-
Name
1H-Benzotriazole
- EINECS 202-394-1
- CAS No. 95-14-7
- Article Data127
- CAS DataBase
- Density 1.348 g/cm3
- Solubility 25 g/l in water
- Melting Point 97-99 °C(lit.)
- Formula C6H5N3
- Boiling Point 276.1 °C at 760 mmHg
- Molecular Weight 119.126
- Flash Point 133.9 °C
- Transport Information
- Appearance yellow to beige solid
- Safety 26-36/37-61-45-36/37/39-28A
- Risk Codes 20/22-36-52/53-5-36/37/38-20/21/22-11
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi,
Xi, F
F
- Synonyms M 318;NSC 3058;RusminR;Seetec BT;Seetec BT-R;Verzone Crystal;Benzotriazole;1,2,3-Benzotriazole (BTA);1,2,3-1H-Benzotriazole;1,2,3-Benzotriazole;1,2,3-Triaza-1H-indene;1,2,3-Triazaindene;1,2-Aminoazophenylene;1H-1,2,3-Benzotriazole;2,3-Diazaindole;Azimidobenzene;Aziminobenzene;BLS 1326;BT 120 (lubricant additive);BTA;Benzeneazimide;C.V.I. Liquid;CVI;Cobratec 35G;Cobratec 99;D32-108;Entek;HY 2;ISK 3;Irgastab I 489;Kemitec TT;
- PSA 41.57000
- LogP 0.95790
Synthetic route

| Conditions | Yield |
|---|---|
| In 1,2-dichloro-ethane at 20 - 25℃; for 2h; | A 100% B 93% |

-
-
3886-69-9
(R)-1-phenyl-ethyl-amine

-
-
183266-61-7
1-(trifluoroacetyl)benzotriazole

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
2354-91-8, 28332-81-2, 39995-50-1, 39995-51-2
N-<(R)-(+)-α-phenylethyl>-2,2,2-trifluoroacetamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 4h; Ambient temperature; | A n/a B 100% |


-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
4714-21-0, 130059-91-5, 130059-95-9, 1657-45-0, 1860-17-9
E-1-methyl-4-styryl-benzene

| Conditions | Yield |
|---|---|
| With sodium naphthalenide In tetrahydrofuran for 25h; Heating; | A 100% B 71% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; sodium nitrite In methanol | 99.9% |
| With K10 Montmorillonite Clay; sodium nitrite In neat (no solvent, solid phase) at 110℃; for 1h; Catalytic behavior; Solvent; Temperature; Time; Microwave irradiation; Green chemistry; | 99% |
| With sodium nitrite In water at 260℃; for 3h; Autoclave; Inert atmosphere; Large scale; | 98.4% |
-
-
117067-51-3
N-(1-Benzotriazol-1-yl-2-methyl-propyl)-thiobenzamide

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
19090-58-5
N-isobutylthiobenzamide

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In tetrahydrofuran for 0.5h; Heating; | A n/a B 99% |
-
-
117067-50-2
N-(1-Benzotriazol-1-yl-nonyl)-benzamide

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
119020-89-2
N-nonylbenzamide

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 3h; Heating; | A n/a B 99% |
-
-
130384-98-4
tert-butyl 1H-benzo[d][1,2,3]triazole-1-carboxylate

-
-
95-14-7
1,2,3-Benzotriazole

| Conditions | Yield |
|---|---|
| With water at 100℃; for 0.0833333h; | 99% |
-
-
119020-88-1
N-(1-benzotriazol-1-yl-1-phenylmethyl)acetamide

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
588-46-5
N-(phenylmethyl)acetamide

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 3h; Heating; | A n/a B 98% |
-
-
117067-55-7
N-(benzotriazol-1-ylmethyl)thiobenzoamide

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
5310-14-5
N-methylthiobenzamide

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In tetrahydrofuran for 0.5h; Heating; | A n/a B 98% |
-
-
117759-81-6
N-<α-(benzotriazol-1-yl)nonyl>thiobenzamide

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
117759-91-8
N-Nonyl-thiobenzamide

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In tetrahydrofuran for 0.5h; Heating; | A n/a B 97% |
-
-
119020-86-9
N-(1-Benzotriazol-1-yl-hexyl)-benzamide

