-
Name
O-CYMENE
- EINECS 208-426-0
- CAS No. 527-84-4
- Article Data94
- CAS DataBase
- Density 0.877 g/mL at 25 °C(lit.)
- Solubility
- Melting Point ?72-?71 °C(lit.)
- Formula C10H14
- Boiling Point 178 °C(lit.)
- Molecular Weight 134.221
- Flash Point 50°C
- Transport Information UN 2046 3
- Appearance
- Safety Moderately toxic by ingestion and intraperitoneal routes. When heated to decomposition it emits acrid smoke and irritating vapors.
- Risk Codes 10
-
Molecular Structure
- Hazard Symbols
- Synonyms o-Cymene(8CI); 1-Isopropyl-2-methylbenzene; 1-Methyl-2-(1-methylethyl)benzene;1-Methyl-2-isopropylbenzene; 2-Isopropyltoluene; NSC 73976; o-Cymol;o-Isopropyltoluene
- PSA 0.00000
- LogP 3.11840
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium phosphate monohydrate; bis[chloro(1,2,3-trihapto-allylbenzene)palladium(II)]; C20H34O3P2 In toluene at 100℃; Suzuki-Miyaura Coupling; Inert atmosphere; | 99% |
-
-
31689-42-6
2-methyl-1-(1-methylethyl)cyclohexa-2,5-diene-1-carboxylic acid

-
-
527-84-4
2-methylisopropylbenzene

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid In dichloromethane at 0℃; for 0.166667h; | 98% |
| With chlorosulfonic acid In dichloromethane at 0℃; for 0.166667h; | 98% |
-
-
31689-42-6
2-methyl-1-(1-methylethyl)cyclohexa-2,5-diene-1-carboxylic acid

-
A
-
527-84-4
2-methylisopropylbenzene

| Conditions | Yield |
|---|---|
| With oxygen; 5,15,10,20-tetraphenylporphyrin In chloroform at -30℃; for 24h; Irradiation; | A 28% B 68% |

-
-
527-84-4
2-methylisopropylbenzene

| Conditions | Yield |
|---|---|
| at 240 - 300℃; Leiten ueber Platin-Katalysatoren; |

| Conditions | Yield |
|---|---|
| With nickel at 90℃; under 88260.9 Torr; Hydrogenation; | |
| With nickel kieselguhr at 40℃; under 44130.5 Torr; Hydrogenation; | |
| With ethanol; sodium |

| Conditions | Yield |
|---|---|
| With copper oxide-chromium oxide at 100℃; under 88260.9 Torr; Hydrogenation; | |
| Multi-step reaction with 2 steps 1: acid 2: hydrogen / 10percent palladium on charcoal / methanol View Scheme | |
| Multi-step reaction with 2 steps 1: Destillation 2: Raney nickel / 90 °C / 88260.9 Torr / Hydrogenation View Scheme | |
| Multi-step reaction with 2 steps 1: 350 °C / beim Leiten ueber ThO2 2: hydrogen; nickel substance / 200 - 220 °C View Scheme | |
| Multi-step reaction with 2 steps 1: potassium pyrosulfate / durch Destillation 2: colloid/al palladium / Hydrogenation View Scheme |
-
-
104-15-4
toluene-4-sulfonic acid

-
-
67-63-0
isopropyl alcohol

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
17821-02-2
1-methyl-2,5-diisopropylbenzene

| Conditions | Yield |
|---|---|
| at 40℃; folgende Wasserdampfdestillation bei 150-160grad; | |
| With sulfuric acid at 40℃; anschliessend Destillation mit Wasserdampf bei 150-160grad; |

-
-
187737-37-7
propene

-
-
109-63-7
trifluoroborane diethyl ether

-
-
108-88-3
toluene

-
-
71-43-2
benzene

-
-
527-84-4
2-methylisopropylbenzene

| Conditions | Yield |
|---|---|
| at 5℃; und 65grad; | |
| at 40℃; Kinetics; |


| Conditions | Yield |
|---|---|
| With sodium |

| Conditions | Yield |
|---|---|
| Multistep reaction; |
-
-
98-82-8
Isopropylbenzene

