-
Name
3-Methyl-2-buten-1-ol
- EINECS 209-141-4
- CAS No. 556-82-1
- Article Data189
- CAS DataBase
- Density 0.844 g/cm3
- Solubility water: 170 g/L (20 °C)
- Melting Point -86oC
- Formula C5H10O
- Boiling Point 141.5 °C at 760 mmHg
- Molecular Weight 86.1338
- Flash Point 43.3 °C
- Transport Information UN 1987 3/PG 3
- Appearance colorless liquid with fruity odor
- Safety 26-36-37-23-16
- Risk Codes 10-22-36/37/38-38-21/22
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms 2-Methyl-2-buten-4-ol;3,3-Dimethylallyl alcohol;3-Methyl-2-buten-1-ol;3-Methyl-2-butene-1-ol;3-Methyl-2-butenol;3-Methyl-2-butenyl alcohol;3-Methylcrotyl alcohol;Dimethylallyl alcohol;Isopent-2-en-1-ol;NSC 158709;Prenol;Prenyl alcohol;g,g-Dimethylallyl alcohol;
- PSA 20.23000
- LogP 0.94490
Synthetic route

| Conditions | Yield |
|---|---|
| With acetylacetonatodicarbonylrhodium(l); trifluorormethanesulfonic acid; carbon monoxide; N-(5-diphenylphosphanylpyrrole-2-carbonyl)guanidine; hydrogen In dichloromethane at 40℃; under 15001.5 Torr; for 20h; Autoclave; | 99% |
| With indium tri-isopropoxide supported on mesoporous SBA-15 In isopropyl alcohol at 80℃; for 7h; Reagent/catalyst; Meerwein-Ponndorf-Verley Reduction; Inert atmosphere; Schlenk technique; chemoselective reaction; | 89.6% |
| With Triisopropyl borate; isopropyl alcohol at 27℃; for 15h; Kinetics; Reagent/catalyst; Meerwein-Ponndorf-Verley reduction; Inert atmosphere; chemoselective reaction; | 87.9% |

| Conditions | Yield |
|---|---|
| With lithium hexafluorophosphate at 0℃; for 8h; Reagent/catalyst; Temperature; Irradiation; Inert atmosphere; | 97% |
| With palladium 10% on activated carbon; oxygen at 80℃; under 3750.38 Torr; for 5h; Reagent/catalyst; Time; Concentration; | 53.05% |
| With pyridine; 2-methyl-2-phenyloxirane; iron pentacarbonyl; carbon monoxide at 70℃; under 3750.38 Torr; for 42h; Reagent/catalyst; Temperature; Inert atmosphere; Autoclave; | |
| With 1,3-bis-(diphenylphosphino)propane; hydrogen; palladium diacetate In acetone at 55℃; under 5250.53 Torr; for 1h; Temperature; Pressure; Autoclave; |

| Conditions | Yield |
|---|---|
| With Amberlyst A 26; carbonate form In tetrahydrofuran for 1h; Heating; | 95% |
| Multi-step reaction with 2 steps 1: NaBH4 / ethanol / 2 h / 15 - 20 °C 2: H2O2 / 2 h / 15 - 20 °C / var. oxidants and time. View Scheme | |
| Multi-step reaction with 4 steps 1.1: potassium carbonate / N,N-dimethyl-formamide / 80 °C / Inert atmosphere 2.1: potassium peroxomonosulfate / methanol; water / 12 h / 0 - 20 °C / Inert atmosphere 3.1: n-butyllithium / tetrahydrofuran; hexane / 0.5 h / -78 - 0 °C / Inert atmosphere 3.2: 0 - 20 °C / Inert atmosphere 4.1: water; dihydrogen peroxide; sodium hydroxide / tetrahydrofuran; hexane / 0.5 h / 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 4 steps 1.1: potassium carbonate / N,N-dimethyl-formamide / 80 °C / Inert atmosphere 2.1: potassium peroxomonosulfate / methanol; water / 12 h / 0 - 20 °C / Inert atmosphere 3.1: n-butyllithium / tetrahydrofuran; hexane / 0.5 h / -78 - 0 °C / Inert atmosphere 3.2: 0 - 20 °C / Inert atmosphere 4.1: water; dihydrogen peroxide; sodium hydroxide / tetrahydrofuran; hexane / 0.5 h / 20 °C / Inert atmosphere View Scheme |
-
-
71821-61-9
3-methyl-1-(trimethylsilyloxy)but-2-ene

