-
Name
2-Ethyl-1,3-cyclopentanedione
- EINECS 212-512-3
- CAS No. 823-36-9
- Article Data39
- CAS DataBase
- Density 1.057 g/cm3
- Solubility
- Melting Point 172-175 °C
- Formula C7H10O2
- Boiling Point 232.9 °C at 760 mmHg
- Molecular Weight 126.155
- Flash Point 84.8 °C
- Transport Information
- Appearance off-white to yellow-beige crystalline powder
- Safety 24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms 2-Ethylcyclopentane-1,3-dione;UNII-91A82TE3OS;1,3-Cyclopentanedione, 2-ethyl-;
- PSA 34.14000
- LogP 0.94460
Synthetic route

| Conditions | Yield |
|---|---|
| With triethylsilane; boron trifluoride diethyl etherate In trifluoroacetic acid Ambient temperature; | 96% |
| With hydrogenchloride; sodium cyanoborohydride In tetrahydrofuran | 75% |
| With hydrogenchloride; sodium cyanoborohydride In tetrahydrofuran at 20℃; | 75% |
-
-
3859-41-4
1,3-cyclopentadione

-
-
75-07-0
acetaldehyde

-
A
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
B
-
1093646-55-9
C13H16O4

| Conditions | Yield |
|---|---|
| With diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; L-proline In dichloromethane at 25℃; for 0.25h; | A 90% B 10% |


| Conditions | Yield |
|---|---|
| With diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; L-proline In dichloromethane at 20℃; for 1h; Inert atmosphere; | 75% |
| With diethyl 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate; L-proline In dichloromethane at 20℃; for 48h; |

| Conditions | Yield |
|---|---|
| (i) semicarbazide*HCl, NaOAc, aq. EtOH, (ii) NaOH, HOCH2CH2OH; Multistep reaction; |
-
-
125113-33-9
2-(1-Ethoxy-propyl)-2-trimethylsilanyloxy-cyclobutanone

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

| Conditions | Yield |
|---|---|
| With Nafion-H; trifluoroacetic acid at 85℃; for 10h; Yield given; |

| Conditions | Yield |
|---|---|
| With 9,10-dihydroanthracene In 1,3,5-trimethyl-benzene at 210℃; Kinetics; Rate constant; different temperatures; ΔG(excit.)300, ΔH(excit.), ΔS(excit.); | A n/a B 88 % Chromat. |
-
-
17082-61-0
1,2-bis(trimethylsiloxy)cyclobutene

-
-
4744-10-9
dimethoxypropane

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

| Conditions | Yield |
|---|---|
| Stage #1: 1,2-bis(trimethylsiloxy)cyclobutene; dimethoxypropane With boron trifluoride diethyl etherate In dichloromethane at -78 - 20℃; for 4h; Stage #2: With trifluoroacetic acid for 24h; Heating; Further stages.; |
-
-
4933-67-9
(3-ethyl-2,4,5-trioxo-cyclopentyl)-glyoxylic acid ethyl ester

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aq. HCl / Heating 2: (i) semicarbazide*HCl, NaOAc, aq. EtOH, (ii) NaOH, HOCH2CH2OH View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Na / ethanol 2: aq. HCl / Heating 3: (i) semicarbazide*HCl, NaOAc, aq. EtOH, (ii) NaOH, HOCH2CH2OH View Scheme |
-
-
14369-94-9
ethyl 4-oxoheptanoate

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol; 5,5-dimethyl-1,3-cyclohexadiene; water; dimethyl sulfoxide |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
57-14-7
1,1-dimethylhydrazine

-
-
124948-39-6
2-ethyl-1,3-cyclopentanedione-dimethylhydrazone

| Conditions | Yield |
|---|---|
| for 4h; Heating; | 99% |
| With toluene-4-sulfonic acid In benzene Heating; | 98% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
932735-55-2
2-ethyl-3-iodocyclopent-2-en-1-one

| Conditions | Yield |
|---|---|
| With iodine; triethylamine; triphenylphosphine In acetonitrile for 8h; Reflux; | 93% |
| With iodine; triethylamine; triphenylphosphine In acetonitrile Heating; | 77% |

| Conditions | Yield |
|---|---|
| [Cp*RuCl(ν2-SMe)]2 In 1,2-dichloro-ethane at 20℃; for 0.5h; | 93% |
-
-
123-75-1
pyrrolidine

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
134622-73-4
2-Ethyl-3-pyrrolidin-1-yl-cyclopent-2-enone

| Conditions | Yield |
|---|---|
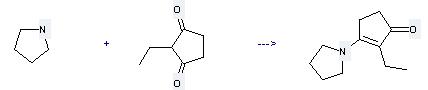
| With acetic acid; propionic acid In toluene for 6h; Heating; | 92% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
107-18-6
allyl alcohol

