-
Name
2-AMINO-4,6-DICHLOROPHENOL
- EINECS 208-421-3
- CAS No. 527-62-8
- Article Data23
- CAS DataBase
- Density 1.56 g/cm3
- Solubility
- Melting Point 97 ºC
- Formula C6H5 Cl2 N O
- Boiling Point 286.5 °C at 760 mmHg
- Molecular Weight 178.018
- Flash Point 127.1 °C
- Transport Information
- Appearance light yellow or white solid
- Safety Mutation data reported. When heated to decomposition it emits toxic vapors of NOx and Cl−.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 2,4-Dichloro-6-aminophenol;2-Amino-4,6-dichlorophenol; 4,6-Dichloro-2-aminophenol;4,6-Dichloro-o-aminophenol; 6-Amino-2,4-dichlorophenol; NSC 658464
- PSA 46.25000
- LogP 2.86240
Synthetic route

| Conditions | Yield |
|---|---|
| With hydrogen; platinum(IV) oxide In ethanol at 20℃; under 750.06 Torr; for 96h; | 99% |
| With tin(ll) chloride In ethanol | 97% |
| With sodium dithionite In ethanol; water at 65℃; for 4h; | 60% |

| Conditions | Yield |
|---|---|
| In ethanol | 80% |
| In ethanol | 80% |
| In ethanol | 80% |
-
-
139137-46-5
trans-2,4-dichloro-6-phenylazo-phenol

-
-
527-62-8
2,4-dichloro-6-aminophenol

| Conditions | Yield |
|---|---|
| With sodium dithionite |
-
-
7647-01-0
hydrogenchloride

-
-
858013-68-0
4,6,4',6'-tetrachloro-2,2'-azo-di-phenol

-
-
527-62-8
2,4-dichloro-6-aminophenol


-
-
527-62-8
2,4-dichloro-6-aminophenol

| Conditions | Yield |
|---|---|
| With hydrogenchloride In benzene |

-
-
527-62-8
2,4-dichloro-6-aminophenol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: ZrCl4 / benzene / 0.2 h / reflux 2: 10 percent HCl / benzene View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: fuming nitric acid 2: tin; hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: chlorine 2: fuming nitric acid 3: tin; hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| In water; sodium hydrogencarbonate |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
219963-60-7
4-methoxy-3-(4-(2,2,2-trichloro-acetyl)piperazin-1-yl)benzene-1-sulfonylchloride

-
-
1331830-34-2
N-(3,5-dichloro-2-hydroxyphenyl)-4-methoxy-3-[4-(2,2,2-trichloroethanoyl)-piperazin-1-yl]-benzenesulfonamide

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane at 0 - 20℃; Inert atmosphere; | 96% |
-
-
75-15-0
carbon disulfide

-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
98279-11-9
5,7-dichloro-2-mercaptobenzoxazole

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol at 20℃; for 6h; Reflux; | 95% |
| Stage #1: carbon disulfide; 2,4-dichloro-6-aminophenol With potassium hydroxide In methanol for 8h; Cooling with ice; Reflux; Stage #2: With acetic acid In methanol pH=6; Cooling with ice; | 84.25% |
| With potassium hydroxide In ethanol for 8h; Heating; | |
| With potassium hydroxide In ethanol Heating; |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
108-24-7
acetic anhydride

-
-
55202-45-4
2-acetamido-4,6-dichlorophenol

| Conditions | Yield |
|---|---|
| for 0.25h; Ambient temperature; | 92% |
-
-
117-80-6
2,3-Dichloro-1,4-naphthoquinone

-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
73397-13-4
6,8,10-trichloro-5H-benzophenoxazin-5-one

| Conditions | Yield |
|---|---|
| With potassium acetate In ethanol at 90 - 100℃; for 20h; | 87% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
506-68-3
bromocyane

-
-
98555-67-0
5,7-dichloro-benzoxazol-2-ylamine

| Conditions | Yield |
|---|---|
| In ethanol at 60℃; for 12h; | 86% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
1427468-73-2
N-(tert-butyl)-5,7-dichlorobenzo[d]oxazol-2-amine

| Conditions | Yield |
|---|---|
| With tetrakis(triphenylphosphine) palladium(0) In 1,4-dioxane at 50℃; for 8h; | 85% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
1395499-28-1
2-(2'-anthraquinonylamino-3'-hydroxy)-4-anilino-6-chloro-s-triazine