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
4773-75-5
N-hexylbenzamide

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 3h; Heating; | A n/a B 97% |
-
-
96-15-1
2-Methylbutylamine

-
-
183266-61-7
1-(trifluoroacetyl)benzotriazole

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
14618-17-8
N-(2-methylbutyl)-2,2,2-trifluoroacetamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 3h; Ambient temperature; | A n/a B 97% |

-
-
128767-05-5
benzo-1,2,3,4-tetrazine 1,3-dioxide

-
-
95-14-7
1,2,3-Benzotriazole

| Conditions | Yield |
|---|---|
| With tin(ll) chloride In ethanol; ethyl acetate at 20℃; | 97% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In water at 80℃; for 4h; | 97% |
| With sodium hydride In N,N-dimethyl acetamide at 60℃; for 2h; Inert atmosphere; | 81% |
| With potassium tert-butylate In dimethyl sulfoxide at 20℃; for 1h; Inert atmosphere; Darkness; Schlenk technique; | 67% |
| With cetyltrimethylammonim bromide; potassium hydroxide In tetrahydrofuran; water at 20℃; for 0.666667h; Green chemistry; |
-
-
111184-75-9
N-(1H-benzotriazol-1-ylmethyl)benzamide

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
88070-48-8
N-methylbenzamide

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 3h; Heating; | A n/a B 96% |
-
-
117067-46-6
N-(1-Benzotriazol-1-yl-2-methyl-propyl)-benzamide

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
5705-57-7
N-isobutyl-benzamide

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 3h; Heating; | A n/a B 96% |
-
-
117067-47-7
N-(1-Benzotriazol-1-yl-butyl)-benzamide

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
2782-40-3
N-butylbenzamide

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 3h; Heating; | A n/a B 96% |

| Conditions | Yield |
|---|---|
| With zinc phthalocyanine; hydrazine hydrate In PEG-400 at 100℃; for 8h; | 96% |

| Conditions | Yield |
|---|---|
| Stage #1: benzotriazol-1-ol With triethylamine In acetonitrile at 20℃; for 0.5h; Stage #2: With tetrahydroxydiboron In acetonitrile at 50℃; for 0.5h; Catalytic behavior; Reagent/catalyst; Temperature; Solvent; | 95% |
| With 2,3-dimethyl-2,3-butane diol In N,N-dimethyl acetamide at 120℃; for 0.25h; Microwave irradiation; Sealed tube; Green chemistry; | 82% |
| With phosphorus; hydrogen iodide at 140 - 150℃; |
-
-
117759-80-5
N-<α-(benzotriazol-1-yl)octyl>thiobenzamide

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
117759-90-7
N-octylbenzothioamide

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In tetrahydrofuran for 0.5h; Heating; | A n/a B 95% |
-
-
75-64-9
tert-butylamine

-
-
183266-61-7
1-(trifluoroacetyl)benzotriazole

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
1960-29-8
N-tert-butyl-2,2,2-trifluoroethanamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 3h; Ambient temperature; | A n/a B 95% |

-
-
183266-61-7
1-(trifluoroacetyl)benzotriazole

-
-
100-51-6
benzyl alcohol

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
351-70-2
benzyl trifluoroacetate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 3h; Heating; | A n/a B 95% |

-
-
92133-96-5
(1H-benzo[d][1,2,3]triazol-1-yl)(3-chlorophenyl)methanone

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
35190-07-9
1,2-bis(3-chlorophenyl)ethane-1,2-dione

| Conditions | Yield |
|---|---|
| With samarium diiodide In tetrahydrofuran at 20℃; for 0.0833333h; | A n/a B 95% |
-
-
313225-01-3
(1H-benzo[d][1,2,3]triazol-1-yl)(4-bromophenyl)methanone

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
35578-47-3
4,4'-dibromobenzil

| Conditions | Yield |
|---|---|
| With samarium diiodide In tetrahydrofuran at 20℃; for 0.0833333h; | A n/a B 95% |

| Conditions | Yield |
|---|---|
| Stage #1: acetic acid; 1,2-diamino-benzene With sodium nitrite In water at 8 - 87℃; for 0.0583333h; Stage #2: at -20℃; Temperature; | 95% |
-
-
54548-46-8
(2R,3R,4R,5R)-4-(acetyloxty)-2-[(acetyloxy)methyl]-5-(1H-1,2,3-benzotriazol-1-yl)tetrahydro-3-furanyl acetate