-
-
59949-74-5
2-ethyl-2-(prop-2-enyl)cyclopentane-1,3-dione

| Conditions | Yield |
|---|---|
| With Ru(Cp*)(η3-C3H5)(p-CH3C6H5SO3)2 In dichloromethane; acetonitrile at 50℃; for 3h; regioselective reaction; | 92% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
78-81-9
isobutylamine

-
-
134622-66-5
2-Ethyl-3-isobutylamino-cyclopent-2-enone

| Conditions | Yield |
|---|---|
| With acetic acid; propionic acid In toluene for 6h; Heating; | 90% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
1422736-27-3
3-(difluoromethoxy)-2-ethylcyclopent-2-en-1-one

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane at 0℃; for 0.333333h; | 89% |
-
-
2016-57-1
1-aminodecane

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
134622-69-8
3-Decylamino-2-ethyl-cyclopent-2-enone

| Conditions | Yield |
|---|---|
| With acetic acid; propionic acid In toluene for 6h; Heating; | 88% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
107-02-8
acrolein

-
-
1416969-76-0
3-(1-ethyl-2,5-dioxocyclopentyl)propanal

| Conditions | Yield |
|---|---|
| In water at 20℃; for 18h; Inert atmosphere; | 87% |
| In water Inert atmosphere; |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
145472-96-4
ethyl 7-(3-methoxyphenyl)-4-oxo(E)-hept-2-enoate

-
-
145472-97-5
ethyl 2-(1-ethyl-2,5-dioxocyclopentyl)-7-(3-methoxyphenyl)-3-oxoheptanoate

| Conditions | Yield |
|---|---|
| With triethylamine In ethyl acetate for 24h; Heating; | 85% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

| Conditions | Yield |
|---|---|
| In ethanol; water for 168h; | 85% |
-
-
110-89-4
piperidine

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
134622-74-5
2-Ethyl-3-piperidin-1-yl-cyclopent-2-enone

| Conditions | Yield |
|---|---|
| With acetic acid; propionic acid In toluene for 6h; Heating; | 84% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
1422736-27-3
3-(difluoromethoxy)-2-ethylcyclopent-2-en-1-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In water at 20℃; for 2h; | 84% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
101387-21-7
ethyl,3-oxo-5-tetrahydropyranoxy-hexyl sulfoxide

-
-
101387-22-8
2-ethyl-2-(3'-oxo-5'-tetrahydropyranoxy-)-hexyl-1,3-cyclopentanedione

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran for 24h; Heating; | 83% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
1395062-13-1
S-bromodifluoromethyl-S-phenyl-2,3,4,5-tetramethylphenylsulfonium triflate

-
-
1422736-27-3
3-(difluoromethoxy)-2-ethylcyclopent-2-en-1-one

| Conditions | Yield |
|---|---|
| Stage #1: 2-ethylcyclopentane-1,3-dione With 1-ethyl-2,2,4,4,4-pentakis(dimethylamino)-2λ5,4λ5-catenadi(phosphazene) In dichloromethane at 0℃; for 0.5h; Inert atmosphere; Stage #2: S-bromodifluoromethyl-S-phenyl-2,3,4,5-tetramethylphenylsulfonium triflate In dichloromethane at 0℃; for 1h; Inert atmosphere; regioselective reaction; | 82% |
-
-
383-62-0
ethyl 2-chloro-2,2-difluoroacetate

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
1422736-27-3
3-(difluoromethoxy)-2-ethylcyclopent-2-en-1-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In N,N-dimethyl-formamide at 60℃; for 7h; Sealed tube; Inert atmosphere; | 82% |
-
-
1629-60-3
1-hexene-3-one

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
77814-26-7
2-Ethyl-2-(3-oxohexyl)cyclopentane-1,3-dione

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol for 6h; Heating; | 77% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
124-40-3
dimethyl amine

-
-
134622-72-3
3-Dimethylamino-2-ethyl-cyclopent-2-enone

| Conditions | Yield |
|---|---|
| With acetic acid; propionic acid In toluene for 6h; Heating; | 77% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
55985-67-6
3-(2,4-Dimethoxy-phenyl)-5-methoxy-pent-1-en-3-ol

-
-
114506-69-3
2-ethyl-2-<5-methoxy-3-(2,4-dimethoxyphenyl)pent-2-enyl>cyclopentane-1,3-dione

| Conditions | Yield |
|---|---|
| With N-benzyl-trimethylammonium hydroxide In xylene Heating; | 76% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
101750-87-2
3-oxo-5-phenylthio-amyl mesylate

-
-
101750-88-3
2-ethyl-2-(3'-oxo-5'-phenylthio-amyl)-cyclopentane-1,3-dione

| Conditions | Yield |
|---|---|
| With triethylamine; hydroquinone In tetrahydrofuran for 14h; Heating; | 75% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
106-95-6
allyl bromide