-
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With tris paraperiodate In benzene Heating; | 95% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 100℃; for 2.5h; | 95% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate; sodium dodecylbenzenesulfonate In cyclohexane; water at 45℃; pH=8; | 92.9% |
| Multi-step reaction with 2 steps 2: aqueous KOH-solution View Scheme |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In tetrahydrofuran at 0 - 20℃; | 91% |
| With lithium aluminium tetrahydride In diethyl ether at 0℃; for 3h; Inert atmosphere; | 72% |
| With diisobutylaluminium hydride In toluene at -78 - 20℃; |
-
-
54105-78-1
1-[(tetrahydro-2H-pyran-2-yl)oxy]-3-methyl-2-butene

-
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With cerium(III) chloride In methanol at 20℃; for 1h; detetrahydropyranylation; | 90% |
-
-
68480-28-4
formiate de methyl-3 butene-2 ol-1

-
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With potassium carbonate In ethanol for 0.333333h; | 84% |
| With potassium hydroxide |

| Conditions | Yield |
|---|---|
| With dmap; formic acid; hexarhodium hexadecacarbonyl; carbon monoxide In tetrahydrofuran at 30℃; under 3800 Torr; for 20h; | A 4 % Chromat. B 81% |
| With dmap; formic acid; hexarhodium hexadecacarbonyl; carbon monoxide In tetrahydrofuran at 30℃; under 3800 Torr; Product distribution; base effect; | |
| With Pt3Fe; hydrogen In ethanol at 70℃; under 750.075 Torr; for 5h; Catalytic behavior; Time; | |
| With Pt0615Fe0385; hydrogen In ethanol at 100℃; under 1800.18 Torr; |

| Conditions | Yield |
|---|---|
| With methanesulfonic acid In tetrahydrofuran; water at 20℃; for 12h; | 74% |
| With sulfuric acid | |
| With dichloro-acetic acid und anschliessende Verseifung; |
-
-
87791-06-8
morpholide of 1-phenylthio-4-hydroxy-2-methylbut-2Z-ene-1-sulfonic acid

-
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With lithium In ammonia at -70℃; for 0.0833333h; | 74% |

-
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With aluminium hydride In tetrahydrofuran at 0℃; for 2h; | 70% |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether for 1.5h; ice bath; | 60% |
| With lithium aluminium tetrahydride In diethyl ether for 5h; | 54% |
| With lithium aluminium tetrahydride; diethyl ether | |
| With lithium aluminium tetrahydride In diethyl ether |
-
-
763-32-6
2-methyl-1-buten-4-ol

-
A
-
107-86-8
3,3-dimethyl acrylaldehyde

-
B
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With 5%-palladium/activated carbon; oxygen at 80℃; under 3750.38 Torr; for 5h; Reagent/catalyst; Time; Concentration; | A 8.78% B 53.06% |
-
-
1438-14-8
2-isopropyloxirane

-
A
-
563-80-4
3-methyl-butan-2-one

-
B
-
763-32-6
2-methyl-1-buten-4-ol

-
C
-
35158-25-9
2-isopropyl-5-methyl-hex-2-enal

-
D
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With magnesium bromide In N,N-dimethyl-formamide at 130℃; for 0.666667h; Further byproducts given; | A 52.2% B 23.2% C 8.7% D 13.7% |

-
-
16722-09-1
trimethylsilyloxirane

-
-
4238-09-9
phenyl isopropyl sulfone

-
A
-
133827-98-2
3-Benzenesulfonyl-3-methyl-1-trimethylsilanyl-butan-1-ol

-
B
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran at -20 - 20℃; for 16h; | A 45% B 31% |


| Conditions | Yield |
|---|---|
| With sulfuric acid; hydrogen bromide; acetic acid das Acetat entsteht; |
-
-
79-43-6
dichloro-acetic acid

