN;2,3,4-TrifluorNITROBENZENE;4-chloro-2-fluroaniline;2,3,4-TRIFLUORONITROBRNZENE
The molecular structure of 1,2,3-Trifluoro-4-nitrobenzene(771-69-7):
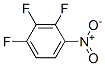
1,2,3-Trifluoro-4-nitrobenzene Uses
1,2,3-Trifluoro-4-nitrobenzene Toxicity Data With Reference
RTECS#: CAS# 771-69-7: None listed
LD50/LC50: RTECS: Not available.
Carcinogenicity: 1,2,3-Trifluoro-4-nitrobenzene - Not listed as a carcinogen by ACGIH, IARC, NTP, or CA Prop 65.
Other: The toxicological properties have not been fully investigated.
1,2,3-Trifluoro-4-nitrobenzene Safety Profile
Safety Statements :
S26: In case of contact with eyes, rinse immediately with plenty of WATER and seek medical advice
S36/37/39: Wear suitable protective clothing, gloves and eye/face protection
S24/25: Avoid contact with skin and eyes
Hazard Codes Xn,Xi
Risk Statements 20/21/22-36/37/38
Safety Statements 26-36/37/39-24/25
RIDADR UN2810
WGK Germany 3
Hazard Note Irritant
HazardClass 6.1
PackingGroup III
1,2,3-Trifluoro-4-nitrobenzene Specification
Conditions to Avoid: Incompatible materials.
Incompatibilities with Other Materials Strong oxidizing agents.
Hazardous Decomposition Products Nitrogen oxides, carbon monoxide, carbon dioxide, hydrogen fluoride gas.
Hazardous Polymerization Has not been reported.
Related Products
- 10,10'-Bis([1,1'-biphenyl]-4-yl)-9,9'-bianthracene
- 10,10'-Dibromo-9,9'-bianthryl
- 10,10-Dimethylanthrone
- 10,10-Oxybisphenoxarsine
- 10-[1,1'-Biphenyl]-4-yl-2-(1-methylethyl)-9-oxo-9H-thioxanthenium hexafluorophosphate
- 10,11-Dehydroimipramine
- 10,11-Dihydro-11-oxodibenzo[b,f][1,4]thiazepine
- 10,11-Dihydro-2-(4-methyl-1-piperazinyl)-11-(2-athiazolyl)-pyridazino(3,4-b)(1,4)benzoxazepine
- 10,11-DIHYDRO-2-(4-METHYL-1-PIPERAZINYL)-11-(3,4-XYLYL)PYRIDAZINO(3,4b)(1,4)-BENZOXAZEPINE MALEATE
- 10,11-Dihydro-5-(3-dimethylamino-2-methylpropyl)-5h-dibenz (b,f)azepine
- 77171-00-7
- 77171-65-4
- 77173-67-2
- 77174-66-4
- 77-17-8
- 77180-80-4
- 77181-26-1
- 77182-82-2
- 77184-95-3
- 7718-51-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View