-
Name
4,5-Dimethoxy-2-nitrobenzoic acid
- EINECS 225-657-2
- CAS No. 4998-07-6
- Article Data33
- CAS DataBase
- Density 1.403 g/cm3
- Solubility Insoluble in water. Soluble in DMSO and methanol. Solubility in hot 1mol/L NaOH (almost transparency).
- Melting Point 195-197 °C(lit.)
- Formula C9H9NO6
- Boiling Point 409.5 °C at 760 mmHg
- Molecular Weight 227.174
- Flash Point 201.4 °C
- Transport Information
- Appearance Yellow crystalline powder
- Safety 26-37/39
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Veratricacid, 6-nitro- (6CI,7CI,8CI);2-Nitro-4,5-dimethoxybenzoic acid;3,4-Dimethoxy-6-nitrobenzoic acid;6-Nitroveratric acid;
- PSA 101.58000
- LogP 1.83340
Synthetic route

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 1-(6-amino-2-bromo-3,4-dimethoxyphenyl)ethan-1-one With 1 ,5-pentanediol for 0.666667h; Stage #2: With nickel(III) oxide; potassium carbonate at 70℃; for 0.666667h; pH=9; Temperature; pH-value; | 96% |
-
-
20357-25-9
2-nitroveratraldehyde

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With sodium chlorite; dihydrogen peroxide; acetic acid In methanol; water at 50℃; Solvent; Temperature; Concentration; Reagent/catalyst; | 93% |
| Stage #1: 6-nitroveratraldehyde With potassium dihydrogenphosphate In water; acetonitrile Stage #2: With sodium chlorite In water; acetonitrile at 25 - 35℃; | 88% |
| With potassium permanganate; sodium hydroxide for 7h; | 83% |

| Conditions | Yield |
|---|---|
| With nitric acid; acetic acid at 90℃; under 11251.1 Torr; for 0.0833333h; Temperature; Pressure; Flow reactor; | 87.6% |
| With nitric acid In water at 60℃; for 6h; Cooling with ice; | 77% |
| With nitric acid at 60℃; for 6.5h; Cooling with ice; | 77% |

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| Oxydation; |
-
-
93-07-2
Veratric acid

-
A
-
709-09-1
3,4-dimethoxynitrobenzene

-
B
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With nitric acid |
-
-
861571-93-9
2-(4,5-dimethoxy-2-nitro-phenyl)-3,5,7-trimethoxy-chromen-4-one

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid
-
-
7697-37-2
nitric acid

-
-
93-07-2
Veratric acid

-
A
-
709-09-1
3,4-dimethoxynitrobenzene

-
B
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid


-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With ethanol |

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With potassium permanganate |

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With potassium permanganate; water |
-
-
16620-92-1
14-nitro-N-methylpapaverinium iodide

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| at 100℃; |
-
-
861363-56-6
(4,5-dimethoxy-2-methyl-3-nitro-phenyl)-hydrazine

-
-
7758-99-8
copper(II) sulfate

-
-
64-19-7
acetic acid

-
A
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
B
-
79025-28-8
3,4-Dimethoxy-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| Oxydation des entstandenen Oels mit Permanganat; |

-
-
26791-93-5
methyl 4,5-dimethoxy-2-nitrobenzoate

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In tetrahydrofuran | |
| With sodium hydroxide In methanol | |
| Stage #1: methyl 4,5-dimethoxy-2-nitrobenzoate With potassium hydroxide; water at 0 - 70℃; Heating / reflux; Stage #2: With acetic acid In water at 20℃; pH=6.6; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: K2CO3 / acetone / Heating 2: HNO3; acetic acid / 50 - 60 °C 3: NaOH / tetrahydrofuran View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: H2SO4 / Heating 2: K2CO3 / acetone / Heating 3: HNO3; acetic acid / 50 - 60 °C 4: NaOH / tetrahydrofuran View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: HNO3; acetic acid / 50 - 60 °C 2: NaOH / tetrahydrofuran View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 3: KMnO4 View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 2: KMnO4 View Scheme | |
| Multi-step reaction with 2 steps 1: nitric acid / 5 - 10 °C 2: KOH-solution; KMnO4 View Scheme | |
| Multi-step reaction with 2 steps 1: nitric acid / 1 h / 20 - 25 °C 2: sodium hydroxide; potassium permanganate / 7 h View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: water; KMnO4 / 80 - 90 °C 2: nitric acid View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: acetic anhydride; nitric acid / 6 h / 0 - 20 °C 2: potassium permanganate; copper(ll) sulfate pentahydrate; potassium hydroxide / benzene / 10 h / Reflux View Scheme |

| Conditions | Yield |
|---|---|
| With potassium permanganate; copper(ll) sulfate pentahydrate; potassium hydroxide In benzene for 10h; Reflux; | 13 g |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
29568-78-3
4,5-dimethoxy-2-nitrobenzoyl chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride In toluene for 3h; Heating; | 100% |
| With phosphorus pentachloride | |
| With thionyl chloride; chloroform |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
115279-57-7
2-(4-aminophenyl)-2-methylpropionitrile