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
13035-61-5
1,2,3,5-tetraacetylribose

| Conditions | Yield |
|---|---|
| With acetic anhydride; trifluoroacetic acid at 95℃; | A n/a B 94.3% |
-
-
117067-48-8
1-benzotriazolyl-N-benzoyl-1-phenylmethylamine

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
1485-70-7
N-benzylbenzamide

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate In ethanol for 3h; Heating; | A n/a B 94% |
-
-
106-48-9
4-chloro-phenol

-
-
183266-61-7
1-(trifluoroacetyl)benzotriazole

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
658-74-2
4-chlorophenyl trifluoroacetate

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 6h; Heating; | A n/a B 94% |


-
A
-
95-14-7
1,2,3-Benzotriazole

| Conditions | Yield |
|---|---|
| With naphthalen-1-yl-lithium In tetrahydrofuran for 0.0833333h; further reagent sodium naphthalenide; | A 94% B 61% |
-
-
91-59-8
naphthalen-2-ylamine

-
-
183266-61-7
1-(trifluoroacetyl)benzotriazole

-
A
-
95-14-7
1,2,3-Benzotriazole

-
B
-
398-48-1
N-(2-naphthyl)-2,2,2-trifluoroacetamide

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 3h; Ambient temperature; | A n/a B 93% |


| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 0.5h; Cooling; | 100% |
| With triethylamine In dichloromethane at 0℃; for 0.5h; Claisen Condensation; Inert atmosphere; | 96% |
| With triethylamine In dichloromethane for 0.5h; Ambient temperature; | 95% |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
50-00-0
formaldehyd

-
-
1120-48-5
n-dioctylamine

-
-
120803-37-4
1-(benzotriazol-1'-yl)methyl-N,N-dioctylamine

| Conditions | Yield |
|---|---|
| 1.) methanol, water, 7 h, 2.) Et2O, reflux, overnight; | 100% |


| Conditions | Yield |
|---|---|
| at 25℃; | 100% |

| Conditions | Yield |
|---|---|
| at 25℃; | 100% |

| Conditions | Yield |
|---|---|
| at 25℃; | 100% |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
131543-46-9
Glyoxal

-
-
111507-88-1
meso-1,2-Di(benzotriazol-1-yl)ethane-1,2-diol

| Conditions | Yield |
|---|---|
| sulfuric acid In water; acetic acid at 25℃; for 24h; | 100% |
| With acetic acid In water at 20℃; | 96% |
| With sulfuric acid; acetic acid In water at 25 - 70℃; for 24h; |

| Conditions | Yield |
|---|---|
| at 25℃; | 100% |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
630-19-3
pivalaldehyde

-
-
111507-83-6
1-Benzotriazol-1-yl-2,2-dimethyl-propan-1-ol

| Conditions | Yield |
|---|---|
| at 25℃; | 100% |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
123-72-8
butyraldehyde

-
-
111098-58-9
1-(1H-Benzotriazol-1-yl)-1-chlorobutane

| Conditions | Yield |
|---|---|
| With thionyl chloride In chloroform for 0.5h; Heating; | 100% |
| With thionyl chloride | |
| With thionyl chloride | |
| With thionyl chloride 1.) benzene, r.t., 0.5 h, 2.) r.t. 2 h; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| at 25℃; | 100% |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
638-29-9
n-valeryl chloride

-
-
84298-28-2
1H-1,2,3-benzotriazol-1-yl(n-butyl)methanone

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 0.5h; Cooling; | 100% |
| With triethylamine In dichloromethane cooling; | 91% |
| at 80 - 100℃; for 0.25h; | 79% |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
78-84-2
isobutyraldehyde

