-
Name
3-Dimethylaminopropylchloride hydrochloride
- EINECS 226-467-2
- CAS No. 5407-04-5
- Article Data9
- CAS DataBase
- Density
- Solubility soluble in water
- Melting Point 187-190 °C(lit.)
- Formula C5H13Cl2N
- Boiling Point 130.7 °C at 760 mmHg
- Molecular Weight 158.071
- Flash Point 32.8 °C
- Transport Information
- Appearance white to yellowish crystalline powder
- Safety 26-61-37/39-36
- Risk Codes 22-36/37/38-52/53
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn,  Xi
Xi
- Synonyms Propylamine,3-chloro-N,N-dimethyl-, hydrochloride (7CI,8CI);(3-Chloropropyl)dimethylaminehydrochloride;(3-Chloropropyl)dimethylammonium chloride;1-Chloro-3-(dimethylamino)propane hydrochloride;1-Dimethylamino-3-chloropropane hydrochloride;3-(Dimethylamino)-1-chloropropane hydrochloride;3-(Dimethylamino)-1-propylchloride hydrochloride;3-Chloro-1-(N,N-dimethylamino)propane hydrochloride;3-Chloro-N,N-dimethylpropane-1-amine hydrochloride;N,N-Dimethyl-3-chloropropylamine hydrochloride;N-(3-Chloropropyl)dimethylamine hydrochloride;
- PSA 3.24000
- LogP 1.97890
Synthetic route

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride In chloroform at 0℃; for 5h; Reflux; | 98% |
| With thionyl chloride In chloroform Reflux; Cooling with ice; | |
| With thionyl chloride In dichloromethane at 0 - 60℃; for 2h; |
-
-
124-40-3
dimethyl amine

-
-
107-05-1
3-chloroprop-1-ene

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| With diatomaceous earth In toluene at 45℃; for 10h; Reagent/catalyst; Temperature; Solvent; | 90% |
-
-
124-40-3
dimethyl amine

-
-
627-30-5
1-chloro-3-hydroxypropane

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: dimethyl amine; 1-chloro-3-hydroxypropane for 4h; Reflux; Stage #2: With thionyl chloride In tetrachloromethane at 20℃; for 4h; Cooling with ice; | 60.8% |
-
-
50-00-0
formaldehyd

-
-
6276-54-6
3-chloropropylamine hydrochloride

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| With formic acid for 30h; Heating; | 30% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
132461-45-1
3,5-Dimethyl-11H-10,11-diaza-benzo[b]fluorene

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In sodium hydroxide; toluene Heating; | 100% |

-
-
14982-15-1
3-benzyloxyestra-1,3,5(10)-trien-17β-ol

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
144-55-8
sodium hydrogencarbonate

| Conditions | Yield |
|---|---|
| With tetra-(n-butyl)ammonium iodide In N,N-dimethyl-formamide; mineral oil | 100% |

-
-
487-89-8
Indole-3-carboxaldehyde

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
169120-64-3
1-(3-dimethylaminopropyl)-1H-indole-3-carbaldehyde

| Conditions | Yield |
|---|---|
| Stage #1: Indole-3-carboxaldehyde With caesium carbonate; potassium iodide In acetonitrile at 0℃; for 0.166667h; Stage #2: 3-(dimethylamino)propyl chloride hydrochloride In acetonitrile at 0℃; for 18h; Reflux; | 100% |
| Stage #1: Indole-3-carboxaldehyde With caesium carbonate; potassium iodide In acetonitrile at 0℃; Inert atmosphere; Stage #2: 3-(dimethylamino)propyl chloride hydrochloride In acetonitrile at 83℃; for 28h; Inert atmosphere; | 89% |
| With caesium carbonate In ethanol for 16h; Reflux; | 42% |

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 2,4,7‑trifluoro‑9H‑carbazole With sodium hydride In N,N-dimethyl-formamide; mineral oil at 20℃; for 0.166667h; Stage #2: 3-(dimethylamino)propyl chloride hydrochloride In N,N-dimethyl-formamide; mineral oil at 50℃; for 2.5h; | 100% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 90℃; Inert atmosphere; Schlenk technique; | 100% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
66715-60-4
3-iodo-N,N-dimethylpropylamine

| Conditions | Yield |
|---|---|
| Stage #1: 3-(dimethylamino)propyl chloride hydrochloride With sodium iodide In cyclohexane; water at 80℃; for 3h; Inert atmosphere; Stage #2: With sodium hydroxide In cyclohexane; water at 0 - 10℃; Solvent; | 99.8% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
507-09-5
thioacetic acid

-
-
66338-43-0
S-3-(dimethylamino)propyl ethanethioate

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 70℃; Inert atmosphere; | 99% |
| With triethylamine In chloroform at 10℃; Inert atmosphere; Reflux; | 82% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| With sodium iodide In acetone for 24h; Reflux; | 99% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
134164-54-8
2-methoxy-11-methyl-6H-indolo[2,3-b]quinoline