-
-
115-18-4
2-methyl-3-buten-2-ol

-
A
-
106-24-1
Geraniol

-
B
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| bei folgendem Verseifen; | |
| und Verseifen; |


| Conditions | Yield |
|---|---|
| With diethyl ether |
-
-
16435-50-0
1-chloro-3-pentene

-
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With alkali |

| Conditions | Yield |
|---|---|
| With isopropyl alcohol at 110℃; | |
| at 110℃; |
-
-
2190-48-9
3-chloro-3-methyl-1-butene

-
-
141-53-7
sodium formate

-
A
-
115-18-4
2-methyl-3-buten-2-ol

-
B
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With formic acid Erwaermen des Reaktionsprodukts mit wss.Kalilauge; |


| Conditions | Yield |
|---|---|
| With tetrahydrofuran; magnesium; mercury dichloride Erhitzen des Reaktionsprodukts mit wss.Kalilauge; |
-
-
503-60-6
3,3-dimethyl-allyl chloride

-
A
-
115-18-4
2-methyl-3-buten-2-ol

-
B
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate at 50℃; |

| Conditions | Yield |
|---|---|
| With sulfuric acid; copper(II) sulfate; acetic acid at 20℃; das Acetat entsteht; Reagens 4: Hydrochinon; | |
| With toluene-4-sulfonic acid; acetic acid das Acetat entsteht; | |
| ueber das Dichloracetat; | |
| With sulfuric acid; copper(II) sulfate; acetic acid at 20℃; das Acetat entsteht; Reagens 4: Hydrochinon; | |
| ueber das Dichloracetat; |
-
-
766-15-4
4,4-dimethyl-1,3-dioxane

-
A
-
16302-35-5
4-methyl-3,6-dihydro-2H-pyran

-
B
-
763-32-6
2-methyl-1-buten-4-ol

-
C
-
2568-33-4
3-methyl-butane-1,3-diol

-
D
-
7525-64-6
4-hydroxy-4-methyltetrahydropyran

-
E
-
115-18-4
2-methyl-3-buten-2-ol

-
F
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With sulfuric acid at 120℃; for 3h; Product distribution; various catalyst, various temperature, various time, extent of conversion, solvent cyclohexane; |


| Conditions | Yield |
|---|---|
| With (1,5-cyclooctadiene)(pyridine)(tricyclohexylphosphine)iridium(I) tetrakis[3,5-bis(trifluoromethyl)phenyl]borate; hydrogen In tetrahydrofuran at 23℃; for 0.5h; | 100% |
| With [Os(η(5)-C5,κ-N-Cp(N))(CH3CN)2]PF6 In tetrahydrofuran-d8 at 60℃; for 1.33333h; Inert atmosphere; | 93% |
| With {(η6-p-cymene)RuCl2}{[3-(3,5-diaza-1-azonia-7-phosphatricyclo[3.3.1.13,7]decan-1-yl)propyl]silanetriyltrioxy} supported on silica-coated ferrite nanoparticles In water at 150℃; under 5171.62 - 6205.94 Torr; for 14h; Inert atmosphere; Microwave irradiation; | 87% |
-
-
103-71-9
phenyl isocyanate

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
105902-61-2
3-methylbut-2-en-1-yl phenylcarbamate

| Conditions | Yield |
|---|---|
| In acetonitrile at 70℃; for 23h; Inert atmosphere; | 100% |
| With triethylamine In dichloromethane at 20℃; Inert atmosphere; | 88% |
| With triethylamine In dichloromethane at 20℃; | 88% |
-
-
110-87-2
3,4-dihydro-2H-pyran

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
54105-78-1
1-[(tetrahydro-2H-pyran-2-yl)oxy]-3-methyl-2-butene

