-
Name
1-Bromo-4-tert-butylbenzene
- EINECS 223-599-2
- CAS No. 3972-65-4
- Article Data61
- CAS DataBase
- Density 1.236 g/cm3
- Solubility Insoluble in water.
- Melting Point 13-16 °C(lit.)
- Formula C10H13Br
- Boiling Point 232 °C at 760 mmHg
- Molecular Weight 213.117
- Flash Point 97.2 °C
- Transport Information
- Appearance clear colorless to slightly yellow liquid
- Safety 23-24/25-37/39-26
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Benzene,1-bromo-4-tert-butyl- (6CI,7CI,8CI);1-Bromo-4-(1,1-dimethylethyl)benzene;1-Bromo-4-t-butylbenzene;1-tert-Butyl-4-bromobenzene;4-Bromo-tert-butylbenzene;4-tert-Butyl-1-bromobenzene;4-tert-Butylbromobenzene;4-tert-Butylphenylbromide;B 1727;NSC 43038;p-Bromo-tert-butylbenzene;p-tert-Butylbromobenzene;
- PSA 0.00000
- LogP 3.74660
Synthetic route
-
-
253185-03-4, 253185-04-5
tert-butylbenzene

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

| Conditions | Yield |
|---|---|
| With bromine; iodine at 0℃; | 100% |
| With carbon dioxide; bromine at 40℃; under 187519 Torr; for 2h; Supercritical conditions; Green chemistry; | 100% |
| With benzyltrimethylazanium tribroman-2-uide; zinc(II) chloride In acetic acid at 70℃; for 2h; | 95% |

| Conditions | Yield |
|---|---|
| 97% |
-
-
214360-66-4
2-(4-tert-butylphenyl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

| Conditions | Yield |
|---|---|
| With copper(ll) bromide In methanol Heating; | 96% |
-
-
108-86-1
bromobenzene

-
-
15512-06-8
3,6-di-tert-butylbenzene-1,2-diol

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

| Conditions | Yield |
|---|---|
| With sulfuric acid | 90% |
-
-
402-43-7
p-trifluoromethylphenyl bromide

-
-
75-24-1
trimethylaluminum

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

| Conditions | Yield |
|---|---|
| In hexane; 1,2-dichloro-ethane at 25 - 50℃; Inert atmosphere; | 89% |
-
-
108-86-1
bromobenzene

-
-
3282-30-2
pivaloyl chloride

-
A
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
B
-
30314-45-5
1-(4-bromophenyl)-2,2-dimethylpropan-1-one

| Conditions | Yield |
|---|---|
| With iodine for 0.166667h; Ambient temperature; Irradiation; | A 8% B 87% |
| With iron(III) sulfate In hexane at 20℃; for 0.75h; Friedel-Crafts acylation; Sonication; | A 8% B 72% |
| With aluminium trichloride In hexane for 0.166667h; Ambient temperature; Irradiation; | A 25% B 50% |

-
-
154318-75-9
4-tert-butylphenyl triflate

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

| Conditions | Yield |
|---|---|
| With [Cp*Ru(CH3CN)3]OTf; lithium bromide at 120℃; for 48h; Inert atmosphere; | 87% |
| Multi-step reaction with 2 steps 1: 86 percent / Et3N / PdCl2(dppf) / dioxane / Heating 2: 96 percent / aq. CuBr2 / methanol / Heating View Scheme |

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

| Conditions | Yield |
|---|---|
| With tetra-N-butylammonium tribromide In tetrahydrofuran; water at 20℃; for 0.333333h; chemoselective reaction; | 86% |
| With tetra-N-butylammonium tribromide In tetrahydrofuran; water at 20℃; for 0.333333h; chemoselective reaction; | 86% |
-
-
176780-03-3, 164927-39-3
(P(o-tol)3)2Pd2(C6H4tBu-4)2Br2

-
-
13716-12-6
tri-tert-butyl phosphine

-
A
-
53199-31-8
bis(tri-t-butylphosphine)palladium(0)

-
B
-
3972-65-4
1-bromo-4-tert-butylbenzene

| Conditions | Yield |
|---|---|
| In benzene-d6 byproducts: P(o-tol)3, biaryl, arene; in C6D6 soln. at 70°C; K(eq) = 3.3(6)E-4; | A n/a B 75% |