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In sodium hydroxide; toluene Heating; | 98% |

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In sodium hydroxide; toluene Heating; | 98% |
-
-
33265-12-2
3-hydroxy-7-methoxy-2H-chromen-2-one

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 3-hydroxy-7-methoxy-2H-chromen-2-one With caesium carbonate In N,N-dimethyl-formamide at 20℃; for 0.5h; Inert atmosphere; Stage #2: 3-(dimethylamino)propyl chloride hydrochloride With caesium carbonate In N,N-dimethyl-formamide at 110℃; for 8h; Inert atmosphere; | 97% |

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 100℃; | 97% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
132445-62-6
9-methoxy-11-methyl-6H-indolo[2,3-b]quinoline

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In sodium hydroxide; toluene Heating; | 96% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
125157-84-8
2-fluoro-11-methyl-6H-indolo[2,3-b]quinoline

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In sodium hydroxide; toluene Heating; | 96% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
106-53-6
para-bromobenzenethiol

-
-
403793-24-8
1-[3-(N,N-Dimethylamino)propylthio]-4-bromobenzene

| Conditions | Yield |
|---|---|
| With potassium carbonate In DMF (N,N-dimethyl-formamide) at 60℃; for 0.25h; | 96% |
| With potassium carbonate In DMF (N,N-dimethyl-formamide) at 60℃; for 0.25h; | 96% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
74-89-5
methylamine

-
-
4543-96-8
N,N,N'-trimethyl-1,3-propanediamine

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 25℃; | 96% |
-
-
7725-93-1
1-(4-methyl-2-thioxo-2,3-dihydrothiazol-5-yl)ethan-1-one

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 1-(4-methyl-2-thioxo-2,3-dihydrothiazol-5-yl)ethan-1-one With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 1h; Stage #2: 3-(dimethylamino)propyl chloride hydrochloride In N,N-dimethyl-formamide at 20℃; | 96% |
-
-
873-62-1
m-cyanophenol

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
182963-94-6
3-(3-(aminomethyl)phenoxy)-N,N-dimethylpropan-1-amine

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; water; N,N-dimethyl-formamide | 95% |

| Conditions | Yield |
|---|---|
| Stage #1: C16H12ClNO With sodium hydride In N,N-dimethyl-formamide for 0.5h; Stage #2: 3-(dimethylamino)propyl chloride hydrochloride In N,N-dimethyl-formamide at 80℃; for 48h; | 95% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
125157-83-7
5,7-Dimethyl-11H-10,11-diaza-benzo[b]fluorene

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In sodium hydroxide; toluene Heating; | 94% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
91221-46-4
1,1-bis(4-hydroxyphenyl)-2-phenyl-1-butene

-
-
1020853-04-6
1,1-bis{4-[3-(dimethylamino)propoxy]phenyl}-2-phenyl-1-butene

| Conditions | Yield |
|---|---|
| Stage #1: 1,1-bis(4-hydroxyphenyl)-2-phenyl-1-butene With sodium hydride In N,N-dimethyl-formamide; paraffin oil at 50℃; for 0.25h; Stage #2: 3-(dimethylamino)propyl chloride hydrochloride In N,N-dimethyl-formamide; paraffin oil at 50℃; for 15h; | 94% |
-
-
1956-45-2, 92313-45-6, 92313-54-7
2-acetylthiophene oxime

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| With potassium iodide; potassium hydroxide In dimethyl sulfoxide at 20℃; for 1h; | 94% |

| Conditions | Yield |
|---|---|
| With potassium iodide; potassium hydroxide In dimethyl sulfoxide at 20℃; for 1h; | 94% |

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| In isopropyl alcohol at 53 - 55℃; for 3h; | 93.3% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
134164-59-3
9-fluoro-11-methyl-6H-indolo[2,3-b]quinoline

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide In sodium hydroxide; toluene Heating; | 93% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
548767-17-5
1-hydroxy-5-iodopyrazole

-
-
572909-06-9
1-(3-N,N-dimethylaminopropyloxy)-5-iodopyrazole

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide for 24h; | 93% |
-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

-
-
6146-52-7
5-nitroindole

-
-
954386-92-6
N,N-dimethyl-3-(5-nitro-1H-indol-1-yl)propan-1-amine

| Conditions | Yield |
|---|---|
| Stage #1: 5-nitroindole With sodium hydride In N,N-dimethyl-formamide; mineral oil at 0 - 5℃; for 0.25h; Stage #2: 3-(dimethylamino)propyl chloride hydrochloride at 0 - 50℃; for 20.25h; | 93% |
-
-
1416136-82-7
2-[3-(benzyloxy)phenyl]-1,1-bis(4-hydroxyphenyl)-1-butene

