-
Name
2-Acetyl-4-methylpyridine
- EINECS
- CAS No. 59576-26-0
- Article Data12
- CAS DataBase
- Density 1.036 g/cm3
- Solubility
- Melting Point 30-34 °C(lit.)
- Formula C8H9NO
- Boiling Point 227.1 °C at 760 mmHg
- Molecular Weight 135.166
- Flash Point 96.5 °C
- Transport Information
- Appearance
- Safety 26-36
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Ketone,methyl 4-methyl-2-pyridyl (6CI);1-(4-Methyl-2-pyridyl)-1-ethanone;Ethanone,1-(4-methyl-2-pyridinyl)-;4-Methyl-2-acetylpyridine;
- PSA 29.96000
- LogP 1.59260
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: 2-Bromo-4-picoline With n-butyllithium In diethyl ether; hexane at -78℃; for 0.166667 - 0.5h; Stage #2: N,N-dimethyl acetamide In diethyl ether; hexane at 20℃; for 2 - 14.5h; Product distribution / selectivity; | 59% |
| Stage #1: 2-Bromo-4-picoline With n-butyllithium In diethyl ether; hexane at -78℃; for 0.166667h; Stage #2: N,N-dimethyl acetamide In diethyl ether; hexane at 20℃; for 14.5h; | 59% |
| With n-butyllithium In diethyl ether; hexane | 37% |
-
-
1620-76-4
2-Cyano-4-methylpyridin

-
-
917-64-6
methyl magnesium iodide

-
-
59576-26-0
2-acetyl-4-methylpyridine

| Conditions | Yield |
|---|---|
| Stage #1: 2-Cyano-4-methylpyridin; methyl magnesium iodide In diethyl ether; benzene at 0 - 20℃; for 2h; Inert atmosphere; Stage #2: With water; ammonium chloride In diethyl ether; benzene Inert atmosphere; | 52% |
| With diethyl ether; benzene Behandeln des Reaktionsgemisches mit wss.Ammoniumchlorid-Loesung; |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide; iron(II) sulfate; trifluoroacetic acid In acetonitrile for 4h; Heating; | 20% |
-
-
4926-28-7
2-Bromo-4-picoline

-
-
59576-26-0
2-acetyl-4-methylpyridine

-
A
-
59576-26-0
2-acetyl-4-methylpyridine

| Conditions | Yield |
|---|---|
| In tert-butyl alcohol; benzene for 5h; Irradiation; | |
| In tert-butyl alcohol; benzene for 5h; Quantum yield; Irradiation; |

-
-
108-89-4
picoline

-
-
59576-26-0
2-acetyl-4-methylpyridine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 30 percent / silver nitrate, ammonium persulfate, conc. H2SO4 / H2O; CH2Cl2 / 3 h / 40 °C 2: benzene; 2-methyl-propan-2-ol / 5 h / Irradiation View Scheme |
-
-
695-34-1
2-Amino-4-methylpyridine

-
-
59576-26-0
2-acetyl-4-methylpyridine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: diazonium reaction View Scheme |
-
-
1620-76-4
2-Cyano-4-methylpyridin

-
-
75-16-1
methylmagnesium bromide

-
-
59576-26-0
2-acetyl-4-methylpyridine

| Conditions | Yield |
|---|---|
| Stage #1: 2-Cyano-4-methylpyridin; methylmagnesium bromide In tetrahydrofuran; diethyl ether at -20 - -10℃; Stage #2: With hydrogenchloride; water In tetrahydrofuran; diethyl ether at -40℃; for 0.0833333h; |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
13846-13-4
2-aminobenzenesulfonic acid sodium salt

| Conditions | Yield |
|---|---|
| In 5,5-dimethyl-1,3-cyclohexadiene; ethanol Reflux; | 97% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 20℃; for 2h; | 97% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

| Conditions | Yield |
|---|---|
| Stage #1: 2-acetyl-4-methylpyridine With trimethylaluminum; 4,4,5,5-tetramethyl-[1,3,2]-dioxaboralane; 3,3'-bis-(3,5-bis-trifluoromethyl-phenyl)-[1,1']binaphthalenyl-2,2'-diol In toluene at 20℃; for 2h; Glovebox; Inert atmosphere; Stage #2: With methanol for 0.5h; enantioselective reaction; | 97% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
5392-12-1
2-methoxy-1-naphthaldehyde