-
-
108-86-1
bromobenzene

-
-
3282-30-2
pivaloyl chloride

-
A
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
B
-
61593-14-4
1-(2-bromophenyl)-2,2-dimethyl-propan-1-one

-
C
-
30314-45-5
1-(4-bromophenyl)-2,2-dimethylpropan-1-one

| Conditions | Yield |
|---|---|
| With iodine for 6h; Ambient temperature; | A 64% B 6% C 17% |
| With aluminium trichloride In hexane for 5h; Ambient temperature; | A 42% B 5% C 12% |


| Conditions | Yield |
|---|---|
| With o-tetrachloroquinone; C16H21IMoO3 In 1,2-dichloro-ethane at 80℃; for 12h; Friedel-Crafts Alkylation; Inert atmosphere; Schlenk technique; Green chemistry; regioselective reaction; | 21.3% |
-
-
769-92-6
4-tert-Butylaniline

-
-
106-95-6
allyl bromide

-
A
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
B
-
27798-45-4
1-allyl-4-(tert-butyl)benzene

| Conditions | Yield |
|---|---|
| With tert.-butylnitrite In acetonitrile at 45 - 50℃; for 1.33h; | A n/a B 19% |


| Conditions | Yield |
|---|---|
| With o-tetrachloroquinone; C16H21IMoO3 In 1,2-dichloro-ethane at 80℃; for 12h; Friedel-Crafts Alkylation; Inert atmosphere; Schlenk technique; Green chemistry; regioselective reaction; | 17.2% |
| (i) AlCl3, MeNO2, (ii) /BRN= 1730872/; Multistep reaction; |

| Conditions | Yield |
|---|---|
| With phosphoric acid at 160 - 170℃; |
-
-
253185-03-4, 253185-04-5
tert-butylbenzene

-
A
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
B
-
7073-99-6
2-tert-butylbromobenzene

| Conditions | Yield |
|---|---|
| With thallium (III) oxide; trifluoroacetic acid; potassium bromide at 20℃; for 6h; | |
| With bromine In hexane at 25℃; for 0.0333333h; Darkness; | |
| With bromine; iodine |

| Conditions | Yield |
|---|---|
| With tetrachloromethane; bromine; iron |

| Conditions | Yield |
|---|---|
| With aluminium trichloride |

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid |
-
-
253185-03-4, 253185-04-5
tert-butylbenzene

-
-
7726-95-6
bromine

-
-
64-19-7
acetic acid

-
A
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
B
-
3972-64-3
1-bromo-3-(tertbutyl)benzene

-
C
-
7073-99-6
2-tert-butylbromobenzene

| Conditions | Yield |
|---|---|
| at 25℃; Kinetics; |

-
-
7601-90-3
perchloric acid

-
-
253185-03-4, 253185-04-5
tert-butylbenzene

-
A
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
B
-
3972-64-3
1-bromo-3-(tertbutyl)benzene

-
C
-
7073-99-6
2-tert-butylbromobenzene

| Conditions | Yield |
|---|---|
| at 25℃; Kinetics; |

-
-
253185-03-4, 253185-04-5
tert-butylbenzene

-
A
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
B
-
1014-60-4
1,3-di-tert-butylbenzene

-
C
-
3972-64-3
1-bromo-3-(tertbutyl)benzene

-
D
-
7073-99-6
2-tert-butylbromobenzene

| Conditions | Yield |
|---|---|
| With bromine at 25℃; for 20h; Irradiation; Yield given; |


| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 98 percent / pyridine / CH2Cl2 / 0.25 h / 0 °C 2: 86 percent / Et3N / PdCl2(dppf) / dioxane / Heating 3: 96 percent / aq. CuBr2 / methanol / Heating View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: H2SO4 2: AlCl3 / 45 °C 3: iron-turnings; tetrachloromethane; bromine View Scheme | |
| Multi-step reaction with 3 steps 1: aqueous H2SO4 2: AlCl3 / 45 °C 3: iron-turnings; tetrachloromethane; bromine View Scheme | |
| Multi-step reaction with 2 steps 1: aqueous H2SO4 2: iron-turnings; tetrachloromethane; bromine View Scheme | |
| Multi-step reaction with 2 steps 1: AlCl3 / 15 - 22 °C 2: iron-turnings; tetrachloromethane; bromine View Scheme | |
| Multi-step reaction with 2 steps 1: HCl; AlCl3 / 60 °C / Einleiten von Isobutylen 2: iron-turnings; tetrachloromethane; bromine View Scheme |
-
-
87199-16-4
3-Formylphenylboronic acid

