-
Name
2-Amino-5-methylbenzoic acid
- EINECS 220-932-3
- CAS No. 2941-78-8
- Article Data35
- CAS DataBase
- Density 1.254 g/cm3
- Solubility
- Melting Point 175 °C (dec.)(lit.)
- Formula C8H9NO2
- Boiling Point 316.6 °C at 760 mmHg
- Molecular Weight 151.165
- Flash Point 145.3 °C
- Transport Information
- Appearance light-brown solid
- Safety 26-36-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms 2-amino-5-methyl-benzoic acid;m-Toluic acid, 6-amino-;Benzoic acid, 2-amino-5-methyl-;6-Amino-m-toluic acid;5-Methylanthranilic acid;2-amino-5-methylbenzoic acid;5-methylanthranilic acid;;L-Tartaric acid diiopropyl ester;
- PSA 63.32000
- LogP 1.85660
Synthetic route
-
-
6492-69-9
5,5'-dimethyl-1H,1'H-[2,2']biindolylidene-3,3'-dione

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 5,5'-dimethyl-1H,1'H-[2,2']biindolylidene-3,3'-dione With bromamine B; sodium hydroxide; palladium dichloride In water; acetonitrile at 60℃; for 2h; pH=12; Stage #2: In water Acidic conditions; | 97% |

| Conditions | Yield |
|---|---|
| With hydrogen; palladium 10% on activated carbon In ethanol at 20℃; | 96% |
| With hydrogen; palladium 10% on activated carbon In ethanol at 20℃; | 96% |
| With palladium on activated carbon; hydrogen In ethyl acetate at 20℃; for 14h; Saturated gas; | 94% |

| Conditions | Yield |
|---|---|
| Stage #1: 5-methyl-1H-indole With bromamine B; sodium hydroxide; palladium dichloride In water; acetonitrile at 60℃; for 3.33333h; pH=12; Stage #2: In water Acidic conditions; | 96% |
| With ruthenium trichloride; osmium(VIII) oxide; bromamine B; sodium hydroxide In water; acetonitrile at 39.84℃; for 4h; | 96% |

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: resin-bound 5-methyl-2-nitrobenzoic acid With aluminium; nickel dichloride at 20℃; for 60h; Stage #2: With trifluoroacetic acid | 85% |

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate; sodium iodide In acetonitrile at 20℃; for 0.666667h; | 80% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; dihydrogen peroxide in der Siedehitze; | |
| With hydroxide; dihydrogen peroxide | |
| With dihydrogen peroxide; sodium hydroxide In water | |
| With sodium hydroxide; dihydrogen peroxide at 10 - 15℃; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 100℃; |
-
-
33900-97-9
2,4-dioxo-3-(p-tolyl)-6-methyl-1,2,3,4-tetrahydroquinazoline

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide |
-
-
5855-94-7
2-(4,6-dimethyl-benzothiazol-2-yl)-4-methyl-aniline

-
A
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
B
-
56536-88-0
3,5-dimethyl-2-aminobenzenethiol

| Conditions | Yield |
|---|---|
| bei der Kalischmelze; |
-
-
86672-48-2
6-methyl-3-phenyl-2,4-(1H,3H)-quinazolinedione

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride |
-
-
7647-01-0
hydrogenchloride

-
-
5925-93-9
2-amino-5-methylbenzonitrile

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| at 100℃; |
-
-
7647-01-0
hydrogenchloride

-
-
3113-72-2
5-methyl-2-nitrobenzoic acid

-
-
2941-78-8
2-Amino-5-methylbenzoic acid


| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: AlCl3; NaCl / 140 °C 2: aqueous KOH View Scheme |

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: tin; fuming hydrochloric acid 2: fuming hydrochloric acid / 100 °C View Scheme |
-
-
64407-07-4
3-(chloromethyl)benzonitrile

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: concentrated sulfuric acid; potassium nitrate 2: tin; fuming hydrochloric acid 3: fuming hydrochloric acid / 100 °C View Scheme |
-
-
64113-86-6
2-nitro-5-methylbenzonitrile

