-
Name
1-Octadecene
- EINECS 204-012-9
- CAS No. 112-88-9
- Article Data65
- CAS DataBase
- Density 0.787 g/cm3
- Solubility Miscible with alcohol, acetone and ether. Immiscible with water.
- Melting Point 14-16 °C(lit.)
- Formula C18H36
- Boiling Point 314.382 °C at 760 mmHg
- Molecular Weight 252.484
- Flash Point 148.889 °C
- Transport Information
- Appearance Clear colourless to amber liquid
- Safety 23-24/25
- Risk Codes 65
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms Octadecylene;alpha-Octadecene;n-1-Octadecene;NSC 66460;a-Octadecene;
- PSA 0.00000
- LogP 7.04380
Synthetic route

| Conditions | Yield |
|---|---|
| With polystyrene-supported(catecholato)oxoRe cat. act. by iPrOH; triphenylphosphine In toluene for 6h; Heating; | 97% |
| With 2,4,6-trimethyl-pyridine; bis(cyclopentadienyl)titanium dichloride; manganese; chloro-trimethyl-silane In tetrahydrofuran at 20℃; Inert atmosphere; | 94% |
| Multi-step reaction with 2 steps 1: sodium hydrogen telluride / ethanol / Heating 2: 92 percent / toluene-p-sulphonyl chloride, pyridine View Scheme | |
| With carbon monoxide; C29H32IrN5O; bis(trifluoromethane)sulfonimide lithium In benzene-d6 at 80℃; under 7500.75 Torr; for 24h; Schlenk technique; chemoselective reaction; | 71 %Spectr. |
-
-
105679-03-6
1-Tellanyl-octadecan-2-ol

-
-
112-88-9
octadec-1-ene

| Conditions | Yield |
|---|---|
| With pyridine; p-toluenesulfonyl chloride | 92% |

| Conditions | Yield |
|---|---|
| With 2,6-C6H3(CH2NMe2)2MnLiCl2 | 91% |

| Conditions | Yield |
|---|---|
| Stage #1: oleoyl alcohol With n-butyllithium; zirconocene dichloride In diethyl ether; hexane at -50 - 34℃; Stage #2: With hydrogenchloride In diethyl ether; hexane at 20℃; | 72% |
-
-
1121-30-8
1-hydroxy-2(1H)-pyridinethione

-
-
37850-75-2
allyl tert-butyl sulfide

-
A
-
24367-44-0
2-(2-tert-butyldisulfanyl)pyridine

-
B
-
112-88-9
octadec-1-ene

-
C
-
89025-53-6
n-pentadecyl-2'-pyridylsulphide

| Conditions | Yield |
|---|---|
| With dmap; n-hexadecanoyl chloride In toluene at 110℃; for 2.5h; | A 11% B 23% C 62% |

-
-
1121-30-8
1-hydroxy-2(1H)-pyridinethione

-
-
112-67-4
n-hexadecanoyl chloride

-
A
-
24367-44-0
2-(2-tert-butyldisulfanyl)pyridine

-
B
-
112-88-9
octadec-1-ene

-
C
-
89025-53-6
n-pentadecyl-2'-pyridylsulphide

| Conditions | Yield |
|---|---|
| With dmap; allyl tert-butyl sulfide In toluene at 110℃; for 2.5h; | A 11% B 23% C 62% |


| Conditions | Yield |
|---|---|
| With bis(cyclopentadienyl)titanium dichloride; water; 2,4,6-collidine hydrochloride In tetrahydrofuran at 20℃; for 24h; Inert atmosphere; | A 30% B 60% |
| With bis(cyclopentadienyl)titanium dichloride; manganese; water In tetrahydrofuran at 20℃; for 24h; Inert atmosphere; | A 42% B 20% |
-
-
3112-87-6
1-methyl-4-(prop-2-ene-1-sulfonyl)benzene

-
-
112-67-4
n-hexadecanoyl chloride

-
A
-
112-88-9
octadec-1-ene

-
B
-
89025-53-6
n-pentadecyl-2'-pyridylsulphide

| Conditions | Yield |
|---|---|
| With 1-hydroxy-2(1H)-pyridinethione; dmap In toluene at 110℃; for 2h; | A 21% B 55% |