-
-
3972-65-4
1-bromo-4-tert-butylbenzene
-
-
52436-75-6
(4-tert-butylphenyl)diazonium tetrafluoroborate

-
A
-
253185-03-4, 253185-04-5
tert-butylbenzene

-
B
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
C
-
701-30-4
1-tert-butyl-4-fluorobenzene

| Conditions | Yield |
|---|---|
| With trimethylsilyl bromide; 3-butyl-1-methyl-1H-imidazol-3-ium hexafluorophosphate at 20℃; | A 9 % Chromat. B 31 % Chromat. C 42 % Chromat. |

-
-
108-86-1
bromobenzene

-
-
507-19-7
t-butyl bromide

-
A
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
B
-
7073-99-6
2-tert-butylbromobenzene

| Conditions | Yield |
|---|---|
| With o-tetrachloroquinone; C16H21IMoO3 In 1,2-dichloro-ethane at 80℃; for 12h; Friedel-Crafts Alkylation; Inert atmosphere; Schlenk technique; Green chemistry; Overall yield = 25.1 %; regioselective reaction; |


| Conditions | Yield |
|---|---|
| Stage #1: 4-tert-Butylaniline With hydrogenchloride; tert.-butylnitrite In methanol; diethyl ether; N,N-dimethyl-formamide at 0℃; for 0.5h; Stage #2: With Bromotrichloromethane; tetrabutylammonium perchlorate In methanol; diethyl ether; N,N-dimethyl-formamide at 20℃; for 3h; Sandmeyer Reaction; Electrochemical reaction; | 53 %Chromat. |

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

| Conditions | Yield |
|---|---|
| With Bromotrichloromethane; [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate In ethyl acetate for 12h; Reagent/catalyst; Schlenk technique; Inert atmosphere; Irradiation; | 74 %Chromat. |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride / dichloromethane / 20 °C / Inert atmosphere; Schlenk technique 2: [4,4’-bis(1,1-dimethylethyl)-2,2’-bipyridine-N1,N1‘]bis [3,5-difluoro-2-[5-(trifluoromethyl)-2-pyridinyl-N]phenyl-C]iridium(III) hexafluorophosphate; Bromotrichloromethane / ethyl acetate / 12 h / Schlenk technique; Inert atmosphere; Irradiation View Scheme |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
16419-60-6
2-Methylphenylboronic acid

| Conditions | Yield |
|---|---|
| With potassium phosphate; palladium-incarcerated polystyrene-based copolymer; tris(o-methoxyphenyl)phosphine In water; toluene for 2h; Suzuki-Miyaura coupling; Heating; | 100% |
| With C25H29N3; potassium carbonate; palladium dichloride In water; N,N-dimethyl-formamide at 90℃; for 2h; Suzuki-Miyaura Coupling; | 99% |
| With dicyclohexyl-(2',6'-dimethoxybiphenyl-2-yl)-phosphane; potassium phosphate; palladium diacetate In toluene at 110℃; for 1.5h; Suzuki-Miyaura reaction; | 98% |

| Conditions | Yield |
|---|---|
| With potassium phosphate; phosphinated polymer incarcerated palladium catalyst In water; toluene for 4h; Suzuki-Miyaura coupling; Heating; | 100% |
| With potassium carbonate In water; isopropyl alcohol at 100℃; for 10h; Catalytic behavior; Suzuki-Miyaura Coupling; Inert atmosphere; | 100% |
| With potassium carbonate In water; isopropyl alcohol at 100℃; for 10h; Suzuki-Miyaura Coupling; Inert atmosphere; | 99% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
122-39-4
diphenylamine