-
-
14531-53-4
methyl cation

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
535-77-3
m-cymene

-
C
-
99-87-6
4-methylisopropylbenzene

| Conditions | Yield |
|---|---|
| Product distribution; Rate constant; with totally tritiated methyl cation, gas and liquid phases at r.t., solid phase at temp. of liquid N2; | A 11 % Chromat. B 39 % Chromat. C 50 % Chromat. |

-
-
74-98-6
propane

-
-
7450-03-5
(2-methylphenyl)trimethylsilane

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
535-77-3
m-cymene

-
C
-
99-87-6
4-methylisopropylbenzene

-
D
-
108-88-3
toluene

| Conditions | Yield |
|---|---|
| With methanol; oxygen; triethylamine Product distribution; Mechanism; reaction of isomeric trimethylsilyltoluenes in gas phase; investigated by mass spectrometric and radiolytic techniques; |

-
-
7399-49-7
1-methyl-2-(prop-1-en-2-yl)benzene

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
60264-83-7
o-1,4-menthadiene

-
C
-
60264-85-9
o-3,6-menthadiene

-
D
-
15582-48-6
o-1-menthene

| Conditions | Yield |
|---|---|
| With hydrogen; calcium hexammine In hexane for 2h; Further byproducts given; | A 92.0 % Chromat. B 4.2 % Chromat. C 0.5 % Chromat. D 0.7 % Chromat. |

-
-
7399-49-7
1-methyl-2-(prop-1-en-2-yl)benzene

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
15582-50-0
o-6-menthene

-
C
-
15582-48-6
o-1-menthene

-
D
-
15582-49-7
o-2-menthene

| Conditions | Yield |
|---|---|
| With hydrogen; calcium hexammine for 24h; Further byproducts given; | A 79.0 % Chromat. B 5.3 % Chromat. C 9.1 % Chromat. D 4.8 % Chromat. |
| With hydrogen; calcium hexammine for 24h; Further byproducts given; | A 79.0 % Chromat. B 5.3 % Chromat. C 9.1 % Chromat. D 4.2 % Chromat. |

-
-
7399-49-7
1-methyl-2-(prop-1-en-2-yl)benzene

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
60264-83-7
o-1,4-menthadiene

-
C
-
60264-85-9
o-3,6-menthadiene

-
D
-
108-88-3
toluene

| Conditions | Yield |
|---|---|
| With hydrogen; calcium hexammine In hexane for 2h; Further byproducts given; | A 96 % Chromat. B 1.70 % Chromat. C 0.2 % Chromat. D 1.1 % Chromat. |

-
-
7399-49-7
1-methyl-2-(prop-1-en-2-yl)benzene

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
60264-83-7
o-1,4-menthadiene

-
C
-
60264-85-9
o-3,6-menthadiene

-
D
-
15582-48-6
o-1-menthene

-
E
-
15582-49-7
o-2-menthene

-
F
-
108-88-3
toluene

| Conditions | Yield |
|---|---|
| With hydrogen; calcium hexammine In hexane for 2h; Product distribution; other mole ratio of hydrogen, other reaction time, other solvent; | A 96.0 % Chromat. B 1.7 % Chromat. C 0.2 % Chromat. D 0.2 % Chromat. E 0.1 % Chromat. F 1.1 % Chromat. |

-
-
60264-83-7
o-1,4-menthadiene

-
-
527-84-4
2-methylisopropylbenzene

| Conditions | Yield |
|---|---|
| With lithium 2-aminoethylamide at 100℃; for 1.5h; | 100 % Chromat. |
| With calcium amide In hexane at 70℃; for 5h; Product distribution; var. react. time, Ca(NH2) to o-1,4-menthadiene 5:1; | 94 % Chromat. |
-
-
60264-83-7
o-1,4-menthadiene

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
15582-50-0
o-6-menthene

-
C
-
15582-48-6
o-1-menthene

-
D
-
15582-49-7
o-2-menthene

-
-
6070-02-6, 6070-03-7, 16580-23-7, 19489-02-2, 19489-03-3
cis-o-menthane

-
-
6070-02-6, 6070-03-7, 16580-23-7, 19489-02-2, 19489-03-3
trans-o-menthane

| Conditions | Yield |
|---|---|
| With zeolite NaX In octane at 120℃; for 12h; Product distribution; var. temperature, oth. zeolite; | A 77 % Chromat. B 2.7 % Chromat. C 10.5 % Chromat. D 3.4 % Chromat. E 2 % Chromat. F 0.5 % Chromat. |