-
-
14370-82-2
allyl phenyl selenide

-
-
112-67-4
n-hexadecanoyl chloride

-
A
-
112-88-9
octadec-1-ene

-
B
-
89025-53-6
n-pentadecyl-2'-pyridylsulphide

| Conditions | Yield |
|---|---|
| With 1-hydroxy-2(1H)-pyridinethione; dmap In toluene at 110℃; for 1.5h; | A 55% B 37% |

-
-
1121-30-8
1-hydroxy-2(1H)-pyridinethione

-
-
37850-75-2
allyl tert-butyl sulfide

-
-
112-67-4
n-hexadecanoyl chloride

-
A
-
112-88-9
octadec-1-ene

-
B
-
89025-53-6
n-pentadecyl-2'-pyridylsulphide

| Conditions | Yield |
|---|---|
| With dmap at 140℃; for 0.5h; Product distribution; Mechanism; reaction with other alkenes; | A 47% B 30% |

-
-
37850-75-2
allyl tert-butyl sulfide

-
-
112-67-4
n-hexadecanoyl chloride

-
A
-
112-88-9
octadec-1-ene

-
B
-
89025-53-6
n-pentadecyl-2'-pyridylsulphide

| Conditions | Yield |
|---|---|
| With 1-hydroxy-2(1H)-pyridinethione; dmap at 140℃; for 0.5h; | A 47% B 30% |

-
-
76126-87-9
3-(2,4,6-trimethylphenylthio)prop-1-ene

-
-
112-67-4
n-hexadecanoyl chloride

-
A
-
112-88-9
octadec-1-ene

-
B
-
89025-53-6
n-pentadecyl-2'-pyridylsulphide

| Conditions | Yield |
|---|---|
| With 1-hydroxy-2(1H)-pyridinethione; dmap In toluene at 110℃; for 2h; | A 25% B 45% |

-
-
91-63-4
2-methylquinoline

-
-
34184-41-3
4-Chlorobenzolsulfonsaeure-octadecylester

-
A
-
6297-03-6
19-oxaheptatriacontane

-
B
-
112-88-9
octadec-1-ene

| Conditions | Yield |
|---|---|
| Stage #1: 2-methylquinoline; 4-Chlorobenzolsulfonsaeure-octadecylester at 140℃; for 5h; Stage #2: With potassium iodide In acetonitrile | A n/a B n/a C n/a D 40% |

-
-
637-69-4
4-Methoxystyrene

-
-
930-02-9
octadecyl-vinyl ether

-
A
-
112-92-5
1-octadecanol

-
B
-
112-88-9
octadec-1-ene

-
D
-
56047-51-9
4-(4-methoxyphenyl)butyraldehyde

| Conditions | Yield |
|---|---|
| With 10-phenyl-9-(2,4,6-trimethylphenyl)acridinium tetrafluoroborate; diphenyldisulfane In 1,2-dichloro-ethane Inert atmosphere; Irradiation; chemoselective reaction; | A 6% B 40% C 14% D 30% |


| Conditions | Yield |
|---|---|
| With tetrabutylammonium tetrafluoroborate In N,N-dimethyl-formamide Electrochemical reaction; Inert atmosphere; | A 33% B 28% C 39% |

-
-
1006-99-1
2-methyl-5-chloro-benzothiazole

-
-
34184-41-3
4-Chlorobenzolsulfonsaeure-octadecylester

-
A
-
6297-03-6
19-oxaheptatriacontane

-
B
-
112-88-9
octadec-1-ene

| Conditions | Yield |
|---|---|
| for 5h; Heating; | A n/a B n/a C n/a D 37% |

-
-
32894-70-5
allyl 4-nitrophenyl sulfide

-
-
112-67-4
n-hexadecanoyl chloride

-
A
-
112-88-9
octadec-1-ene

-
B
-
89025-53-6
n-pentadecyl-2'-pyridylsulphide

| Conditions | Yield |
|---|---|
| With 1-hydroxy-2(1H)-pyridinethione; dmap In toluene at 110℃; for 1.5h; | A 12% B 27% |