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane at 20℃; for 4h; | 100% |
| With pyridinium p-toluenesulfonate In tetrahydrofuran | 100% |
| With Aluminum(III) chloride hexahydrate at 30℃; for 0.5h; | 98% |
-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
18511-56-3
(3,3-dimethyloxiranyl)methanol

| Conditions | Yield |
|---|---|
| With dihydrogen peroxide; {[CF3(CF2)7(CH2)3]3N(+)}12[WZn3(H2O)2(ZnW9O34)2] In ethyl acetate at 22℃; for 8h; | 100% |
| With 3,3-dimethyldioxirane In acetone at 23℃; Kinetics; Further Variations:; Solvents; | 100% |
| With tetrabutylphosphonium peroxotantalate; dihydrogen peroxide In water at 0℃; for 1.5h; Catalytic behavior; Reagent/catalyst; Schlenk technique; chemoselective reaction; | 100% |

| Conditions | Yield |
|---|---|
| indium(III) chloride In acetonitrile at 20℃; for 0.5h; | 100% |
| With zirconium(IV) chloride In dichloromethane at 20℃; for 1h; | 100% |
| With pyridine at 20℃; for 2.5h; | 99% |
-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
188263-82-3
1-((tert-butyldiphenylsilyl)oxy)-3-methyl-2-butene

| Conditions | Yield |
|---|---|
| With 1H-imidazole In dichloromethane; N,N-dimethyl-formamide at 0 - 20℃; | 100% |
| With 1H-imidazole In dichloromethane at 0 - 20℃; for 0.5h; | 98% |
| With dmap; triethylamine In dichloromethane for 2h; Ambient temperature; | 82% |
-
-
5707-04-0
Phenylselenyl chloride

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
99018-46-9
3-methyl-2-(phenylselenyl)-1,3-butanediol

| Conditions | Yield |
|---|---|
| With water In acetonitrile at 20℃; for 24h; | 100% |
| In water; acetonitrile for 2h; Ambient temperature; | 86% |
-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
114701-65-4
tert-butyldimethyl((3-methylbut-2-en-1-yl)oxy)silane

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; | 100% |
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 6h; | 94% |
| With 1H-imidazole In dichloromethane at 0 - 20℃; | 93% |
-
-
76-83-5
trityl chloride

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
141561-63-9
(((3-methylbut-2-en-1-yl)oxy)methanetrityl)tribenzene

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 0 - 23℃; | 100% |
| With dmap; triethylamine In dichloromethane at 0 - 23℃; Inert atmosphere; | 100% |
| With dmap; 4 A molecular sieve; triethylamine In dichloromethane at 20℃; for 12h; | 65% |

| Conditions | Yield |
|---|---|
| at 20℃; for 0.5h; Inert atmosphere; neat (no solvent); | 100% |

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 20℃; Mitsunobu reaction; Cooling with ice; | 100% |
-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
108-12-3
isopentanoyl chloride

-
-
89026-29-9
3-methyl-2-buten-1-yl 3-methylbutanoate

| Conditions | Yield |
|---|---|
| With pyridine; dmap In dichloromethane at 0 - 20℃; for 2.5h; | 100% |
-
-
1445-45-0
Trimethyl orthoacetate

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
7796-72-7
ethyl 3,3-dimethylpent-4-enoate

| Conditions | Yield |
|---|---|
| With phenol at 130 - 160℃; for 16h; Johnson-Claisen Rearrangement; Inert atmosphere; | 100% |
-
-
1003872-51-2
2,6-dichloro-N-cyclohexyl-pyridine-3-carboxamide

-
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: 3-methyl-2-buten-1-ol With sodium hydride In N,N-dimethyl-formamide; mineral oil for 0.75h; Inert atmosphere; Stage #2: 2,6-dichloro-N-cyclohexyl-pyridine-3-carboxamide In N,N-dimethyl-formamide; mineral oil at 0 - 20℃; for 4h; Inert atmosphere; | 100% |

-
-
41725-90-0
methyl 2-methylbut-2-enoate

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
765300-33-2
3-methyl-2-butenyl angelate

| Conditions | Yield |
|---|---|
| In toluene | 99.8% |

-
-
4530-20-5
BOC-glycine

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
82706-45-4
tert-butoxycarbonylamino-acetic acid 3-methyl-but-2-enyl ester