-
-
1395499-23-6
C29H18Cl2N6O4

| Conditions | Yield |
|---|---|
| In nitrobenzene at 60 - 65℃; for 5h; | 83% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
137076-22-3
tert butyl 4-formylpiperidine-1-carboxylate

| Conditions | Yield |
|---|---|
| With zinc dibromide In toluene Heating; | 83% |

| Conditions | Yield |
|---|---|
| With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane | 82% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
119072-55-8, 7188-38-7
tert-butylisonitrile

-
-
1427468-91-4
N-(tert-butyl)-2-(tert-butylimino)-6,8-dichloro-2H-benzo[b][1,4]oxazin-3-amine

| Conditions | Yield |
|---|---|
| With palladium dichloride In 1,4-dioxane at 50℃; for 4h; | 82% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
63969-35-7
2,4-dichloro-6-diazocyclohexa-2,4-dien-1-one

| Conditions | Yield |
|---|---|
| Stage #1: 2,4-dichloro-6-aminophenol With hydrogenchloride In ethanol; water at 0℃; for 0.166667h; Inert atmosphere; Schlenk technique; Stage #2: With sodium nitrite In ethanol; water at 0℃; for 2.16667h; Inert atmosphere; Schlenk technique; | 81% |
| Stage #1: 2,4-dichloro-6-aminophenol With hydrogenchloride In ethanol Stage #2: With 1-nitro-3-methylbutane In methanol | 80% |
| Stage #1: 2,4-dichloro-6-aminophenol With hydrogenchloride In ethanol; water at 0℃; for 0.166667h; Stage #2: With sodium nitrite In ethanol; water at 0℃; for 2.16667h; Stage #3: With potassium carbonate In dichloromethane; water Cooling with ice; |
-
-
527-62-8
2,4-dichloro-6-aminophenol

| Conditions | Yield |
|---|---|
| With 1,1'-carbonyldiimidazole In tetrahydrofuran at 20℃; for 16h; | 81% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride; trifluoroacetic acid In 1,2-dichloro-ethane for 1h; Reflux; | 80% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
1060757-29-0
5-(4-trifluoromethylphenyl)-6-oxa-4-azaspiro[2.4]hept-4-ene-7-carboxylic acid

| Conditions | Yield |
|---|---|
| Stage #1: 5-(4-trifluoromethylphenyl)-6-oxa-4-azaspiro[2.4]hept-4-ene-7-carboxylic acid With 1-hydroxy-7-aza-benzotriazole; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane for 0.25h; Inert atmosphere; Cooling with ice; Stage #2: 2,4-dichloro-6-aminophenol With 2,4,6-trimethyl-pyridine In dichloromethane at 20℃; for 12h; Inert atmosphere; | 79% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In ethanol; water at 140℃; for 1.5h; Microwave irradiation; Sealed tube; | 71% |

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride; trifluoroacetic acid In 1,2-dichloro-ethane at 50℃; for 8h; | 68% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
50-00-0
formaldehyd

-
-
1022250-47-0
2-dimethylamino-4,6-dichlorophenol

| Conditions | Yield |
|---|---|
| Stage #1: 2,4-dichloro-6-aminophenol; formaldehyd With sodium cyanoborohydride; acetic acid In water; acetonitrile at 0℃; for 2h; Stage #2: With sodium hydrogencarbonate In water; acetonitrile | 64% |
| With sodium cyanoborohydride; acetic acid In water; acetonitrile at 20℃; for 12h; Cooling with ice; | 55.16% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

| Conditions | Yield |
|---|---|
| Stage #1: 2,4-dichloro-6-aminophenol; 4-hydroxyquinazolin-7-benzaldehyde With titanium(IV) isopropylate In 1,2-dichloro-ethane for 5h; Reflux; Stage #2: With sodium cyanoborohydride In 1,2-dichloro-ethane Reflux; | 63% |

| Conditions | Yield |
|---|---|
| With sulfur; 2,2'-azobis-(2,4-dimethylvaleronitrile); sodium hydrogencarbonate; bis(pinacol)diborane In N,N-dimethyl-formamide at 100℃; for 15h; Reagent/catalyst; Solvent; Inert atmosphere; | 62% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
308846-06-2
5-(bromomethyl)pyridine-2-carbonitrile

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; potassium iodide In acetonitrile for 2h; | 60% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
109-97-7
pyrrole