-
-
5296-64-0
allyl phenyl thioether

-
-
112-67-4
n-hexadecanoyl chloride

-
A
-
112-88-9
octadec-1-ene

-
B
-
89025-53-6
n-pentadecyl-2'-pyridylsulphide

| Conditions | Yield |
|---|---|
| With 1-hydroxy-2(1H)-pyridinethione; dmap In toluene at 110℃; for 1.5h; | A 20% B 22% |

-
-
69113-52-6
3‐(2‐ethylpropane‐2‐sulfinyl)prop‐1‐ene

-
-
112-67-4
n-hexadecanoyl chloride

-
-
112-88-9
octadec-1-ene

| Conditions | Yield |
|---|---|
| With 1-hydroxy-2(1H)-pyridinethione; dmap In toluene at 110℃; for 1h; | 7% |

| Conditions | Yield |
|---|---|
| With 1-hydroxy-2(1H)-pyridinethione; dmap In toluene at 20℃; for 16h; darkness; | 3% |

| Conditions | Yield |
|---|---|
| With phosphoric acid at 100℃; entsteht vermutlich im Gemisch mit Isomeren; | |
| With phosphoric acid; pyrographite at 280 - 300℃; under 25 Torr; entsteht vermutlich im Gemisch mit Isomeren; | |
| With phosphoric acid at 280 - 300℃; under 25 Torr; entsteht vermutlich im Gemisch mit Isomeren; |
-
-
408321-28-8
2-ethoxy-1-bromo-octadecane

-
-
112-88-9
octadec-1-ene

| Conditions | Yield |
|---|---|
| With i-Amyl alcohol; zinc |

| Conditions | Yield |
|---|---|
| at 520 - 525℃; thermische Zersetzung an Glaswolle; |

| Conditions | Yield |
|---|---|
| under 120 - 140 Torr; Destillation; |

| Conditions | Yield |
|---|---|
| under 120 - 140 Torr; |

| Conditions | Yield |
|---|---|
| at 300℃; |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In dimethyl sulfoxide; benzene |

| Conditions | Yield |
|---|---|
| With quinoline; hydrogen; Lindlar's catalyst In Petroleum ether |

| Conditions | Yield |
|---|---|
| at 350 - 400℃; |
-
-
3386-32-1
n-octadecyl p-toluenesulfonate

-
-
865-47-4
potassium tert-butylate

-
A
-
112-88-9
octadec-1-ene

-
B
-
61548-84-3
1-tert-butoxy-octadecane

| Conditions | Yield |
|---|---|
| In dimethyl sulfoxide; benzene |

-
-
2426-02-0
3,4,5,6-Tetrahydrophthalic anhydride

-
-
112-88-9
octadec-1-ene

-
-
85672-82-8
10-Hexadecyl-8-oxa-tricyclo[4.3.2.01,6]undecane-7,9-dione

| Conditions | Yield |
|---|---|
| In cyclohexene for 20h; Heating; Irradiation; | 100% |

| Conditions | Yield |
|---|---|
| With fac-[Mn(1,2-bis(di-isopropylphosphino)ethane)(CO)3(CH2CH2CH3)]; hydrogen In diethyl ether at 25℃; under 37503.8 Torr; for 18h; | 99% |
| With platinum(IV) oxide under 2206.5 Torr; Hydrogenation; | |
| With triethylsilane; palladium dichloride In ethanol at 20℃; for 24h; | 96 % Chromat. |
-
-
112-88-9
octadec-1-ene