-
-
111507-81-4
1-Benzotriazol-1-yl-2-methyl-propan-1-ol

| Conditions | Yield |
|---|---|
| at 25℃; | 100% |

| Conditions | Yield |
|---|---|
| In benzene Ambient temperature; | 100% |

-
-
95-14-7
1,2,3-Benzotriazole

-
-
407-25-0
trifluoroacetic anhydride

-
-
183266-61-7
1-(trifluoroacetyl)benzotriazole

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 0.5h; Ambient temperature; | 100% |
| In tetrahydrofuran |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
66176-39-4
4-(bromomethyl)benzene-1-sulfonyl chloride

| Conditions | Yield |
|---|---|
| With triethylamine In diethyl ether at 20℃; for 3h; Substitution; | 100% |
-
-
95-14-7
1,2,3-Benzotriazole

-
-
124-63-0
methanesulfonyl chloride

-
-
37073-15-7
1-(methanesulfonyl)-1H-1,2,3-benzotriazole

| Conditions | Yield |
|---|---|
| With pyridine In toluene at 0 - 20℃; Inert atmosphere; | 100% |
| With pyridine In toluene at 0 - 20℃; | 93.8% |
| With pyridine In toluene at 20℃; Inert atmosphere; Cooling with ice; | 93% |
-
-
110-91-8
morpholine

-
-
95-14-7
1,2,3-Benzotriazole

-
-
66-99-9
β-naphthaldehyde

-
-
433232-15-6
[1-(4-morpholino)-1-(2-naphthyl)]methyl-1H-1,2,3-benzotriazole

| Conditions | Yield |
|---|---|
| In ethanol at 20℃; for 15h; | 100% |

-
-
95-14-7
1,2,3-Benzotriazole

-
-
62-23-7
4-nitro-benzoic acid

-
-
4231-71-4
1H-1,2,3-benzotriazol-1-yl-4-nitrophenylmethanone

| Conditions | Yield |
|---|---|
| With thionyl chloride In dichloromethane at 20℃; for 8.5h; | 100% |
| With thionyl chloride In dichloromethane at 20℃; | 91% |
| With thionyl chloride In dichloromethane at 20℃; for 2h; | 91.4% |

-
-
95-14-7
1,2,3-Benzotriazole

-
-
56876-11-0
α-Phthalimido-β-(3-methoxyphenyl)-propionamid

-
-
872690-16-9
N-[benzotriazol-1-yl-(4-bromo-phenyl)-methyl]-2-(1,3-dioxo-1,3-dihydro-isoindol-2-yl)-3-(3-methoxy-phenyl)-propionamide

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 100% |

-
-
95-14-7
1,2,3-Benzotriazole

-
-
56876-11-0
α-Phthalimido-β-(3-methoxyphenyl)-propionamid

-
-
100-52-7
benzaldehyde

-
-
872690-15-8
N-(benzotriazol-1-yl-phenyl-methyl)-2-(1,3-dioxo-1,3-dihydro-isoindol-2-yl)-3-(3-methoxy-phenyl)-propionamide

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 100% |

-
-
95-14-7
1,2,3-Benzotriazole

-
-
104-88-1
4-chlorobenzaldehyde

-
-
21946-94-1
(S)-2-(1,3-dioxoisoindolin-2-yl)-3-phenylpropanamide

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 100% |

-
-
95-14-7
1,2,3-Benzotriazole

-
-
100-52-7
benzaldehyde

-
-
21946-94-1
(S)-2-(1,3-dioxoisoindolin-2-yl)-3-phenylpropanamide

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 100% |

-
-
95-14-7
1,2,3-Benzotriazole

-
-
1122-91-4
4-bromo-benzaldehyde

-
-
21946-94-1
(S)-2-(1,3-dioxoisoindolin-2-yl)-3-phenylpropanamide

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In benzene Heating; | 100% |

-
-
95-14-7
1,2,3-Benzotriazole

-
-
50-00-0
formaldehyd

-
-
103-67-3
benzyl-methyl-amine

-
-
57684-27-2
N-((1H-benzo[d][1,2,3]triazol-1-yl)methyl)-N-methyl-1-phenylmethanamine

| Conditions | Yield |
|---|---|
| In methanol; diethyl ether; water Reflux; | 100% |
| In methanol; water at 50℃; for 15h; | 85% |
| In water |