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: fuming hydrochloric acid / 150 °C / im geschlossenen Rohr 2: tin; hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: durch Nitrieren 2: fuming hydrochloric acid / 150 °C / im geschlossenen Rohr 3: tin; hydrochloric acid View Scheme | |
| Multi-step reaction with 3 steps 1: concentrated hydrochloric acid / im Druckrohr 2: fuming nitric acid 3: tin; hydrochloric acid View Scheme | |
| Multi-step reaction with 3 steps 1: concentrated hydrochloric acid / im Druckrohr 2: fuming nitric acid 3: tin; hydrochloric acid View Scheme | |
| Multi-step reaction with 3 steps 1: concentrated hydrochloric acid / im Druckrohr 2: fuming nitric acid 3: tin; hydrochloric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: fuming nitric acid 2: tin; hydrochloric acid View Scheme | |
| Multi-step reaction with 2 steps 1: fuming nitric acid 2: tin; hydrochloric acid View Scheme | |
| Multi-step reaction with 2 steps 1: fuming nitric acid 2: tin; hydrochloric acid View Scheme | |
| Multi-step reaction with 2 steps 1: (nitration) 2: H2 / Pd-C View Scheme |
-
-
18595-16-9
2-amino-5-methylbenzoic acid methyl ester

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In tetrahydrofuran; ethanol; water at 50℃; for 3h; Inert atmosphere; |
-
-
67-56-1
methanol

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
18595-16-9
2-amino-5-methylbenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| With sulfuric acid for 72h; Reflux; | 100% |
| With sulfuric acid for 72h; Reflux; | 92% |
| With thionyl chloride at 0℃; for 24h; | 82% |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
34161-81-4
5-methyl-2-((4-methylphenyl)sulfonamido)benzoic acid

| Conditions | Yield |
|---|---|
| With sodium carbonate In water at 70℃; for 0.333333h; | 100% |
| Stage #1: 2-Amino-5-methylbenzoic acid; p-toluenesulfonyl chloride With sodium carbonate In water at 60 - 85℃; Stage #2: With hydrogenchloride In water | |
| With triethylamine In tetrahydrofuran; water at 0 - 20℃; for 3h; |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
34897-84-2
2-amino-5-methylbenzyl alcohol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; Inert atmosphere; | 100% |
| Stage #1: 2-Amino-5-methylbenzoic acid With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; for 2h; Stage #2: With water; sodium hydroxide In tetrahydrofuran | 100% |
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; for 2h; | 100% |
-
-
186581-53-3, 908094-01-9
diazomethane

-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
18595-16-9
2-amino-5-methylbenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| In diethyl ether for 1h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| In pyridine; toluene at 80℃; for 1h; Cyclization; | 99% |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
80-11-5
N-methyl-N-nitrosotoluene-p-sulfonamide

-
-
18595-16-9
2-amino-5-methylbenzoic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: N-methyl-N-nitrosotoluene-p-sulfonamide With potassium hydroxide In diethyl ether; water Stage #2: 2-Amino-5-methylbenzoic acid In diethyl ether for 1h; | 99% |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
4692-99-3
6-methyl-1H-benzo[d][1,3]oxazine-2,4-dione

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; for 5h; Inert atmosphere; Large scale; | 99% |
| With pyridine In acetonitrile at 55℃; for 2h; Cooling with ice; | 94% |
| With pyridine In acetonitrile at 55℃; for 2h; Cooling with ice; | 93.6% |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
917-54-4
methyllithium

-
-
25428-06-2
2-amino-5-methyl-acetophenone

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0℃; for 6h; | 99% |
| In N,N-dimethyl acetamide at 0℃; for 2h; |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
98-09-9
benzenesulfonyl chloride