-
-
26038-78-8
1,2-dibromooctadecane

| Conditions | Yield |
|---|---|
| With bromine In dichloromethane at 20℃; for 1h; | 99% |
| With poly(diallyldimethylammonium chloride) supported tribromide for 0.833333h; | 80% |
| With bromine |

| Conditions | Yield |
|---|---|
| With platinum on carbon nanotubes In neat (no solvent) at 20℃; for 24h; | 99% |
| dihydrogen hexachloroplatinate In diethyl ether | 85% |
| dihydrogen hexachloroplatinate | |
| With dihydrogen hexachloroplatinate | |
| With SiliaCat Pt(0) - mesoporous organosilica microspheres doped with Pt nanoparticles In neat (no solvent) at 65℃; for 1h; | 91 %Spectr. |

| Conditions | Yield |
|---|---|
| With palladium diacetate In N,N-dimethyl-formamide at 130℃; for 0.0333333h; Schlenk technique; Microwave irradiation; regioselective reaction; | 99% |

| Conditions | Yield |
|---|---|
| With platinum(0)-1,3-divinyl-1,1,3,3-tetramethyldisiloxane complex at 80℃; | 99% |

| Conditions | Yield |
|---|---|
| With platinum on carbon nanotubes In neat (no solvent) at 20℃; for 24h; | 98% |
| With SiliaCat Pt(0) - mesoporous organosilica microspheres doped with Pt nanoparticles In neat (no solvent) at 65℃; for 1h; | 86 %Spectr. |

| Conditions | Yield |
|---|---|
| With [1,3-bis(2,4,6-trimethylphenyl)-2-imidazolidinylidene]dichloro-(3-phenyl-1H-inden-1-ylidene)(tricyclohexylphosphine)ruthenium(II) In dichloromethane at 80℃; for 15h; Cross Metathesis; Inert atmosphere; Sealed tube; diastereoselective reaction; | 98% |
-
-
112-88-9
octadec-1-ene

-
-
762-04-9
phosphonic acid diethyl ester

-
-
16165-72-3
octadecylphosphonate de diethyle

| Conditions | Yield |
|---|---|
| With di-tert-butyl peroxide at 135℃; for 6h; | 97% |
-
-
112-88-9
octadec-1-ene

-
-
36587-77-6
2-octadecene

| Conditions | Yield |
|---|---|
| With trimethylaluminum; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene; cobalt acetylacetonate In N,N-dimethyl acetamide at 90℃; for 16h; | 97% |
| With [(2,6-iPrC6H3NC(Me)C(Me)N-2,6-iPrC6H3)Pd(CH3)(Cl)]; sodium tetrakis[(3,5-di-trifluoromethyl)phenyl]borate; acetonitrile In chlorobenzene at 25℃; for 3h; Darkness; Inert atmosphere; | 53% |
| With selenium at 200℃; for 20h; Reagent/catalyst; Inert atmosphere; | |
| With trimethylaluminum; cobalt(II) aceylacetonate; 4,5-bis(diphenylphosphino)-9,9-dimethylxanthene In N,N-dimethyl acetamide; toluene at 90℃; for 16h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With N-Bromosuccinimide; 1,8-diazabicyclo[5.4.0]undec-7-ene In 1,2-dichloro-ethane at 50℃; | 96% |

-
-
112-88-9
octadec-1-ene

-
-
105-74-8
dilauryl peroxide

-
-
104-15-4
toluene-4-sulfonic acid

-
-
839-90-7
1,3,5-tris(2-hydroxyethyl)-S-triazine-2,4,6-trione

-
-
107-96-0
3-mercaptopropionic acid

| Conditions | Yield |
|---|---|
| In water; toluene | 94.6% |


| Conditions | Yield |
|---|---|
| With di-tert-butyl peroxide at 135℃; for 8h; | 94% |
| With 2,2'-azobis(isobutyronitrile) In benzene |

| Conditions | Yield |
|---|---|
| In neat (no solvent) HSiCl3 and 1-octadecene at about 300°C under pressure;; | 94% |
| In neat (no solvent) HSiCl3 and octadecene at 300°C under pressure;; | 94% |
| In neat (no solvent) HSiCl3 and 1-octadecene at about 300°C under pressure;; | 94% |
| In neat (no solvent) HSiCl3 and octadecene at 300°C under pressure;; | 94% |
| With SiliaCat Pt(0) - mesoporous organosilica microspheres doped with Pt nanoparticles In neat (no solvent) at 65℃; for 1h; | 87 %Spectr. |
-
-
507-63-1
1-iodoheptadecafluorooctane

-
-
112-88-9
octadec-1-ene

-
-
176979-19-4
1,1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8-heptadecafluoro-10-iodohexacosane