-
-
36809-23-1
N,N-diphenyl-N-(4-tert-butylphenyl)amine

| Conditions | Yield |
|---|---|
| With bis(tri-t-butylphosphine)palladium(0); potassium tert-butylate In toluene at 20℃; for 4h; | 100% |
| With palladium diacetate; P(i-BuNCH2CH2)3N; sodium t-butanolate In toluene at 80℃; | 96% |
| for 3h; | 96% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; palladium diacetate; sodium t-butanolate In toluene for 12h; | 99.7% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; palladium diacetate; sodium t-butanolate In toluene for 12h; | 99.6% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

| Conditions | Yield |
|---|---|
| With tri-tert-butyl phosphine; palladium diacetate; sodium t-butanolate In toluene for 12h; | 99.6% |

| Conditions | Yield |
|---|---|
| Stage #1: poly(methacrylic acid) With sodium carbonate In water Stage #2: With palladium dichloride Stage #3: 1-bromo-4-tert-butylbenzene With 2,6-di-tert-butyl-4-methyl-phenol; sodium carbonate In water at 130 - 135℃; under 9000.9 - 12001.2 Torr; for 2h; Autoclave; Inert atmosphere; | 99.6% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
253185-03-4, 253185-04-5
tert-butylbenzene

| Conditions | Yield |
|---|---|
| With potassium phosphate In N,N-dimethyl-formamide; cyclohexanol at 110℃; for 12h; | 99% |
| With n-butyllithium; samarium diiodide; dipyrrolidinomethylaminophosphoric acid triamide; tert-butyl alcohol In tetrahydrofuran; hexane at -66 - 21℃; | 94% |
| With triethylamine In acetonitrile at 20℃; for 15h; Irradiation; Inert atmosphere; | 84% |

| Conditions | Yield |
|---|---|
| With tetraethylammonium tosylate; tetrakis(triphenylphosphine) palladium(0) In N,N-dimethyl-formamide Ambient temperature; electrolysis; | 99% |
| With DPEPhos; potassium tert-butylate; palladium diacetate; bis(pinacol)diborane for 12h; Heating; | 99% |
| With potassium carbonate In ethanol; water at 80℃; for 4h; Ullmann Condensation; Schlenk technique; Sealed tube; | 88% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
544-92-3
copper(I) cyanide

-
-
4210-32-6
4-tert-butyl benzonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 1-bromo-4-tert-butylbenzene; copper(I) cyanide In N,N-dimethyl-formamide for 8h; Heating; Stage #2: With hydrogenchloride; iron(III) chloride In water; N,N-dimethyl-formamide at 70℃; for 0.5h; | 99% |
| In N,N-dimethyl-formamide for 5h; Heating; | 71% |

| Conditions | Yield |
|---|---|
| With di-tert-butyl{2′-isopropoxy-[1,1′-binaphthalen]-2-yl}phosphane; sodium hydride; bis(dibenzylideneacetone)-palladium(0) In o-xylene at 110℃; for 18h; Inert atmosphere; | 99% |
| With caesium carbonate; copper(l) iodide In 1-methyl-pyrrolidin-2-one at 195℃; for 2h; microwave irradiation; | 89% |
| With potassium phosphate; copper(l) iodide; N,N′-bis(2-phenylphenyl)oxalamide In N,N-dimethyl-formamide at 90℃; for 24h; Schlenk technique; Inert atmosphere; Sealed tube; | 88% |
| With copper(I) trifluoromethanesulfonate benzene; 1-naphthalenecarboxylic acid; caesium carbonate; ethyl acetate In toluene at 110℃; | 81% |
-
-
110-91-8
morpholine