-
-
60264-83-7
o-1,4-menthadiene

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
o-1,5-menthadiene -
527-83-3
o-1,5-menthadiene

-
C
-
72188-50-2
o-1,3-menthadiene

-
D
-
71889-93-5
1-isopropyl-5-ethylidenecyclopentane

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In dimethyl sulfoxide at 25℃; for 80h; Product distribution; var. react. time, oth. temperature; | A 24 % Chromat. B 34 % Chromat. C 25 % Chromat. D 17 % Chromat. |

-
-
60264-83-7
o-1,4-menthadiene

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
527-83-3
o-1,5-menthadiene

-
C
-
72188-50-2
o-1,3-menthadiene

-
D
-
99805-90-0
4-methyl-3-(1-methylethylidene)-1-cyclohexene

-
E
-
99805-89-7
o-1(7),4-menthadiene

-
F
-
99805-91-1
o-4,6-menthadiene

| Conditions | Yield |
|---|---|
| With aluminosilicate diatomaceous earth In hexane at 270℃; for 0.00694444h; Product distribution; Mechanism; other time, other temperature; |

-
-
60264-83-7
o-1,4-menthadiene

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
71889-93-5
1-isopropyl-5-ethylidenecyclopentane

| Conditions | Yield |
|---|---|
| With silica gel In hexane at 350℃; for 0.014h; Product distribution; in a stream of nitrogen, oth. temperature; | A 52 % Chromat. B 39.5 % Chromat. |
-
-
54580-22-2
2,5-dimethyl-4-isopropyl-3-isopropylidenehexa-1,4-diene

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
106-42-3
para-xylene

| Conditions | Yield |
|---|---|
| at 750℃; under 0.003 Torr; for 2.77778E-07h; Further byproducts given; |
-
-
126949-89-1
(1α,6α)-6,9,9-Trimethyl-7,8-diazabicyclo<4.3.0>nona-2,5,7-trien

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
2228-74-2
1,7,7-Trimethyl-tropiliden

-
C
-
2228-73-1
2,7,7-Trimethyl-cycloheptatrien

| Conditions | Yield |
|---|---|
| In benzene at 28℃; other solvents C6D6 and CDCl3, other temperatures; also by photolysis; | A 3 % Chromat. B 3 % Chromat. C 94 % Chromat. |
| In benzene-d6 at 39.2℃; Mechanism; Kinetics; Product distribution; energy data: ΔH(excit.), ΔS(excit.), ΔG(excit.); other solvents C6H6 or CDCl3, other temperatures; other educt (1α,6α)-6-Trideuteriomethyl-2,9,9-trimethyl-7,8-diazabicyclo<4.3.0>nona-2,4,7-trien; | A 3 % Chromat. B 3 % Chromat. C 94 % Chromat. |

-
-
69856-67-3
3,3,3',3',5,5,5',5'-octamethyl-3,3',5,5'-tetrahydro-4,4'-bi(4H-pyrazolylidene)

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
106-42-3
para-xylene

-
C
-
54580-22-2
2,5-dimethyl-4-isopropyl-3-isopropylidenehexa-1,4-diene

-
D
-
94526-06-4
1-isopropylidene-2,2,4,4,5,5-hexamethylspiropentane

-
E
-
94526-08-6
2-(2,3-dimethylbut-1-en-3-yl)-1-isopropyl-3,3-dimethylcyclopropene

| Conditions | Yield |
|---|---|
| at 400 - 800℃; for 2.77778E-07h; Product distribution; |

-
-
136667-03-3
α-(o-tolyl)isobutyrophenone

-
A
-
7399-49-7
1-methyl-2-(prop-1-en-2-yl)benzene

-
B
-
527-84-4
2-methylisopropylbenzene

-
C
-
100-52-7
benzaldehyde

| Conditions | Yield |
|---|---|
| In benzene Quantum yield; Ambient temperature; Irradiation; |

-
-
99805-92-2
trans-o-4-menthen-8-ol

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
60264-83-7
o-1,4-menthadiene

-
C
-
15582-48-6
o-1-menthene

-
D
-
13427-84-4
o-2(8),3-menthadiene

-
E
-
99805-93-3
o-2(8),4-menthadiene

-
-
99805-94-4
trans-o-4,8-menthadiene

| Conditions | Yield |
|---|---|
| With potassium hydrogensulfate Product distribution; 1.)140 deg C, 1 h 2.)180 deg C, 15 min; |