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at -5 - 25℃; for 8h; | 99.15% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 0 - 20℃; | 91% |
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; | |
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 2h; Solvent; Large scale; | 129 kg |

| Conditions | Yield |
|---|---|
| With bis(acetylacetonato)dioxidomolybdenum(VI); dimethyl sulfoxide at 100℃; for 7h; | 99% |
| With oxygen; Pd561phen60(OAc)180 In benzene at 60℃; for 24h; | 99% |
| With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; oxygen In acetonitrile at 75℃; under 760.051 Torr; for 11h; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: benzoyl chloride; 3-methyl-2-buten-1-ol In dichloromethane at 20℃; Stage #2: With poly{trans-bicyclo[2.2.1]hept-5-ene-2,3-di(chlorocarbonyl)} In dichloromethane Heating; | 99% |
| With dmap; triethylamine In dichloromethane at 0 - 20℃; | 91% |
| With pyridine at 20℃; for 0.5h; | 89% |

| Conditions | Yield |
|---|---|
| With N-iodo-succinimide In dichloromethane at -78 - 0℃; | 99% |
| Stage #1: 4-Methoxystyrene; 3-methyl-2-buten-1-ol In dichloromethane at 20℃; for 0.0833333h; Stage #2: With N-iodo-succinimide In dichloromethane at -78 - 0℃; Further stages.; | 99% |

| Conditions | Yield |
|---|---|
| With 1H-imidazole In N,N-dimethyl-formamide at 20℃; for 22h; | 99% |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| Stage #1: 3-methyl-2-buten-1-ol With n-butyllithium In tetrahydrofuran; hexane at 0℃; for 1.5h; Stage #2: di-tert-butyl dicarbonate In tetrahydrofuran; hexane at 0 - 24℃; for 20h; | 99% |
-
-
79-30-1
isobutyryl chloride

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
76649-23-5
3-methyl-2-butenyl isobutanoate

| Conditions | Yield |
|---|---|
| With pyridine; dmap In dichloromethane at 0 - 20℃; for 2.5h; | 99% |
| With dmap; triethylamine In dichloromethane at 20℃; for 18h; Inert atmosphere; |

| Conditions | Yield |
|---|---|
| With carbon dioxide at 46.02℃; under 66081.6 Torr; for 3.5h; Reagent/catalyst; High pressure; Supercritical conditions; Green chemistry; | 99% |
-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
62875-08-5
(R,S)-3-methyl-1,2,3-butanetriol

| Conditions | Yield |
|---|---|
| Stage #1: 3-methyl-2-buten-1-ol With dihydrogen peroxide; bis(2,4-pentanedionato)dioxomolybdenum(VI) In acetonitrile at 30℃; Stage #2: With diphenyl sulfide regioselective reaction; | 99% |
| Multi-step reaction with 2 steps 1: 3-chloro-benzenecarboperoxoic acid / acetonitrile / 168 h / 20 °C 2: water; perchloric acid / 0.17 h View Scheme |

| Conditions | Yield |
|---|---|
| With pseudomonas fuorescens lipase immobilized on multiwall carbon nano-tubes at 50℃; for 4h; Green chemistry; | 99% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; | 99% |
| In acetonitrile at 70℃; for 23h; Inert atmosphere; | 78% |

| Conditions | Yield |
|---|---|
| With pyridine at 0 - 24℃; for 24.5h; Inert atmosphere; | 99% |
| With pyridine at 0 - 24℃; for 24.5h; | 99% |
-
-
28131-17-1
2-(4-bromophenyl)acrylic acid

-
-
556-82-1
3-methyl-2-buten-1-ol

| Conditions | Yield |
|---|---|
| With 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane at 20℃; for 3h; Inert atmosphere; | 99% |