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
84736-47-0
4-(4-(tert-butyl)phenyl)morpholine

| Conditions | Yield |
|---|---|
| With sodium t-butanolate; 1-(2-di-isopropylphosphinophenyl)-3,5-diphenyl-1H-pyrazole; tris(dibenzylideneacetone)dipalladium (0) In toluene at 85℃; | 99% |
| With 5-(di-tert-butylphosphino)-1′, 3′, 5′-triphenyl-1′H-[1,4′]bipyrazole; potassium hydroxide; tris(dibenzylideneacetone)dipalladium (0) In tert-Amyl alcohol; water at 90℃; for 5h; | 99% |
| With palladium diacetate; P(i-BuNCH2CH2)3N; sodium t-butanolate In toluene at 80℃; | 99% |

| Conditions | Yield |
|---|---|
| With palladium diacetate; P(i-BuNCH2CH2)3N; sodium t-butanolate In toluene at 80℃; | 99% |
| With potassium tert-butylate; (ϖ-allyl)(diphosphinecyclobutene)palladium at 20℃; for 12h; | 97% |
| With tris-(dibenzylideneacetone)dipalladium(0); (S)-(1,1'-binaphthalene)-2,2'-diylbis(diphenylphosphine); sodium t-butanolate In toluene at 80℃; for 21h; Schlenk technique; Inert atmosphere; | 95% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
70729-05-4
1-Bromo-4-tert-butyl-2-nitro-benzene

| Conditions | Yield |
|---|---|
| With sulfuric acid; nitric acid at 20℃; for 12h; | 99% |
| With sulfuric acid; nitric acid | 95% |
| With sulfuric acid; nitric acid Inert atmosphere; | 95% |

| Conditions | Yield |
|---|---|
| With tris(dibenzylideneacetone)dipalladium (0); lithium hexamethyldisilazane; CyJohnPhos In tetrahydrofuran at 65℃; for 15h; | 99% |
| With copper(l) iodide; 2-carboxyquinoline N-oxide; potassium carbonate; ammonium hydroxide In dimethyl sulfoxide at 80℃; for 23h; Inert atmosphere; | 90% |
| With bis(tri-ortho-tolylphosphine)palladium(0); (R)-(-)-1-[(S)-2-(dicyclohexylphosphino)ferrocenyl]ethyl-di-tert-butylphosphine; ammonia; sodium t-butanolate In 1,4-dioxane at 80℃; for 6h; Inert atmosphere; | 88% |

| Conditions | Yield |
|---|---|
| With monophosphine 1,2,3,4,5-pentaphenyl-1'-(di-tert-butylphosphino)ferrocene; palladium compound; sodium t-butanolate In toluene at 100℃; for 4h; | 99% |
| With sodium t-butanolate; monophosphine 1,2,3,4,5-pentaphenyl-1'-(di-tert-butylphosphino)ferrocene; palladium diacetate In toluene | 99% |

| Conditions | Yield |
|---|---|
| With sodium t-butanolate; 1-(di-2-tert-butylphosphinophenyl)-2,5-diphenyl-1H-pyrrole; tris(dibenzylideneacetone)dipalladium (0) In toluene at 85℃; | 99% |
| With 5-(di-t-butylphosphanyl)-1-naphthalen-1-yl-1H-pyrazole; potassium hydroxide; tris(dibenzylideneacetone)dipalladium (0) In tert-Amyl alcohol; water at 90℃; | 90% |

| Conditions | Yield |
|---|---|
| With sodium t-butanolate; 1-(2-di-tert-butylphosphinophenyl)-3,5-diphenyl-1H-pyrazole; tris(dibenzylideneacetone)dipalladium (0) In tert-Amyl alcohol at 85℃; | 99% |
| With 5-(di-tert-butylphosphino)-1′, 3′, 5′-triphenyl-1′H-[1,4′]bipyrazole; potassium hydroxide; tris(dibenzylideneacetone)dipalladium (0) In tert-Amyl alcohol; water at 90℃; for 5h; | 99% |
| With sodium t-butanolate; dicyclohexyl[2-(1-phenylvinyl)phenyl]phosphane; palladium diacetate In toluene at 100℃; | 72% |

| Conditions | Yield |
|---|---|
| With tris(dibenzylideneacetone)dipalladium (0); 2,8,9-tribenzyl-2,3,8,9-tetraaza-1-phosphabicyclo[3,3,3]undecane; cesium fluoride In tetrahydrofuran at 20℃; for 10h; Stille cross-coupling; | 99% |
| With cesium fluoride; palladium diacetate In 1,4-dioxane at 100℃; for 3h; Stille cross-coupling; | 99% |
| With bis(tri-t-butylphosphine)palladium(0) In water at 20℃; for 4h; Stille Cross Coupling; Inert atmosphere; | 92% |