-
-
5407-04-5
3-(dimethylamino)propyl chloride hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: 2-[3-(benzyloxy)phenyl]-1,1-bis(4-hydroxyphenyl)-1-butene With sodium hydride In N,N-dimethyl-formamide; paraffin oil at 50℃; for 0.25h; Stage #2: 3-(dimethylamino)propyl chloride hydrochloride In N,N-dimethyl-formamide; paraffin oil at 50℃; for 22h; | 92% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 45℃; for 2h; | 91.07% |
| With triethylamine In diethyl ether at 20℃; | 83% |
| With sodium hydroxide In water at 45 - 50℃; for 2h; | 80% |
3-Dimethylaminopropylchloride hydrochloride Chemical Properties
Molecule structure of 1-Propanamine, 3-chloro-N,N-dimethyl-, hydrochloride (1:1) (CAS NO.5407-04-5):
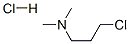
IUPAC Name: 3-Chloro-N,N-dimethylpropan-1-amine hydrochloride
Molecular Formula: C5H13Cl2N
Formula Weight: 158.07 g/mol
Melting Point: 187-190 °C(lit.)
Flash Point: 32.8 °C
Boiling Point: 130.7 °C at 760 mmHg
Vapour Pressure: 9.61 mmHg at 25 °C
Water Solubility: soluble
Sensitive: hygroscopic
Appearence: white to yellowish crystalline powder
H-Bond Donor: 1
H-Bond Acceptor: 1
Rotatable Bond Count: 3
Exact Mass: 157.042505
MonoIsotopic Mass: 157.042505
Topological Polar Surface Area: 3.2
Heavy Atom Count: 8
Canonical SMILES: CN(C)CCCCl.Cl
InChI: InChI=1S/C5H12ClN.ClH/c1-7(2)5-3-4-6;/h3-5H2,1-2H3;1H
InChIKey: LJQNMDZRCXJETK-UHFFFAOYSA-N
EINECS: 224-971-7
Classification Code: Mutation data; Tumor data
3-Dimethylaminopropylchloride hydrochloride Uses
1-Propanamine, 3-chloro-N,N-dimethyl-, hydrochloride (1:1) (CAS NO.5407-04-5) is used as intermediate for the syntheses of pharmaceuticals (e.g. cyclobenzaprine, imipramine, benzydamine, amitriptyline and bencyclane).
3-Dimethylaminopropylchloride hydrochloride Production
Put dimethylamine and propylene alcohol into the reaction tank, in the presence of sodium hydroxide, synthesizing at the temperature of 130-150 °C and under pressure of 127.5-147.1kPa, 1-dimethylamino-C-3-ol is obtained by reversed flow for 24h. Then use thionyl chloride to chlorination, 1-Propanamine, 3-chloro-N,N-dimethyl-, hydrochloride (1:1) is synthesized at the temperature of 55-90 °C for 9-10h.
3-Dimethylaminopropylchloride hydrochloride Toxicity Data With Reference
| 1. | mic-sat 10 mg/L | ENMUDM Environmental Mutagenesis. 3 (1981),33. |
3-Dimethylaminopropylchloride hydrochloride Consensus Reports
Reported in EPA TSCA Inventory.
3-Dimethylaminopropylchloride hydrochloride Safety Profile
Hazard Codes:  Xn,
Xn,  Xi
Xi
Risk Statements: 22-36/37/38-52/53
R22:Harmful if swallowed.
R36/37/38:Irritating to eyes, respiratory system and skin.
R52/53:Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment.
Safety Statements: 26-61-37/39-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S61:Avoid release to the environment. Refer to special instructions / safety data sheets.
S37/39:Wear suitable gloves and eye/face protection.
S36:Wear suitable protective clothing.
WGK Germany: 3
RTECS: TY1225000
F: 3
Hazard Note: Irritant
Mutation data reported. When heated to decomposition it emits toxic vapors of NOx and Cl−.
3-Dimethylaminopropylchloride hydrochloride Specification
1-Propanamine, 3-chloro-N,N-dimethyl-, hydrochloride (1:1) (CAS NO.5407-04-5) is also named as 1-Chloro-3-(dimethylamino)propane hydrochloride ; 3-(Dimethylamino)-1-propyl chloride hydrochloride ; 3-(Dimethylamino)chloropropane hydrochloride ; 3-(Dimethylamino)propyl chloride hydrochloride ; 3-Chloro-N',N'-dimethylaminopropane hydrochloride ; 3-Chloro-N,N-dimethyl-1-propanamine hydrochloride ; 3-Chloro-N,N-dimethyl-1-propylamine hydrochloride ; 3-Chloro-N,N-dimethylpropylamine hydrochloride ; AI3-26681 ; CCRIS 7054 ; Dimethylaminopropyl chloride, hydrochloride ; N,N-Dimethyl-3-chloropropylamine hydrochloride ; NSC 5368 .
Related Products
- 3-Dimethylaminopropylchloride hydrochloride
- 540-72-7
- 540-73-8
- 5407-51-2
- 5407-58-9
- 54076-73-2
- 54077-16-6
- 54078-29-4
- 5407-87-4
- 5407-91-0
- 5407-94-3
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View