-
-
138964-56-4
5-methyl-2-[(phenylsulfonyl)amino]benzoic acid

| Conditions | Yield |
|---|---|
| With sodium carbonate In water at 60 - 80℃; for 6.03333h; Heating / reflux; | 98.7% |
| With sodium carbonate In water at 80℃; for 6h; | 0.22 g |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
162046-61-9
2-trifluoromethoxy-benzoyl chloride

| Conditions | Yield |
|---|---|
| With triethylamine In toluene at 20℃; for 48h; Cyclization; | 98% |
| With triethylamine In n-heptane; toluene |

| Conditions | Yield |
|---|---|
| Stage #1: terephthalic acid With thionyl chloride In toluene at 0 - 75℃; for 7h; Stage #2: 2-Amino-5-methylbenzoic acid In N,N-dimethyl acetamide; toluene at 0 - 5℃; for 2h; Cooling with ice-methanol; Stage #3: With acetic anhydride In N,N-dimethyl acetamide; toluene for 1.5h; Product distribution / selectivity; Heating / reflux; | 98% |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Amino-5-methylbenzoic acid; terephthaloyl chloride In N,N-dimethyl acetamide at -3 - 4℃; for 2h; Cooling with ice-methanol; Stage #2: With acetic anhydride In N,N-dimethyl acetamide; toluene for 1.5h; Product distribution / selectivity; Heating / reflux; | 98% |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
13091-43-5
2-amino-3-bromo-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| With bromine In acetic acid at 20℃; for 3h; Bromination; | 97% |
| With bromine In acetic acid | |
| With N-Bromosuccinimide In N,N-dimethyl-formamide at 20℃; for 14h; |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
79791-29-0
2-(3-(2,5-dimethylphenoxy)propyl)-2-methylpropionyl chloride

| Conditions | Yield |
|---|---|
| With sodium hydroxide In 1,4-dioxane; water at 0℃; for 10.5h; | 96.7% |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
320740-16-7
2-amino-3-iodo-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| With hydrogenchloride; Iodine monochloride at 50℃; for 0.5h; | 96% |
| With iodine In acetic acid for 72h; Iodination; Heating; | 16% |

| Conditions | Yield |
|---|---|
| With triethylamine In ethanol for 3h; Reflux; | 96% |
| With triethylamine In ethanol |

| Conditions | Yield |
|---|---|
| With oxygen; N,N,N',N'-tetramethylguanidine In acetonitrile at 30℃; under 760.051 Torr; for 120h; Irradiation; Green chemistry; | 96% |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
76-83-5
trityl chloride

-
-
85167-27-7
5-methyl-2-<(triphenylmethyl)amino>benzoic acid

| Conditions | Yield |
|---|---|
| With pyridine 1.) reflux, 5 min, 2.) room temperature; | 95% |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
18063-02-0
2,6-difluorobenzoylchloride

| Conditions | Yield |
|---|---|
| With triethylamine In toluene at 20℃; for 48h; Cyclization; | 95% |
| With triethylamine In n-heptane; toluene |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
6089-09-4
4-pentynoic acid

-
-
1227624-58-9
3a,7-dimethyl-3,3a-dihydro-1H-benzo[d]pyrrolo[2,1-b][1,3]-oxazine-1,5(2H)-dione

| Conditions | Yield |
|---|---|
| With Echavarren's catalyst In 1,1-dichloroethane at 120℃; for 12h; Sealed vial; | 95% |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
622-78-6
Benzyl isothiocyanate

-
-
852239-39-5
2-mercapto-3-benzyl-4-oxo-6-methyl-3H-quinazoline

| Conditions | Yield |
|---|---|
| With triethylamine In ethanol for 2h; Reflux; | 95% |
| With triethylamine In ethanol Reflux; | |
| With triethylamine In ethanol | |
| With triethylamine In ethanol |

| Conditions | Yield |
|---|---|
| Stage #1: 2-Amino-5-methylbenzoic acid With thionyl chloride In toluene for 2h; Reflux; Stage #2: benzyl alcohol In toluene at 20℃; | 95% |

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 120℃; | 95% |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
79-30-1
isobutyryl chloride

-
-
890982-57-7
2-(isobutyramido)-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran Reflux; | 94.6% |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 110℃; for 72h; | 94% |
| With hydrogenchloride for 2h; Heating; | 72% |
| With thionyl chloride for 18h; Reflux; Inert atmosphere; Cooling with ice; | |
| With sulfuric acid at 80℃; |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

-
-
503-38-8
trichloromethyl chloroformate

-
-
4692-99-3
6-methyl-1H-benzo[d][1,3]oxazine-2,4-dione

| Conditions | Yield |
|---|---|
| In 1,4-dioxane for 4h; Heating / reflux; | 94% |
| In 1,4-dioxane at 20℃; for 4h; Product distribution / selectivity; Heating / reflux; | 94% |
| In 1,4-dioxane at 20 - 110℃; for 2h; | 93% |
-
-
2941-78-8
2-Amino-5-methylbenzoic acid