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol at 20℃; for 1h; Inert atmosphere; | 96% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
95-92-1
oxalic acid diethyl ester

-
-
741288-24-4
ethyl 4-(4-methylpyridin-2-yl)-2,4-dioxobutanoate

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol at 20℃; | 95% |
| Stage #1: oxalic acid diethyl ester With sodium ethanolate In ethanol at 20℃; for 0.166667h; Stage #2: 2-acetyl-4-methylpyridine In ethanol for 0.5h; Stage #3: With hydrogenchloride; water Product distribution / selectivity; more than 3 stages; | 82% |
| Stage #1: 2-acetyl-4-methylpyridine; oxalic acid diethyl ester With sodium ethanolate In ethanol at 20 - 60℃; for 9h; Stage #2: With water In diethyl ether; ethanol Product distribution / selectivity; |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
1563017-51-5
1-(4-methyl-1-oxido-2-pyridinyl)ethanone

| Conditions | Yield |
|---|---|
| With 3-chloro-benzenecarboperoxoic acid In dichloromethane at 0℃; | 88% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
247174-18-1
6-bromo-2-methoxynaphthalene-1-carbaldehyde

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol at 5 - 10℃; Inert atmosphere; | 83% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
405-18-5
trans-2-phenylethene-1-sulfonyl fluoride

| Conditions | Yield |
|---|---|
| With nickel(II) nitrate hexahydrate; potassium carbonate In acetonitrile at 20℃; for 24h; | 82% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
75-15-0
carbon disulfide

-
-
75-03-6
ethyl iodide

-
-
170653-78-8
2-(3',3'-bis(ethylthio)-1'-oxoprop-2'-en-1'-yl)-4-methylpyridine

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran Ambient temperature; | 81% |

-
-
59576-26-0
2-acetyl-4-methylpyridine

| Conditions | Yield |
|---|---|
| With hydrogen bromide; bromine; acetic acid at 20℃; for 2h; Inert atmosphere; | 81% |
| With bromine In HBr-AcOH; diethyl ether; acetic acid | 10.8 g (63 %) |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
5973-71-7
3,4-dimethylbenzaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: 3,4-dimethylbenzaldehyde With sodium hydroxide In ethanol; water at 20℃; Stage #2: 2-acetyl-4-methylpyridine In ethanol; water at 20℃; for 0.166667h; | 81% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
78570-34-0
3,3-bis(methylsulfanyl)-1-pyridin-2-ylprop-2-en-1-one

-
-
108295-36-9
4-methyl-4'-(methylthio)-2,2':6',2"-terpyridine

| Conditions | Yield |
|---|---|
| Stage #1: 2-acetyl-4-methylpyridine With potassium tert-butylate In tetrahydrofuran at 20℃; for 0.166667h; Stage #2: 3,3-bis(methylsulfanyl)-1-pyridin-2-ylprop-2-en-1-one In tetrahydrofuran for 16h; Stage #3: With ammonium acetate; acetic acid In tetrahydrofuran for 4h; Heating; | 77% |
| With potassium tert-butylate; ammonium acetate 2) AcOH, heating; Multistep reaction; |

| Conditions | Yield |
|---|---|
| In ethanol | 74% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol Claisen-Schmidt Condensation; Reflux; | 72% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
1384956-00-6
3,5-dimethyl-[1,2,3]triazolo[1,5-a]pyridine

| Conditions | Yield |
|---|---|
| Stage #1: 2-acetyl-4-methylpyridine With hydrazine hydrate In ethanol for 1h; Reflux; Stage #2: With [bis(acetoxy)iodo]benzene In dichloromethane at 20℃; for 0.5h; | 70% |
| Multi-step reaction with 2 steps 1: methanol / Cooling with ice 2: morpholine / 90 - 100 °C View Scheme | |
| Multi-step reaction with 2 steps 1: hydrazine hydrate / ethanol / 1 h / Reflux 2: [bis(acetoxy)iodo]benzene / dichloromethane / 0.5 h / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| In ethanol; acetonitrile Heating; | 70% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
25688-12-4
N-(2-bromophenyl)hydrazinecarbothioamide

| Conditions | Yield |
|---|---|
| With acetic acid In methanol for 2h; Reflux; | 69% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
56252-09-6
2,3-Dimethoxy-naphthalene-1-carbaldehyde

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol at 5 - 10℃; Inert atmosphere; | 67% |
-
-
110-86-1
pyridine