-
-
30433-91-1
[2-(2-thyenyl)ethyl]amine

-
-
95-14-7
1,2,3-Benzotriazole

-
-
50-00-0
formaldehyd

-
-
1201693-98-2
benzotriazol-1-ylmethyl-(2-thiophen-2-yl-ethyl)-amine

| Conditions | Yield |
|---|---|
| In diethyl ether; water at 20℃; | 100% |


| Conditions | Yield |
|---|---|
| With thionyl chloride In dichloromethane at 20℃; for 8.5h; | 100% |
| Stage #1: 1,2,3-Benzotriazole With thionyl chloride In dichloromethane at 25℃; for 0.5h; Stage #2: 3-nitrobenzoic acid In dichloromethane at 25℃; for 3h; | 89% |
| Stage #1: 3-nitrobenzoic acid With iodine; triphenylphosphine In dichloromethane at 0℃; for 0.5h; Stage #2: 1,2,3-Benzotriazole In dichloromethane at 0℃; for 0.166667h; Stage #3: With triethylamine In dichloromethane at 20℃; for 1h; | 70% |
| Stage #1: 3-nitrobenzoic acid With 1,3,5-trichloro-2,4,6-triazine; triethylamine In dichloromethane at 0℃; for 0.166667h; Stage #2: 1,2,3-Benzotriazole In dichloromethane at 0 - 25℃; for 1h; | 70% |
| With thionyl chloride In dichloromethane at 20℃; |