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane at 25℃; for 16h; | 94% |
2-Amino-5-methylbenzoic acid Specification
The IUPAC name of 2-Amino-5-methylbenzoic acid is 2-Amino-5-methylbenzoic acid. With the CAS registry number 2941-78-8, it is also named as 6-Amino-m-toluic acid. The product's categories are Carboxylicacid; Fine Chemical & Intermediates; Amino Acids and Derivatives; Aromatic Carboxylic Acids, Amides, Anilides, Anhydrides & Salts; Benzoic acid; Organic acids; Aromatics Compounds; Aromatics. Besides, it is light-brown solid, which should be stored in closed, cool and dry place. In addition, its molecular formula is C8H9NO2 and molecular weight is 151.16.
The other characteristics of this product can be summarized as: (1)EINECS: 220-932-3; (2)ACD/LogP: 1.67; (3)# of Rule of 5 Violations: 0; (4)ACD/LogD (pH 5.5): 0.73; (5)ACD/LogD (pH 7.4): -0.95; (6)ACD/BCF (pH 5.5): 1.25; (7)ACD/BCF (pH 7.4): 1; (8)ACD/KOC (pH 5.5): 21.98; (9)ACD/KOC (pH 7.4): 1; (10)H bond acceptors: 3; (11)H bond donors: 3; (12)Freely Rotating Bonds: 2; (13)Index of Refraction: 1.618; (14)Molar Refractivity: 42.24 cm3; (15)Molar Volume: 120.5 cm3; (16)Surface Tension: 58.2 dyne/cm; (17)Density: 1.254 g/cm3; (18)Flash Point: 145.3 °C; (19)Melting point: 174-177 °C; (20)Enthalpy of Vaporization: 58.9 kJ/mol; (21)Boiling Point: 316.6 °C at 760 mmHg; (22)Vapour Pressure: 0.000171 mmHg at 25 °C.
Preparation of 2-Amino-5-methylbenzoic acid: this chemical can be prepared by 5-Methyl-2-nitro-benzoic acid.
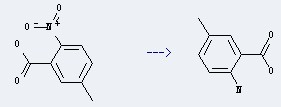
This reaction needs N2H4*H2O, Ethanol and Raney-Ni at temperature of 40 °C.The yield is 65 %.
Uses of 2-Amino-5-methylbenzoic acid: it can react with Acetic acid anhydride to get 2-Acetylamino-5-methyl-benzoic acid.
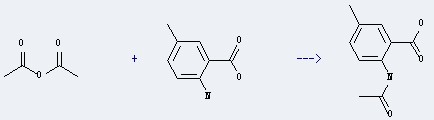
This reaction needs Acetylation by heating for 1 hour. The yield is 93 %.
When you are using this chemical, please be cautious about it as the following: 2-Amino-5-methylbenzoic acid is irritating to eyes, respiratory system and skin. You should wear suitable protective clothing, gloves and eye/face protection when use it. Moreover, in case of contact with eyes, please rinse immediately with plenty of water and seek medical advice.
People can use the following data to convert to the molecule structure.
(1)SMILES:O=C(O)c1cc(ccc1N)C
(2)InChI:InChI=1/C8H9NO2/c1-5-2-3-7(9)6(4-5)8(10)11/h2-4H,9H2,1H3,(H,10,11)
(3)InChIKey:NBUUUJWWOARGNW-UHFFFAOYAI
(4)Std. InChI:InChI=1S/C8H9NO2/c1-5-2-3-7(9)6(4-5)8(10)11/h2-4H,9H2,1H3,(H,10,11)
(5)Std. InChIKey:NBUUUJWWOARGNW-UHFFFAOYSA-N
Related Products
- 2-Amino-5-methylbenzoic acid
- 294181-95-6
- 29418-67-5
- 294187-78-3
- 29419-14-5
- 29419-15-6
- 29420-49-3
- 2942-06-5
- 2942-07-6
- 2942-10-1
- 294210-79-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View