-
-
1067193-04-7
N-[4-(cyano-dimethyl-methyl)-phenyl]-4,5-dimethoxy-2-nitro-benzamide

| Conditions | Yield |
|---|---|
| Stage #1: 4,5-dimethoxy-2-nitro-benzoic acid With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane at 20℃; for 1h; Stage #2: 2-(4-aminophenyl)-2-methylpropionitrile With triethylamine In dichloromethane at 20℃; | 100% |

| Conditions | Yield |
|---|---|
| With silver carbonate In dimethyl sulfoxide at 120℃; for 16h; | 100% |
| With copper(l) iodide; triethylamine In dimethyl sulfoxide at 140℃; under 760.051 Torr; for 20h; Catalytic behavior; Reagent/catalyst; Solvent; Temperature; Inert atmosphere; Schlenk technique; | 95% |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
1444018-10-3
potassium 2-nitro-4,5-dimethoxybenzoate

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In ethanol at 20℃; for 3h; Inert atmosphere; | 100% |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
31839-20-0
2-nitro-4-methoxy-5-hydroxybenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4,5-dimethoxy-2-nitro-benzoic acid With sodium hydroxide; water at 100℃; for 3h; Stage #2: With hydrogenchloride In water at 0℃; pH=< 2; | 99% |
| With water; sodium hydroxide at 100℃; for 3h; Inert atmosphere; | 99% |
| With water; sodium hydroxide at 100℃; for 6h; | 97.6% |

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In methanol at 20℃; under 7600.51 Torr; for 6h; | 97% |
| With palladium 10% on activated carbon; hydrogen In methanol at 20℃; for 24h; | 92% |
| With hydrazine hydrate In ethanol at 60℃; for 5h; Sealed tube; Green chemistry; chemoselective reaction; | 87% |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

| Conditions | Yield |
|---|---|
| With dmap; copper diacetate In acetonitrile at 100℃; for 6h; Schlenk technique; Sealed tube; Inert atmosphere; | 94% |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
349411-81-0
N,N-dimethyl-4,5-dimethoxy-2-nitrobenzamide

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 120℃; for 1h; Sealed tube; | 93% |
| With trichlorophosphate at 120℃; for 1h; | 93% |

| Conditions | Yield |
|---|---|
| Stage #1: 4,5-dimethoxy-2-nitro-benzoic acid With thionyl chloride for 0.833333h; Heating; Stage #2: With ammonium hydroxide | 88.5% |
| Stage #1: 4,5-dimethoxy-2-nitro-benzoic acid With triethylamine; Diphenylphosphinic chloride In chloroform Stage #2: With ammonia | |
| Multi-step reaction with 2 steps 1: SOCl2 2: NH4OH View Scheme |

| Conditions | Yield |
|---|---|
| With triethylamine In acetone for 1.5h; | 86% |

| Conditions | Yield |
|---|---|
| In methanol; water at 20℃; | 86% |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
54431-30-0
1-(4,5-dimethoxy-2-nitrobenzoyl)-4-hydroxypyrrolidine-2-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: 4,5-dimethoxy-2-nitro-benzoic acid With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane at 20℃; for 16h; Stage #2: (2S,4R)-4-hydroxy-2-proline methyl ester 4-toluenesulfonate With triethylamine In dichloromethane at -20 - 20℃; for 16h; | 85% |
| With oxalyl dichloride; TEA In dichloromethane; N,N-dimethyl-formamide at 20℃; | 57% |

| Conditions | Yield |
|---|---|
| With silver carbonate; palladium(II) trifluoroacetate In dimethyl sulfoxide; N,N-dimethyl-formamide at 120℃; for 3h; Heck olefination; | 85% |

| Conditions | Yield |
|---|---|
| With triethanolamine; N,N-dimethyl-formamide In 4-(dicyanomethylene)-2-methyl-6-(p-dimethylaminostyryl)-4H-pyran | 85% |

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
66003-76-7
Diphenyliodonium triflate