-
-
99805-92-2
trans-o-4-menthen-8-ol

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
60264-83-7
o-1,4-menthadiene

-
C
-
15582-48-6
o-1-menthene

-
D
-
99805-93-3
o-2(8),4-menthadiene

-
-
99805-94-4
trans-o-4,8-menthadiene

| Conditions | Yield |
|---|---|
| With acetic anhydride at 150℃; for 12h; Product distribution; sealed ampoule; |

-
-
77804-19-4, 77849-06-0
methyl-3 dimethylcarbinol-4 cyclohexene trans

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
60264-83-7
o-1,4-menthadiene

-
C
-
54356-60-4
o-2(8),5-menthadiene

-
D
-
527-83-3
o-1,5-menthadiene

-
-
54356-60-4, 99805-95-5, 99805-96-6
trans-o-5,8-menthadiene

| Conditions | Yield |
|---|---|
| With potassium hydrogensulfate Product distribution; 1.)140 deg C, 1 h 2.)180 deg C, 15 min; |

-
-
77804-19-4, 77849-06-0
methyl-3 dimethylcarbinol-4 cyclohexene trans

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
13427-84-4
o-2(8),3-menthadiene

-
C
-
54356-60-4
o-2(8),5-menthadiene

-
D
-
527-83-3
o-1,5-menthadiene

-
-
54356-60-4, 99805-95-5, 99805-96-6
cis-o-5,8-menthadiene

-
-
54356-60-4, 99805-95-5, 99805-96-6
trans-o-5,8-menthadiene

| Conditions | Yield |
|---|---|
| With acetic anhydride at 150℃; for 12h; Product distribution; sealed ampoule; |

-
-
77804-19-4, 77849-06-0
methyl-3 dimethylcarbinol-4 cyclohexene cis

-
A
-
527-84-4
2-methylisopropylbenzene

-
B
-
13427-84-4
o-2(8),3-menthadiene

-
C
-
54356-60-4
o-2(8),5-menthadiene

-
D
-
527-83-3
o-1,5-menthadiene

-
-
54356-60-4, 99805-95-5, 99805-96-6
cis-o-5,8-menthadiene

-
-
54356-60-4, 99805-95-5, 99805-96-6
trans-o-5,8-menthadiene

| Conditions | Yield |
|---|---|
| With acetic anhydride at 150℃; for 12h; Product distribution; sealed ampoule; |

-
-
527-84-4
2-methylisopropylbenzene

-
A
-
6502-22-3
2-isopropylbenzaldehyde

-
B
-
128260-56-0
1-Isopropyl-2-nitromethyl-benzene

-
C
-
128260-55-9
1-Isopropyl-2-nitrooxymethyl-benzene

| Conditions | Yield |
|---|---|
| With ammonium cerium(IV) nitrate In acetonitrile at 17℃; for 0.666667h; Irradiation; | A 14% B 5% C 69% |

-
-
527-84-4
2-methylisopropylbenzene

-
-
434-45-7
1,1,1-trifluoroacetophenone

-
A
-
1547-69-9
meso-1,1,1,4,4,4-Hexafluor-2,3-diphenylbutandiol(2,3)

-
B
-
118893-11-1
C20H26

-
C
-
118893-12-2
1,1,1-Trifluoro-3-(2-isopropyl-phenyl)-2-phenyl-propan-2-ol

| Conditions | Yield |
|---|---|
| In acetonitrile at 30℃; | A 35% B 7% C 43% |

-
-
527-84-4
2-methylisopropylbenzene

-
A
-
1547-69-9
meso-1,1,1,4,4,4-Hexafluor-2,3-diphenylbutandiol(2,3)

-
B
-
118893-11-1
C20H26

-
C
-
118893-12-2
1,1,1-Trifluoro-3-(2-isopropyl-phenyl)-2-phenyl-propan-2-ol

| Conditions | Yield |
|---|---|
| With 2,2,2-Trifluoroacetophenone In acetonitrile at 30℃; | A 35% B 7% C 43% |