-
-
59576-26-0
2-acetyl-4-methylpyridine

| Conditions | Yield |
|---|---|
| With iodine at 20℃; | 65% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water at 20℃; for 3h; | 62% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
4637-24-5
N,N-dimethyl-formamide dimethyl acetal

-
-
166196-85-6
3-(dimethylamino)-1-(4-methylpyridin-2-yl)prop-2-en-1-one

| Conditions | Yield |
|---|---|
| for 20h; Heating; | 61% |
| for 3h; Reflux; Inert atmosphere; Schlenk technique; | 0.6 g |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
1122-91-4
4-bromo-benzaldehyde

-
-
878007-29-5
1-(3-oxo-3-[2-(4-methylpyridyl)]propen-1-yl)-4-bromobenzene

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol; water at 25℃; for 1h; Claisen-Schmidt condensation; | 60% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
920518-65-6
3-dimethyl-amino-2-methylpropenal

-
-
1134-35-6
4,4'-dimethyl-2,2'-bipyridines

| Conditions | Yield |
|---|---|
| With ammonium acetate; sodium hydroxide; acetic acid In tetrahydrofuran; toluene | 59.7% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

| Conditions | Yield |
|---|---|
| In 5,5-dimethyl-1,3-cyclohexadiene; ethanol Reflux; | 59% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
78-94-4
methyl vinyl ketone

-
A
-
1567389-02-9
8-acetyl-1-aza-7-ethylbicyclo[4.3.0]nona-2,4,6,8-tetraene

-
B
-
1567389-18-7
8-acetyl-1-aza-7-ethyl-9-(3-oxobutyl)bicyclo[4.3.0]nona-2,4,6,8-tetraene

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate In water; acetonitrile at 0℃; for 12h; Baylis-Hillman Reaction; Reflux; Overall yield = 80 %; | A 57% B 23% |

-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
620-23-5
m-tolyl aldehyde

| Conditions | Yield |
|---|---|
| With ammonia; potassium hydroxide In ethanol; water at 60℃; for 24h; Kroehnke Pyridine Synthesis; | 51% |
-
-
59576-26-0
2-acetyl-4-methylpyridine

-
-
54884-55-8
2-methoxy-6-methyl-benzaldehyde

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol at 20℃; for 1h; Inert atmosphere; | 48% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-acetyl-4-methylpyridine; 4-formylphenylboronic acid, With sodium hydroxide In ethanol at 25℃; for 10h; Stage #2: With ammonium hydroxide In ethanol; water for 20h; Reflux; | 47% |
2-Acetyl-4-methylpyridine Chemical Properties
Empirical Formula: C8H9NO
Molecular Weight: 135.1632
Freely Rotating Bonds: 1
Polar Surface Area: 29.96 Å2
Index of Refraction: 1.512
Molar Refractivity: 39.19 cm3
Molar Volume: 130.4 cm3
Polarizability: 15.53× 10-24 cm3
Surface Tension: 38.1 dyne/cm
Density: 1.036 g/cm3
Flash Point: 96.5 °C
Enthalpy of Vaporization: 46.37 kJ/mol
Melting Point: 30-34 °C(lit.)
Boiling Point: 227.1 °C at 760 mmHg
Vapour Pressure: 0.0788 mmHg at 25°C
The Cas Register Number of 2-Acetyl-4-methylpyridine is 59576-26-0.The chemical synonyms of 2-Acetyl-4-methylpyridine (CAS NO.59576-26-0) are 1-(4-Methyl-2-pyridinyl)ethanone and ethanone, 1-(4-methyl-2-pyridinyl)- .The molecular structure of 2-Acetyl-4-methylpyridine (CAS NO.59576-26-0) is
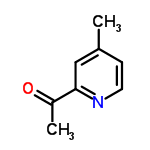 .
.
2-Acetyl-4-methylpyridine Uses
2-Acetyl-4-methylpyridine (CAS NO.59576-26-0) is used as organic intermediate .
2-Acetyl-4-methylpyridine Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38: Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36: Wear suitable protective clothing.
WGK Germany: 3
Related Products
- 2-Acetyl-4-methylpyridine
- 5957-75-5
- 595-77-7
- 5957-80-2
- 59578-63-1
- 5957-96-0
- 5958-24-7
- 595-84-6
- 59-58-5
- 59586-13-9
- 59587-08-5
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View