-
-
556-82-1
3-methyl-2-buten-1-ol

-
-
1334485-04-9
3-methylbut-2-enyl 4-(2,2-dimethyl-4-oxo-4H-1,3-dioxin-6-yl)-3-oxobutanoate

| Conditions | Yield |
|---|---|
| In toluene at 55℃; Inert atmosphere; Schlenk technique; | 99% |
| In toluene at 55℃; for 4h; regioselective reaction; | 4.33 g |
3-Methyl-2-buten-1-ol Chemical Properties
Molecular Structure of 3-Methyl-2-buten-1-ol (CAS No.556-82-1):
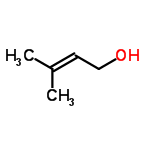
Molecular Formula: C5H10O
Molecular Weight: 86.13
IUPAC Name: 3-Methylbut-2-en-1-ol
CAS No: 556-82-1
EINECS: 209-141-4
H bond acceptors: 1
H bond donors: 1
Freely Rotating Bonds: 2
Polar Surface Area: 9.23 Å2
Index of Refraction: 1.435
Molar Refractivity: 26.64 cm3
Molar Volume: 101.9 cm3
Surface Tension: 26.4 dyne/cm
Density: 0.844 g/cm3
Flash Point: 43.3 °C
Enthalpy of Vaporization: 44.12 kJ/mol
Boiling Point: 141.5 °C at 760 mmHg
Vapour Pressure: 2.38 mmHg at 25°C
Refractive Index: 1.442-1.444
Water Solubility: 170 g/L (20 ºC)
Storage Temp.: Flammables area
Classification Code: Skin/Eye Irritant
3-Methyl-2-buten-1-ol Uses
3-Methyl-2-buten-1-ol (CAS No.556-82-1) is used occasionally in perfumery.It occurs naturally in citrus fruits, cranberry, bilberry, currants, grapes, raspberry, blackberry, tomato, white bread, hop oil, coffee, arctic bramble, cloudberry and passion fruit.
3-Methyl-2-buten-1-ol Production
3-Methyl-2-buten-1-ol (CAS No.556-82-1) is produced industrially by the reaction of formaldehyde with isobutene, followed by the isomerization of the resulting isoprenol (3-methyl-3-buten-1-ol).
.jpg)
3-Methyl-2-buten-1-ol Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rabbit | LD50 | skin | 3900mg/kg (3900mg/kg) | Food and Cosmetics Toxicology. Vol. 17, Pg. 895, 1979. | |
| rat | LD50 | oral | 810mg/kg (810mg/kg) | Food and Cosmetics Toxicology. Vol. 17, Pg. 895, 1979. |
3-Methyl-2-buten-1-ol Consensus Reports
3-Methyl-2-buten-1-ol (CAS No.556-82-1) is reported in EPA TSCA Inventory.
3-Methyl-2-buten-1-ol Safety Profile
Safety Information of 3-Methyl-2-buten-1-ol (CAS No.556-82-1):
Hazard Codes:  Xn
Xn
Risk Statements: 10-22-36/37/38-38-21/22
R10:Flammable.
R22:Harmful if swallowed.
R36/37/38:Irritating to eyes, respiratory system and skin.
R38:Irritating to skin.
R21/22:Harmful in contact with skin and if swallowed.
Safety Statements: 26-36-37-23-16
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36:Wear suitable protective clothing.
S37:Wear suitable gloves.
S23:Do not breathe vapour.
S16:Keep away from sources of ignition.
RIDADR: UN 1987 3/PG 3
WGK Germany: 1
RTECS: EM9472500
HazardClass: 3
PackingGroup: III
HS Code: 29052990
3-Methyl-2-buten-1-ol Specification
3-Methyl-2-buten-1-ol (CAS No.556-82-1), it also can be called 3,3-Dimethylallyl alcohol ; Prenyl alcohol ; 2-Buten-1-ol, 3-methyl- ; 2-Methyl-2-buten-4-ol ; 3-Methylbut-2-en-1-ol . It is clear colourless to very slightly yellow liquid. 3-Methyl-2-buten-1-ol (CAS No.556-82-1) is a natural alcohol. It is one of the most simple terpenes. It is a clear colorless oil that is reasonably soluble in water and miscible with most common organic solvents.
Related Products
- 3-Methyl-2-buten-1-ol
- 55682-48-9
- 55682-83-2
- 55682-91-2
- 5568-33-2
- 55684-37-2
- 55-68-5
- 55685-75-1
- 55686-22-1
- 55687-02-0
- 55687-33-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View