-
B
-
59949-74-5
2-ethyl-2-(prop-2-enyl)cyclopentane-1,3-dione

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 60℃; for 10h; | A n/a B 75% |
| With sodium hydroxide In water at 60℃; for 10h; Yields of byproduct given; |

-
-
26825-94-5
10-bromodecanoic acid methyl ester

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

| Conditions | Yield |
|---|---|
| With sodium hydride In dimethyl sulfoxide at 20℃; for 24h; | 74% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
56475-35-5
5-Methoxy-3-(4-methoxy-phenyl)-pent-1-en-3-ol

-
-
114506-70-6
2-ethyl-2-<5-methoxy-3-(p-methoxyphenyl)pent-2-enyl>cyclopentane-1,3-dione

| Conditions | Yield |
|---|---|
| With N-benzyl-trimethylammonium hydroxide In xylene Heating; | 73% |

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

| Conditions | Yield |
|---|---|
| In ethanol; water at 25℃; for 48h; | 71.6% |
-
-
110-91-8
morpholine

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
134622-75-6
2-Ethyl-3-morpholin-4-yl-cyclopent-2-enone

| Conditions | Yield |
|---|---|
| With acetic acid; propionic acid In toluene for 6h; Heating; | 71% |
-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
106-96-7
propargyl bromide

-
B
-
71450-35-6
2-ethyl-2-(prop-2-ynyl)cyclopentane-1,3-dione

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 60℃; for 7.5h; | A n/a B 70% |

-
-
823-36-9
2-ethyl-1,3-cyclopentanedione

-
-
1422736-27-3
3-(difluoromethoxy)-2-ethylcyclopent-2-en-1-one

| Conditions | Yield |
|---|---|
| In para-xylene at 60℃; for 3h; Schlenk technique; Inert atmosphere; | 67% |
2-Ethyl-1,3-cyclopentanedione Specification
The 2-Ethyl-1,3-cyclopentanedione, with the CAS registry number 823-36-9, is also known as 1,3-Cyclopentanedione, 2-ethyl-. It belongs to the product categories of Miscellaneous; Ketone. Its EINECS number is 212-512-3. This chemical's molecular formula is C7H10O2 and molecular weight is 126.15. What's more, its systematic name is 2-ethylcyclopentane-1,3-dione. When using it, you must avoid contact with eyes. It is stable at common pressure and temperature, and it should be sealed and stored in a cool and dry place. Moreover, it should be protected from strong oxidants. It is used as a drug intermediate.
Physical properties of 2-Ethyl-1,3-cyclopentanedione are: (1)ACD/LogP: -0.25; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.25; (4)ACD/LogD (pH 7.4): -0.25; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 17.41; (8)ACD/KOC (pH 7.4): 17.41; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 1; (12)Polar Surface Area: 34.14 Å2; (13)Index of Refraction: 1.459; (14)Molar Refractivity: 32.64 cm3; (15)Molar Volume: 119.2 cm3; (16)Polarizability: 12.94×10-24cm3; (17)Surface Tension: 34.4 dyne/cm; (18)Density: 1.057 g/cm3; (19)Flash Point: 84.8 °C; (20)Enthalpy of Vaporization: 46.96 kJ/mol; (21)Boiling Point: 232.9 °C at 760 mmHg; (22)Vapour Pressure: 0.0574 mmHg at 25°C.
Preparation: this chemical can be prepared by 2-acetylcyclopentane-1,3-dione at the ambient temperature. This reaction will need reagents boron trifluoride etherate, triethylsilane and solvent trifluoroacetic acid. The yield is about 96%.

Uses of 2-Ethyl-1,3-cyclopentanedione: it can be used to produce 2-ethyl-3-pyrrolidin-1-yl-cyclopent-2-enone by heating. It will need reagent propanoic acid and solvent toluene with the reaction time of 6 hours. The yield is about 92%.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C1CCC(=O)C1CC
(2)Std. InChI: InChI=1S/C7H10O2/c1-2-5-6(8)3-4-7(5)9/h5H,2-4H2,1H3
(3)Std. InChIKey: YDFBIBUYOUFJMR-UHFFFAOYSA-N
Related Products
- 2-Ethyl-1-(3-methyl-1-oxo-2-butenyl)piperidine
- 2-Ethyl-1,3-cyclopentanedione
- 2-Ethyl-1,3-hexanediol
- 2-Ethyl-1,4-dimethoxybenzene
- 2-Ethyl-1-butanol
- 2-Ethyl-1-butene
- 2-Ethyl-1-hexanethiol
- 2-Ethyl-1-hexanol silicate
- 2-Ethyl-1-Hexene
- 2-Ethyl-1H-indene
- 82-33-7
- 82338-74-7
- 82338-76-9
- 82338-98-5
- 82339-08-0
- 823-39-2
- 82339-27-3
- 823-40-5
- 82344-98-7
- 823-45-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View