-
-
527-84-4
2-methylisopropylbenzene

-
A
-
4571-14-6
2-isopropyl-5-nitrotoluene

-
B
-
175540-88-2
2-isopropyl-3-nitrotoluene

-
C
-
103096-00-0
2-isopropyl-4-nitrotoluene

-
D
-
175540-89-3
2-isopropyl-6-nitrotoluene

| Conditions | Yield |
|---|---|
| With nitric acid In sulfuric acid at 25℃; for 0.0833333h; Further byproducts given; | A 37.6% B 3.8% C 27.8% D 12.7% |
| With nitric acid In sulfuric acid at 25℃; Further byproducts given; | A 35.3% B 3.8% C 27.1% D 12.8% |
| With nitric acid In sulfuric acid at 25℃; Further byproducts given; | A 34.5% B 3.7% C 27.9% D 12.3% |

-
-
527-84-4
2-methylisopropylbenzene

-
A
-
88-72-2
1-methyl-2-nitrobenzene

-
B
-
4571-14-6
2-isopropyl-5-nitrotoluene

-
C
-
175540-88-2
2-isopropyl-3-nitrotoluene

-
D
-
103096-00-0
2-isopropyl-4-nitrotoluene

| Conditions | Yield |
|---|---|
| With nitric acid In sulfuric acid at 25℃; for 2h; Further byproducts given; | A 23% B 35.3% C 3.8% D 27.1% |
| With nitric acid In sulfuric acid at 25℃; for 2h; Further byproducts given; | A 23% B 35.3% C 3.8% D 27.1% |

-
-
119-61-9
benzophenone

-
-
527-84-4
2-methylisopropylbenzene

-
A
-
464-72-2
tetraphenylethane-1,2-diol

-
B
-
118893-14-4
2-Methyl-1,1-diphenyl-2-o-tolyl-propan-1-ol

-
C
-
118893-13-3
2-(2-Isopropyl-phenyl)-1,1-diphenyl-ethanol

| Conditions | Yield |
|---|---|
| In acetonitrile at 30℃; Irradiation; | A 35% B 4% C 13% |

-
-
527-84-4
2-methylisopropylbenzene

-
-
3029-30-9
naphthalene-1,4-dicarbonitrile

-
A
-
118893-08-6
4-(2-Isopropyl-benzyl)-naphthalene-1-carbonitrile

-
-
118893-09-7
(1R,2R)-2-(2-Isopropyl-benzyl)-1,2-dihydro-naphthalene-1,4-dicarbonitrile

-
-
118893-10-0
C22H20N2

| Conditions | Yield |
|---|---|
| In acetonitrile at 17℃; for 20h; | A 15% B 23% C 27% |


| Conditions | Yield |
|---|---|
| With 5% platinum on alumina at 300℃; | |
| at 300℃; Leiten ueber Platin-Aluminiumoxyd-Kontakte; |
-
-
527-84-4
2-methylisopropylbenzene

-
A
-
87-83-2
pentabromotoluene

-
B
-
854403-37-5
1,2,3-tribromo-5-isopropyl-4-methyl-benzene

| Conditions | Yield |
|---|---|
| With sulfuric acid; bromine; iodine; iron at 0℃; Lichtausschluss; |
-
-
527-84-4
2-methylisopropylbenzene

-
-
16580-23-7
1-isopropyl-2-methylcyclohexane

| Conditions | Yield |
|---|---|
| With hydrogen; nickel at 170 - 180℃; |

| Conditions | Yield |
|---|---|
| With chlorosulfonic acid; chloroform und anschliessend mit wss.NH3; |
-
-
527-84-4
2-methylisopropylbenzene

-
-
13387-56-9
2-(o-tolyl)propyl-2-hydroperoxide

| Conditions | Yield |
|---|---|
| With oxygen; lead acetate |
-
-
527-84-4
2-methylisopropylbenzene

-
-
17821-06-6
4-bromo-1-isopropyl-2-methyl-benzene

| Conditions | Yield |
|---|---|
| With bromine; iodine; iron at 0℃; Lichtausschluss; |
-
-
527-84-4
2-methylisopropylbenzene

-
-
854403-39-7
1,2-dibromo-4-isopropyl-5-methyl-benzene

| Conditions | Yield |
|---|---|
| With bromine; iodine; iron at 0℃; Lichtausschluss; |
-
-
527-84-4
2-methylisopropylbenzene