-
-
92200-31-2
4,5-dimethoxy-2-nitrobiphenyl

| Conditions | Yield |
|---|---|
| With silver carbonate; bis[2-(diphenylphosphino)phenyl] ether; palladium dichloride In dimethyl sulfoxide at 150℃; for 1h; | 85% |

| Conditions | Yield |
|---|---|
| With oxygen; silver carbonate; potassium hydroxide; copper dichloride In dimethyl sulfoxide; N,N-dimethyl-formamide at 130 - 140℃; | 85% |
| With copper(l) iodide; oxygen; copper(l) chloride In dimethyl sulfoxide at 160℃; under 760.051 Torr; for 30h; Schlenk technique; Sealed tube; | 65% |
| With 2.9-dimethyl-1,10-phenanthroline; oxygen; copper (I) acetate; silver sulfate; sodium chloride In dimethyl sulfoxide at 160℃; for 24h; Schlenk technique; | 52% |
| With 2.9-dimethyl-1,10-phenanthroline; oxygen; copper diacetate; silver sulfate; sodium chloride In dimethyl sulfoxide at 160℃; under 760.051 Torr; for 20h; Schlenk technique; | 52% |
-
-
591-50-4
iodobenzene

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
92200-31-2
4,5-dimethoxy-2-nitrobiphenyl

| Conditions | Yield |
|---|---|
| With 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; N,N-dimethyl-aniline; silver carbonate; palladium dichloride at 150℃; for 2h; | 82% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; Steglich Esterification; | 82% |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
513-42-8
3-hydroxy-2-methyl-1-propene

-
-
1286732-04-4
2-methylallyl 4,5-dimethoxy-2-nitrobenzoate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; Steglich Esterification; | 82% |
-
-
610-94-6
2-bromobenzoic acid methyl ester

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
108012-00-6
2,3-dimethoxydibenzopyran-6-one

| Conditions | Yield |
|---|---|
| With palladium(II) acetylacetonate; potassium carbonate; bis[2-(diphenylphosphino)phenyl] ether; copper(l) chloride In 1-methyl-pyrrolidin-2-one at 165℃; for 16h; Inert atmosphere; Molecular sieve; chemoselective reaction; | 81% |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
870-08-6
dioctyltin(IV) oxide

-
-
1229818-23-8
n-Oct2Sn(4,5-dimethoxy-2-nitrobenzoate)2

| Conditions | Yield |
|---|---|
| In toluene byproducts: H2O; Stoich. amounts of HL and n-Oct2SO were mixed in toluene, reflux for 6-7h, H2O was removed using a Dean-Stark apparatus; Toluene was evapd. under reduced pressure, mass was crystd. from CHCl3:acetone (1:1); elem. anal.; | 80% |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
1461-22-9
tributyltin chloride

-
-
1229818-10-3
n-Bu3Sn(4,5-dimethoxy-2-nitrobenzoate)

| Conditions | Yield |
|---|---|
| With Et3N In chloroform byproducts: Et3NHCl; Et3N (1 mmol) was added to dissolved in solvent HL (1 mmol), mixt. was refluxed 30 min, 1 mmol n-Bu3SnCl was added, solution was refluxed for 4-7 h, mixt. was kept at 25°C overnight; Et3NHCl was filtered off, solvent was evapd. under redused pressure, mass was crystd. from CHCL3:n-hexane (1:1); elem. anal; | 80% |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
639-58-7
triphenyltin chloride

-
-
1229818-12-5
Ph3Sn(4,5-dimethoxy-2-nitrobenzoate)

| Conditions | Yield |
|---|---|
| With Et3N In chloroform byproducts: Et3NHCl; Et3N (1 mmol) was added to dissolved in solvent HL (1 mmol), mixt. was refluxed 30 min, 1 mmol n-Bu3SnCl was added, solution was refluxed for 4-7 h, mixt. was kept at 25°C overnight.; Et3NHCl was filtered off, solvent was evapd. under redused pressure, mass was crystd. from CHCl3:n-hexane (1:1); elem. anal; | 80% |
-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
22296-40-8
4,4',5,5'-tetramethoxy-2,2'-dinitro-1,1'-biphenyl

| Conditions | Yield |
|---|---|
| With triphenylphosphine; silver carbonate; palladium dichloride In dimethyl sulfoxide at 130℃; for 6h; Inert atmosphere; | 80% |
-
-
53484-50-7
(E)-p-methoxy-cinnamyl alcohol

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
1620682-50-9
4-methoxycinnamyl 4,5-dimethoxy-2-nitrobenzoate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; Steglich Esterification; | 80% |
-
-
864755-03-3
(2S,4R)-2-t-butyldimethylsilyloxymethyl-4-oxyacetylpyrrolidine