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile) at 80℃; Inert atmosphere; | 94% |

| Conditions | Yield |
|---|---|
| Stage #1: octadec-1-ene With diethylenetriaminepentaacetic acid (DTPA)-functionalized silica supported Pt at 60℃; for 0.5h; Stage #2: Dichloromethylsilane at 60℃; for 4h; Reagent/catalyst; | 93.5% |
| at 300℃; | |
| dihydrogen hexachloroplatinate In diethyl ether; isopropyl alcohol at 60℃; for 12h; |

| Conditions | Yield |
|---|---|
| With C24H30ClN3NiO2; sodium t-butanolate In tetrahydrofuran at 20℃; for 6h; Inert atmosphere; Glovebox; | 93% |
-
-
67-56-1
methanol

-
-
112-88-9
octadec-1-ene

-
-
201230-82-2
carbon monoxide

-
-
29238-06-0
dimethyl 2-hexadecylsuccinate

| Conditions | Yield |
|---|---|
| With copper(l) chloride; palladium on activated charcoal In tetrahydrofuran at 25℃; under 760 Torr; for 216h; | 92% |
| With copper(l) chloride; palladium on activated charcoal In tetrahydrofuran at 25℃; under 760 Torr; for 216h; | 92% |


| Conditions | Yield |
|---|---|
| With sodium hypophosphite monohydrate; 2,2'-azobis(isobutyronitrile); sulfuric acid In ethanol for 26h; Inert atmosphere; Reflux; | 91% |

| Conditions | Yield |
|---|---|
| With dihydrogen hexachloroplatinate In isopropyl alcohol at 80℃; for 3h; | 91% |
-
-
112-88-9
octadec-1-ene

-
-
4142-85-2
1,1,1,3,3,3-hexachloro-1,3-disilapropane

-
-
1621184-16-4
1,1,1,3,3-pentachloro-1,3-disilaheneicosane

| Conditions | Yield |
|---|---|
| Stage #1: 1,1,1,3,3,3-hexachloro-1,3-disilapropane With trihexyl(tetradecyl)phosphonium chloride; Dichloromethylsilane at 80 - 90℃; Inert atmosphere; Autoclave; Stage #2: octadec-1-ene With dihydrogen hexachloroplatinate In tetrahydrofuran at 90 - 110℃; Inert atmosphere; | 91% |

| Conditions | Yield |
|---|---|
| With rhodium(III) chloride trihydrate at 120℃; for 12h; | 89.5% |
-
-
112-88-9
octadec-1-ene

-
-
423-62-1
1-Iodo-perfluorodecane

-
-
1257261-90-7
1,1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,9,9,10,10-henicosafluoro-12-iodooctacosane

| Conditions | Yield |
|---|---|
| With 2,2'-azobis(isobutyronitrile) at 80℃; Inert atmosphere; | 89% |

| Conditions | Yield |
|---|---|
| Stage #1: methanol; octadec-1-ene With mercury(II) diacetate at 0℃; Stage #2: With sodium In methanol at 0℃; | A n/a B 87% |

-
-
950665-39-1
(R)-tridec-1-en-4-yl (2R)-2-methoxy-2-phenylacetate

-
-
112-88-9
octadec-1-ene

-
-
950665-40-4
nonacos-12-en-10-yl 2-methoxy-2-phenylacetate

| Conditions | Yield |
|---|---|
| 1,3-bis(2,4,6-triMePh)imidazole-2yl-(Cl2)Ru(PCy3)(=CH-Ph) In dichloromethane at 45℃; for 4h; Heating; | 87% |
-
-
112-88-9
octadec-1-ene

| Conditions | Yield |
|---|---|
| With 1,1'-binaphthalene-2,2'-diamine; oxygen; palladium diacetate; sodium hydrogencarbonate; p-benzoquinone In 1,2-dichloro-ethane at 100℃; for 24h; | A n/a B 86% |