| Conditions | Yield |
|---|---|
| With thionyl chloride In dichloromethane at 20℃; for 8.5h; | 100% |
| Stage #1: ortho-nitrobenzoic acid With trichloroisocyanuric acid; triphenylphosphine In dichloromethane at 20℃; for 0.5h; Stage #2: 1,2,3-Benzotriazole In dichloromethane at 20℃; | 64% |
| With thionyl chloride In dichloromethane at 20℃; |
1H-Benzotriazole Chemical Properties
Empirical Formula: C6H5N3
Molecular Weight: 119.124
Nominal Mass: 119 Da
Average Mass: 119.124 Da
Monoisotopic Mass: 119.048347 Da
EINECS: 202-394-1
Index of Refraction: 1.715
Molar Refractivity: 34.71 cm3
Molar Volume: 88.3 cm3
Surface Tension: 73.9 dyne/cm
Density: 1.348 g/cm3
Flash Point: 133.9 °C
Enthalpy of Vaporization: 51.46 kJ/mol
Boiling Point: 276.1 °C at 760 mmHg
Vapour Pressure: 0.00491 mmHg at 25 °C
Melting point: 97-99 °C(lit.)
Water solubility: 025 g/l in water (20 °C)
Appearance: White to light tan crystals or white powder
Structure of 1H-Benzotriazole (CAS NO.95-14-7):
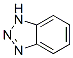
IUPAC Name: 2H-Benzotriazole
Canonical SMILES: C1=CC2=NNN=C2C=C1
InChI: InChI=1S/C6H5N3/c1-2-4-6-5(3-1)7-9-8-6/h1-4H,(H,7,8,9)
InChIKey: QRUDEWIWKLJBPS-UHFFFAOYSA-N
Product Category of 1H-Benzotriazole (CAS NO.620-17-7): Industrial/Fine Chemicals;Heterocyclic Compounds;Imidaxoles;Copper corrosion inhibitor;Organic Chemicals
1H-Benzotriazole History
It is produced by reaction of o-phenylenediamine, sodium nitrite and acetic acid. The conversion proceeds via diazotization of one of the amine groups.
1H-Benzotriazole Uses
1H-Benzotriazole (CAS NO.95-14-7) is used as a corrosion inhibitor, an aircraft deicer and anti-icer fluid as well as for silver protection in dishwashing detergents.
1H-Benzotriazole Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | oral | 500mg/kg (500mg/kg) | AUTONOMIC NERVOUS SYSTEM: OTHER (DIRECT) PARASYMPATHOMIMETIC BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) LUNGS, THORAX, OR RESPIRATION: CYANOSIS | Gigiena i Sanitariya. For English translation, see HYSAAV. Vol. 46(11), Pg. 70, 1981. |
| mouse | LD50 | intraperitoneal | 400mg/kg (400mg/kg) | Farmakologiya i Toksikologiya Vol. 41, Pg. 708, 1978. Link to PubMed | |
| mouse | LD50 | intravenous | 238mg/kg (238mg/kg) | Journal of Pharmacology and Experimental Therapeutics. Vol. 105, Pg. 486, 1952. Link to PubMed | |
| mouse | LD50 | oral | 615mg/kg (615mg/kg) | National Technical Information Service. Vol. AD-A067-313, | |
| rabbit | LDLo | skin | 450mg/kg (450mg/kg) | National Technical Information Service. Vol. OTS0520182, | |
| rat | LC50 | inhalation | 1910mg/m3/3H (1910mg/m3) | LUNGS, THORAX, OR RESPIRATION: STRUCTURAL OR FUNCTIONAL CHANGE IN TRACHEA OR BRONCHI LUNGS, THORAX, OR RESPIRATION: DYSPNEA LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES | National Technical Information Service. Vol. OTS0520637, |
| rat | LD50 | oral | 560mg/kg (560mg/kg) | National Technical Information Service. Vol. OTS0516797, | |
| rat | LD50 | skin | > 1gm/kg (1000mg/kg) | National Technical Information Service. Vol. OTS0516745, |
1H-Benzotriazole (CAS NO.95-14-7) hasn't been listed as a carcinogen by NTP, IARC, ACGIH, or CA Prop 65. And the toxicological properties have not been fully investigated. You can see actual entry in RTECS for complete information.
1H-Benzotriazole Safety Profile
Hazard Codes:  Xn,
Xn, Xi,
Xi, F
F
Risk Statements: 20/22-36-52/53-5-36/37/38-20/21/22-11
R11:Highly flammable.
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed.
R20/22:Harmful by inhalation and if swallowed.
R36/37/38:Irritating to eyes, respiratory system and skin.
R36:Irritating to eyes.
R52/53:Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment.
Safety Statements: 26-36/37-61-45-36/37/39-28A
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S28:After contact with skin, wash immediately with plenty of soap-suds.
S36/37:Wear suitable protective clothing and gloves.
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S61:Avoid release to the environment. Refer to special instructions / safety data sheets.
1H-Benzotriazole Specification
1H-Benzotriazole , its cas register number is 95-14-7. It also can be called 1,2,3-Benzotriazole ; Benzotriazole ; 1,2,-Aminozophenylene ; and Azimidobenzene . It is hazardous, so the first aid measures and others should be known. Such as: When on the skin: first, should flush skin with plenty of water immediately for at least 15 minutes while removing contaminated clothing. Secondly, get medical aid. Or in the eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Then get medical aid soon. While, it's inhaled: Remove from exposure and move to fresh air immediately. Give artificial respiration while not breathing. When breathing is difficult, give oxygen. And as soon as to get medical aid. Then you have the ingesting of the product: Wash mouth out with water, and get medical aid immediately. Notes to physician: Treat supportively and symptomatically.
In addition, 1H-Benzotriazole (CAS NO.95-14-7) is light sensitive. Dusts may form an explosion hazard. It is not compatible with strong oxidizing agents, heavy metals, and you must not take it with incompatible materials, light, ignition sources, heat. And also prevent it to broken down into hazardous decomposition products: Nitrogen oxides, carbon monoxide, carbon dioxide.
Related Products
- 1H-Benzotriazole
- 1H-Benzotriazole, 1-methyl
- 1H-Benzotriazole,1-(1-ethoxy-2-propen-1-yl)-
- 1H-Benzotriazole,1-(trimethylsilyl)-
- 1H-Benzotriazole,1-chloro-
- 1H-Benzotriazole,5,6-dimethyl-
- 1H-Benzotriazole,6-bromo-
- 1H-Benzotriazole,6-fluoro-
- 1H-Benzotriazole,6-methoxy-
- 1H-Benzotriazole,7-methyl-
- 95153-31-4
- 95154-01-1
- 951-55-3
- 95-15-8
- 951624-83-2
- 951625-97-1
- 951626-15-6
- 951650-22-9
- 951677-39-7
- 951677-47-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View