| Conditions | Yield |
|---|---|
| With 5-(di-t-butylphosphanyl)-1-naphthalen-1-yl-1H-pyrazole; potassium hydroxide; tris(dibenzylideneacetone)dipalladium (0) In tert-Amyl alcohol; water at 90℃; | 99% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
97302-05-1
di-4-methoxyphenylzinc

| Conditions | Yield |
|---|---|
| With catalyst 1 (aliphatic, phosphine based pincer complex of palladium) In 1,4-dioxane; 1-methyl-pyrrolidin-2-one at 100℃; for 0.25h; Negishi cross-coupling reaction; | 99% |
-
-
2116-65-6
4-benzyl pyridine

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
1393444-74-0
(4-tert-butylphenyl)(4-pyridyl)phenylmethane

| Conditions | Yield |
|---|---|
| With KN(SiMe3)2; palladium diacetate; nixantphos In cyclopentyl methyl ether at 24℃; for 12h; Inert atmosphere; | 99% |
| With BF4(1-)*C20H25N2O2(1+); palladium diacetate; sodium hexamethyldisilazane In toluene at 60℃; for 12h; Reagent/catalyst; Inert atmosphere; | 96% |
| With BF4(1-)*C20H25N2O2(1+); palladium diacetate; sodium hexamethyldisilazane In toluene at 60℃; for 16h; Reagent/catalyst; Solvent; Temperature; Inert atmosphere; | 95% |
| With C25H21N2O(1+)*Cl(1-); palladium diacetate; sodium hexamethyldisilazane In toluene at 60℃; for 12h; Reagent/catalyst; Inert atmosphere; | 89% |
| With bis(1,5-cyclooctadiene)nickel (0); sodium hexamethyldisilazane; nixantphos at 110℃; for 16h; | 80% |
-
-
101-82-6
2-Benzylpyridine

-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
1393444-73-9
(4-tert-butylphenyl)(2-pyridyl)phenylmethane

| Conditions | Yield |
|---|---|
| With diethylene glycol dimethyl ether; NiXantphos; palladium diacetate; sodium hexamethyldisilazane at 23℃; for 12h; Catalytic behavior; Reagent/catalyst; Glovebox; Inert atmosphere; Sealed tube; | 99% |
| With bis(1,5-cyclooctadiene)nickel (0); sodium hexamethyldisilazane; nixantphos at 20℃; for 16h; Catalytic behavior; Temperature; Concentration; Time; | 99% |
| With KN(SiMe3)2; palladium diacetate; nixantphos In cyclopentyl methyl ether at 24℃; for 12h; Inert atmosphere; | 96% |

| Conditions | Yield |
|---|---|
| With KN(SiMe3)2; palladium diacetate; nixantphos In cyclopentyl methyl ether at 24℃; for 12h; Inert atmosphere; | 99% |
| With bis(1,5-cyclooctadiene)nickel (0); sodium hexamethyldisilazane; nixantphos at 110℃; for 16h; | 81% |

| Conditions | Yield |
|---|---|
| With bis(1,5-cyclooctadiene)nickel (0); sodium hexamethyldisilazane; nixantphos at 50℃; for 16h; | 99% |
| With KN(SiMe3)2; palladium diacetate; nixantphos In tetrahydrofuran at 85℃; for 12h; Inert atmosphere; | 87% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
101-81-5
Diphenylmethane

-
-
26167-26-0
((4-(tert-butyl)phenyl)methylene)dibenzene

| Conditions | Yield |
|---|---|
| With 15-crown-5; NiXantphos; palladium diacetate; sodium hexamethyldisilazane at 23℃; for 12h; Catalytic behavior; Reagent/catalyst; Temperature; Solvent; Concentration; Glovebox; Inert atmosphere; Sealed tube; | 99% |
| With KN(SiMe3)2; palladium diacetate; nixantphos In cyclopentyl methyl ether at 24℃; for 12h; Inert atmosphere; | 95% |
| Stage #1: 1-bromo-4-tert-butylbenzene; Diphenylmethane With palladium diacetate; potassium hexamethylsilazane; nixantphos at 24℃; for 12h; Inert atmosphere; Glovebox; Stage #2: With depe at 24℃; for 0.666667h; | 91% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
611-45-0
1-Benzyl-naphthalene