| Conditions | Yield |
|---|---|
| Stage #1: octadec-1-ene With Wilkinson's catalyst for 0.0833333h; Stage #2: Triethoxysilane at 90℃; for 5h; | 85.8% |
| With dihydrogen hexachloroplatinate(IV) hexahydrate In isopropyl alcohol at 140℃; for 18h; Catalytic behavior; Time; Solvent; Temperature; Inert atmosphere; regioselective reaction; | 68% |
| With SiliaCat Pt(0) - mesoporous organosilica microspheres doped with Pt nanoparticles In neat (no solvent) at 75℃; for 2h; Temperature; | 98 %Spectr. |
| With platinum(II) with naphthalenolimine and cyclo-1,5-octadiene binary ligands anchored onto mesoporous silica SBA-15 support catalyst In neat (no solvent) at 60℃; for 2h; Catalytic behavior; Temperature; Sealed tube; regioselective reaction; | |
| With SiO2-coated nano-Fe3O4-supported biIMI-PtCl2 at 60℃; for 2h; regioselective reaction; |
1-Octadecene Specification
The CAS registry number of 1-Octadecene is 112-88-9. The IUPAC name is octadec-1-ene. In addition, it is a long-chain hydrocarbon and an alkene with the molecular formula C18H36. It is a relatively inexpensive solvent and is compatible with oleic acid. What's more, it is a clear colourless to amber liquid which can be used in the synthesis of colloidal quantum dots.
Physical properties about 1-Octadecene are: (1)ACD/LogP: 9.81; (2)# of Rule of 5 Violations: 1; (3)ACD/LogD (pH 5.5): 9.81; (4)ACD/LogD (pH 7.4): 9.81; (5)ACD/BCF (pH 5.5): 1000000; (6)ACD/BCF (pH 7.4): 1000000; (7)ACD/KOC (pH 5.5): 5170237.5; (8)ACD/KOC (pH 7.4): 5170237.5; (9)#Freely Rotating Bonds: 15; (10)Index of Refraction: 1.443; (11)Molar Refractivity: 85.16 cm3; (12)Molar Volume: 320.7 cm3; (13)Polarizability: 33.76×10-24cm3; (14)Surface Tension: 27.9 dyne/cm; (15)Density: 0.787 g/cm3; (16)Flash Point: 148.9 °C; (17)Enthalpy of Vaporization: 53.34 kJ/mol; (18)Boiling Point: 314.4 °C at 760 mmHg; (19)Vapour Pressure: 0.000863 mmHg at 25°C.
Preparation of 1-Octadecene: this chemical can be prepared by 1-tellanyl-octadecan-2-ol. This reaction will need reagents toluene-p-sulphonyl chloride and pyridine. The yield is about 92%.
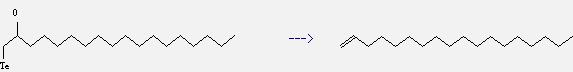
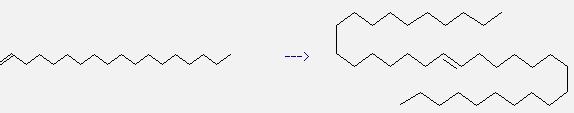
Uses of 1-Octadecene: it can be used to produce (E)-17-tetratriacontene. This reaction will need reagents WCl6, Sn(CH3)4 and ethyl acetate. The reaction time is 35 hours at temperature of 80 °C. And the yield is about 49%.
When you are using this chemical, please be cautious about it as the following:
This chemical may irritate eyes and skin, and may cause lung damage if swallowed. When you are using it, do not breathe gas/fumes/vapour/spray (appropriate wording to be specified by the manufacturer) and avoid contact with skin and eyes.
You can still convert the following datas into molecular structure:
(1)SMILES: C=C\CCCCCCCCCCCCCCCC
(2)Std. InChI: InChI=1S/C18H36/c1-3-5-7-9-11-13-15-17-18-16-14-12-10-8-6-4-2/h3H,1,4-18H2,2H3
(3)Std. InChIKey: CCCMONHAUSKTEQ-UHFFFAOYSA-N
Related Products
- 1-Octadecene
- 112889-02-8
- 112-89-0
- 112892-88-3
- 112897-97-9
- 112897-99-1
- 112898-00-7
- 112898-06-3
- 112898-10-9
- 112898-18-7
- 112898-19-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View