-
-
4998-07-6
4,5-dimethoxy-2-nitro-benzoic acid

-
-
864755-12-4
(2S)(4R)-N-(4,5-dimethoxy-2-nitrobenzoyl)-2-(tert-butyldimethylsilyloxymethyl)-4-oxyacetylpyrrolidine

| Conditions | Yield |
|---|---|
| Stage #1: 4,5-dimethoxy-2-nitro-benzoic acid With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In DMF (N,N-dimethyl-formamide); dichloromethane at 0 - 20℃; for 1h; Stage #2: (2S,4R)-2-t-butyldimethylsilyloxymethyl-4-oxyacetylpyrrolidine In dichloromethane at 20 - 55℃; for 18h; | 79.9% |

| Conditions | Yield |
|---|---|
| With triphenyl-arsane; silver carbonate; palladium dichloride In dimethyl sulfoxide at 150℃; for 6h; | 79% |
| With 2,2'-bis-(diphenylphosphino)-1,1'-binaphthyl; N,N-dimethyl-aniline; silver carbonate; palladium dichloride at 150℃; for 2h; | 72% |
4,5-Dimethoxy-2-nitrobenzoic acid Chemical Properties
Molecule structure of 4,5-Dimethoxy-2-nitrobenzoic acid (CAS NO.4998-07-6) :
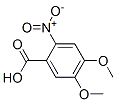
IUPAC Name: 4,5-dimethoxy-2-nitrobenzoic acid
Molecular Weight: 227.17086 g/mol
Molecular Formula: C9H9NO6
Density: 1.403 g/cm3
Melting Point: 195-197 °C(lit.)
Boiling Point: 409.5 °C at 760 mmHg
Flash Point: 201.4 °C
Appreance: yellow crystalline powder
Molar Volume: 161.8 cm3
Polarizability: 21.04*10-24 cm3
Surface Tension: 53.7 dyne/cm
Enthalpy of Vaporization: 69.77 kJ/mol
Vapour Pressure: 1.94E-07 mmHg at 25 °C
log P (octanol-water): 0.990
Water Solubility: 4.00E+04 mg/L
Atmospheric OH Rate Constant: 2.81E-12 cm3/molecule-sec
XLogP3-AA: 1.2
H-Bond Donor: 1
H-Bond Acceptor: 6
Rotatable Bond Count: 3
Exact Mass: 227.042987
MonoIsotopic Mass: 227.042987
Topological Polar Surface Area: 98.9
Heavy Atom Count: 16
Complexity: 276
Canonical SMILES: COC1=C(C=C(C(=C1)C(=O)O)[N+](=O)[O-])OC
InChI: InChI=1S/C9H9NO6/c1-15-7-3-5(9(11)12)6(10(13)14)4-8(7)16-2/h3-4H,1-2H3,(H,11,12)
InChIKey: WWCMFGBGMJAJRX-UHFFFAOYSA-N
EINECS: 225-657-2
Product Categories: Aromatic Carboxylic Acids, Amides, Anilides, Anhydrides & Salts; Organic acids; Aromatics; C9; Carbonyl Compounds; Carboxylic Acids
4,5-Dimethoxy-2-nitrobenzoic acid Uses
4,5-Dimethoxy-2-nitrobenzoic acid (CAS NO.4998-07-6) is used as pharmaceutical intermediate, mainly used to be prazosin hydrochloride intermediate.
4,5-Dimethoxy-2-nitrobenzoic acid Safety Profile
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38: Irritating to eyes, respiratory system and skin
Safety Statements: 26-37/39
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
S37/39: Wear suitable gloves and eye/face protection
WGK Germany: 3
4,5-Dimethoxy-2-nitrobenzoic acid Specification
4,5-Dimethoxy-2-nitrobenzoic acid (CAS NO.4998-07-6) is also called 6-Nitroveratriceacid ; Rarechem AL BO 1523 ; 2-Nitro-4,5-dimethoxybenzoic acid ; 6-Nitroveratric acid ; 3,4-Dimethoxy-6-nitrobenzoic acid ; 6-Nitroveratricacid,99% .
Related Products
- 4-06-00-02342 (Beilstein Handbook Reference)
- 4,10-Ace-1,2-benzanthracene
- 4,10-Dioxatricyclo[5.2.1.0(2,6)]decan-8-en-3-one
- 4-(1,1,2,2-Tetrafluoroethoxy)benzoicacid
- 4-(1,1,2,2-Tetrafluoroethoxy)chlorobenzene
- 4-(1,1,2,2-Tetrafluoroethoxy)nitrobenzene
- 4-(1,1,2,2-Tetrafluoroethoxy)toluene
- 4-(1,1-Difluoropropan-2-yl)benzene-1-sulfonyl chloride
- 4-(1,1-Dioxothiazolidin-2-yl)benzoate
- 4′-(1,2,3,4-TETRAHYDRO-4-(4-HYDROXY-2-OXO-2H-1-BENZOPYRAN-3-YL)-2-NAPHTHALENYL)(1,1′-BIPHENYL)-4-CARBONITRILE, cis-
- 499-80-9
- 499-81-0
- 499-83-2
- 4998-38-3
- 4998-57-6
- 499-86-5
- 4998-76-9
- 499-90-1
- 4999-79-5
- 50-00-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View