-
-
1393444-66-0
(4-tert-butylphenyl)(1-naphthyl)phenylmethane

| Conditions | Yield |
|---|---|
| With KN(SiMe3)2; palladium diacetate; nixantphos In cyclopentyl methyl ether at 24℃; for 12h; Inert atmosphere; | 99% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
587-79-1
4-fluorodiphenylmethane

-
-
1393444-68-2
(4-tert-butylphenyl)(4-fluorophenyl)phenylmethane

| Conditions | Yield |
|---|---|
| With 1,1,4,7,10,10-hexamethyltriethylenetetramine; NiXantphos; palladium diacetate; sodium hexamethyldisilazane at 23℃; for 12h; Catalytic behavior; Reagent/catalyst; Glovebox; Inert atmosphere; Sealed tube; | 99% |
| With KN(SiMe3)2; palladium diacetate; nixantphos In cyclopentyl methyl ether at 24℃; for 12h; Inert atmosphere; | 96% |
-
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
13132-15-5
2-benzylthiophene

-
-
1393444-77-3
(4-tert-butylphenyl)(2-thienyl)phenylmethane

| Conditions | Yield |
|---|---|
| With KN(SiMe3)2; palladium diacetate; nixantphos In cyclopentyl methyl ether at 24℃; for 12h; Inert atmosphere; | 99% |
1-Bromo-4-tert-butylbenzene Chemical Properties
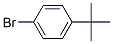
IUPAC Name:1-bromo-4-tert-butylbenzene
Molecular Formula:C10H13Br
Molecular Weight:213.114220 g/mol
Appearance:Colorless liquid
Melting Point:13-16 °C(lit.)
Boiling Point:80-81 °C2 mm Hg(lit.)
Flash Point:97 °C
Solubility:benzene, xylene, chlorobenzene and other solvents.
Insolubility:water
density:1.229 g/mL at 25 °C(lit.)
refractive index n20/D 1.533(lit.)
BRN:1859117
CAS DataBase Reference:3972-65-4(CAS DataBase Reference)
NIST Chemistry Reference:3972-65-4(NIST)
Synonyms of 1-Bromo-4-tert-butylbenzene(3972-65-4):
P-BROMO-T-BUTYLBENZENE;4-BROMO-TERT-BUTYLBENZENE;2-(4'-BROMOPHENYL)-2-METHYLPROPANE;1-BROMO-P-T-BUTYLBENZENE;1-BROMO-4-T-BUTYLBENZENE;1-BROMO-4-TERT-BUTYLBENZENE;P-BROMO-TERT-BUTYLBENZENE;4-TERT-BUTYLBROMOBENZENE
Categories of 1-Bromo-4-tert-butylbenzene(3972-65-4):
Halides;Phenyls & Phenyl-Het;Phenyls & Phenyl-Het;Aryl;C9 to C12;Halogenated Hydrocarbons;Bromine Compounds
1-Bromo-4-tert-butylbenzene Uses
1-Bromo-4-tert-butylbenzene(3972-65-4) is used as a intermediate for the synthesis of Miticide Fenazaquin.
1-Bromo-4-tert-butylbenzene Safety Profile
Safety Information of 1-Bromo-4-tert-butylbenzene(3972-65-4):
Hazard Codes : Xi 
Risk Statements:36/37/38
36/37/38:Irritating to eyes, respiratory system and skin
Safety Statements:23-24/25-37/39-26
23:Do not breathe gas/fumes/vapor/spray (appropriate wording to be specified by the manufacturer)
24/25:Avoid contact with skin and eyes
37/39:Wear suitable gloves and eye/face protection
26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
WGK Germany:3
Hazard Note:Irritant
HS Code:29036990
Related Products
- 1-Bromo-4-tert-butylbenzene
- 397308-78-0
- 3973-08-8
- 3973-17-9
- 3973-18-0
- 397328-49-3
- 397328-86-8
- 3973-62-4
- 39736-26-0
- 3973-70-4
- 39741-26-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View