-
-
121789-62-6
2-(3,5-Dichlor-2-hydroxyphenyl)-1H-pyrrol

| Conditions | Yield |
|---|---|
| Stage #1: 2,4-dichloro-6-aminophenol With tert.-butylnitrite In dimethyl sulfoxide at 20℃; for 0.5h; Inert atmosphere; Stage #2: pyrrole With chlorpromazine hydrochloride In dimethyl sulfoxide at 0℃; Inert atmosphere; | 55% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride; N-ethyl-N,N-diisopropylamine In 1,2-dichloro-ethane at 20℃; for 18h; | 55% |
| With sodium tris(acetoxy)borohydride In ethanol; 1,2-dichloro-ethane at 20℃; for 18h; | 38% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
131747-68-7
2-cyanopyridine-5-carboxyaldehyde

| Conditions | Yield |
|---|---|
| With sodium cyanoborohydride; acetic acid In methanol at 60℃; | 53% |

| Conditions | Yield |
|---|---|
| With sulfur; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; sodium t-butanolate In N,N-dimethyl-formamide at 70℃; for 3h; Molecular sieve; Inert atmosphere; | 52% |
| With sulfur; sodium t-butanolate In N,N-dimethyl-formamide at 70℃; for 3h; Molecular sieve; | 52% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

| Conditions | Yield |
|---|---|
| With 2,2'-azobis-(2,4-dimethylvaleronitrile); sodium hydrogencarbonate; sulfur; bis(pinacol)diborane In N,N-dimethyl-formamide at 100℃; for 15h; Inert atmosphere; | 52% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

| Conditions | Yield |
|---|---|
| With sodium tris(acetoxy)borohydride; trifluoroacetic acid In 1,2-dichloro-ethane for 1h; Reflux; | 50% |
-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
50-78-2
aspirin

-
-
81665-46-5
salicylic acid-(3,5-dichloro-2-hydroxy-anilide)

| Conditions | Yield |
|---|---|
| With sodium hydroxide; thionyl chloride In N,N-dimethyl-aniline; acetone | 49% |

-
-
527-62-8
2,4-dichloro-6-aminophenol

-
-
28621-54-7
N,N-diethyl-N'-chlorosulfonyl chloroformamidine

-
-
1155876-12-2
7,9-dichloro-4-(diethylamino)-2,2-dioxo-2λ6-5H-[1,2,3,5]benzoxathiadiazepine

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In various solvent(s) at 20℃; | 48% |
2-Amino-4,6-dichlorophenol Chemical Properties
Empirical Formula: C6H5Cl2NO
Molecular Weight: 178.016
EINECS: 208-421-3
Index of Refraction: 1.661 Molar
Density: 1.56 g/cm3
Flash Point: 127.1 °C
Enthalpy of Vaporization: 54.67 kJ/mol
Boiling Point: 286.5 °C at 760 mmHg
Vapour Pressure: 0.00153 mmHg at 25 °C
Melting point: 97 °C
Appearance: Long needles
Product categories of 2-Amino-4,6-dichlorophenol (CAS.527-62-8): Aromatic Phenols
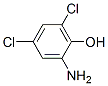
Structure of 2-Amino-4,6-dichlorophenol (CAS.527-62-8):
2-Amino-4,6-dichlorophenol Toxicity Data With Reference
| 1. | mnt-ham-lng 200 mg/L | MUREAV Mutation Research. 368 (1996),149. | ||
| 2. | sce-ham-lng 100 mg/L | MUREAV Mutation Research. 368 (1996),149. |
2-Amino-4,6-dichlorophenol Safety Profile
Mutation data reported. When heated to decomposition it emits toxic vapors of NOx and Cl−.
Hazard Note: Irritant
2-Amino-4,6-dichlorophenol Specification
2-Amino-4,6-dichlorophenol (CAS.527-62-8) also can be called 2,4-Dichloro-6-aminophenol and; Phenol, 2-amino-4,6-dichloro- . It is probably combustible, and may react with strong oxidizers and mineral acids or bases .
Related Products
- 2-AMINO-4-γ-DIETHYLAMINOPROPYLAMINO-5,6-DIMETHYLPYRIMIDINE
- 2-Amino-4-(1'-hydroxyethyl)pyridine
- 2-AMINO-4-(2-(5-NITRO-2-FURYL)VINYL) THIAZOLE
- 2-Amino-4-(2-chloroethoxy)-5-methoxybenzoic acid methyl ester
- 2-Amino-4-(3,4-difluorophenyl)thiazole
- 2-Amino-4-(3-chloropropoxy)-5-methoxybenzonitrile
- 2-Amino-4-(3-pyridyl)thiazole
- 2-Amino-4-(4-bromophenyl)thiazole
- 2-Amino-4-(4-chlorophenyl)thiazole
- 2-Amino-4-(4-methoxyphenyl)pyrimidine
- 52763-21-0
- 52-76-6
- 52766-27-5
- 52767-84-7
- 527-69-5
- 52771-21-8
- 52771-22-9
- 527-72-0
- 52772-11-9
- 527-73-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View