-
-
854403-38-6
1,2,3,4-tetrabromo-5-isopropyl-6-methyl-benzene

| Conditions | Yield |
|---|---|
| With bromine; iodine; iron at 0 - 50℃; Lichtausschluss; |

| Conditions | Yield |
|---|---|
| at 410℃; Leiten ueber aktivierte Bleicherde; |
-
-
107-39-1
2,4,4-trimethyl-1-pentene

-
-
527-84-4
2-methylisopropylbenzene

-
-
29577-23-9
6-Tert-Butyl-1,1,3,4-tetramethyl-3-neopentyl indan

| Conditions | Yield |
|---|---|
| With sulfuric acid at 5℃; |
-
-
527-84-4
2-methylisopropylbenzene

-
-
60264-83-7
o-1,4-menthadiene

| Conditions | Yield |
|---|---|
| With calcium hexammine In hexane |

| Conditions | Yield |
|---|---|
| With nitronium tetrafluoborate In sulfolane |
-
-
527-84-4
2-methylisopropylbenzene

-
-
15582-50-0
o-6-menthene

| Conditions | Yield |
|---|---|
| With calcium hexammine In hexane |
-
-
527-84-4
2-methylisopropylbenzene

-
-
60264-85-9
o-3,6-menthadiene

| Conditions | Yield |
|---|---|
| With calcium hexammine In hexane |
2-Isopropyltoluene Chemical Properties
IUPAC Name: 1-Methyl-2-propan-2-ylbenzene
Molecular Formula: C10H14
Molecular Weight: 134.24g/mol
EINECS: 208-426-0
Density: 0.877 g/mL at 25 °C(lit.)
Melting Point: 72-71 °C(lit.)
Boiling Point: 178.1 °C at 760 mmHg
Flash Point: 50.6 °C
liansport Information: UN 2046 3
Freely Rotating Bonds: 1
Polar Surface Area: 0 Å2
Index of Refraction: 1.492
Molar Refractivity: 45.26 cm3
Molar Volume: 155.7 cm3
Polarizability: 17.94 ×10-24 cm3
Surface Tension: 28.5 dyne/cm
Enthalpy of Vaporization: 39.74 kJ/mol
Vapour Pressure: 1.36 mmHg at 25°C
The Cas Register Number of 2-Isopropyltoluene is 527-84-4.The chemical synonyms of 2-Isopropyltoluene (CAS NO.527-84-4) are O-Cymene ; 1-(1-Methylethyl)-2-methylbenzene ; 1-Methyl,2-n-isopropylbenzene ; 1-Methyl-2-(1-methylethyl)-benzen ; 1-Methyl-2-(1-methylethyl)benzene ; 1-Methyl-2-(1-methylethyl)-benzene ; 1-Methyl-2-isopropylbenzol ; Benzene, 1-methyl-2-(1-methylethyl) .The molecular structure of 2-Isopropyltoluene (CAS NO.527-84-4) is 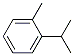 .
.
2-Isopropyltoluene Toxicity Data With Reference
| 1. | orl-rat LD50:2130 mg/kg | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 29 (3)(1985),49. | ||
| 2. | orl-mus LD50:2024 mg/kg | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 29 (3)(1985),49. | ||
| 3. | ihl-mus LC50:10,300 mg/m3 | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 29 (3)(1985),49. | ||
| 4. | ipr-mus LD50:1315 mg/kg | GTPZAB Gigiena Truda i Professionalnye Zabolevaniia. Labor Hygiene and Occupational Diseases. 29 (3)(1985),49. |
2-Isopropyltoluene Consensus Reports
Reported in EPA TSCA Inventory.
2-Isopropyltoluene Safety Profile
Moderately toxic by ingestion and intraperitoneal routes. When heated to decomposition it emits acrid smoke and irritating vapors.
Risk Statements:10
R10:Flammable.
Safety Statements:23-24/25
S23:Do not breathe vapour.
S24/25:Avoid contact with skin and eyes.
HazardClass:3.2
2-Isopropyltoluene Specification
Downstream products of 2-Isopropyltoluene (CAS NO.527-84-4) is Cresol .
Related Products
- 2-Isopropyltoluene
- 52785-06-5
- 527-85-5
- 52786-55-7
- 52787-14-1
- 52-78-8
- 52788-02-0
- 52788-53-1
- 52789-62-5
- 52789-73-8
- 52791-05-6
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View