-
Name
4-Dimethylaminopyridine
- EINECS 214-353-5
- CAS No. 1122-58-3
- Article Data109
- CAS DataBase
- Density 1.012 g/cm3
- Solubility miscible with water
- Melting Point 83-86 °C(lit.)
- Formula C7H10N2
- Boiling Point 194.9 °C at 760 mmHg
- Molecular Weight 122.17
- Flash Point 71.7 °C
- Transport Information UN 2811 6.1/PG 2
- Appearance white to yellow crystalline powder
- Safety 36/37/39-45-28A-26-28-36/37-53-27-22
- Risk Codes 25-34-24/25-36/37/38-27-36-24-20-61-40-23/24/25
-
Molecular Structure
-
Hazard Symbols
 T,
T, C,
C, T+
T+
- Synonyms Pyridine, 4-(dimethylamino)-;N,N-Dimethylpyridin-4-amine;4-Pyridinamine, N,N-dimethyl-;N,N-dimethyl-1H-pyridin-4-amine;4-(Dimethylamino)pyridine;p-Dimethylaminopyridine;4-Dimethylamino-pyridine(DMAP);4-Dimethylaminopyridine(DMAP);gamma-(Dimethylamino)pyridine;4-Pyridinamine,N,N-dimethyl-;DMAP (catalyst);4-Dimethylamino pyridine;4-(Dimethylamino) Pyridine;DMAP 4-Dimethylaminopyridine;4-Dimethylaminopy ridine;4-Dimethyl aminopyridine;4-Dimethylaminopyridine DMAP;4-Di(methylamino)pyridine;4-Dimethylamino-pyridine;DMAP (4-Dimethylaminopyridine);4-Dimethylaminopyridine (dmap);4-DMAP;4-Dimethyl amino pyridine;N4,N4-Dimethylpyridin-4-amine;4-(N,N-Dimethylamino)pyridine;
- PSA 16.13000
- LogP 1.14760
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: pyridine-4-carbonitrile With hydrogenchloride; acrylic acid at 90℃; for 1.5h; Inert atmosphere; Stage #2: dimethyl amine at 70℃; for 2.5h; Inert atmosphere; Stage #3: With sodium hydroxide at 70 - 90℃; for 3.5h; Temperature; Reagent/catalyst; Inert atmosphere; | 99.4% |
| Stage #1: pyridine-4-carbonitrile With hydrogenchloride; acrylic acid In water at 70℃; for 4h; Inert atmosphere; Stage #2: dimethyl amine In water at 50℃; for 3h; Temperature; Inert atmosphere; | 99% |
| Stage #1: pyridine-4-carbonitrile With hydrogenchloride; hydroquinone; acrylic acid In water at 90℃; for 6h; Stage #2: dimethyl amine In water for 3h; Temperature; Reflux; | 98.6% |

| Conditions | Yield |
|---|---|
| With bis(triphenyl)oxodiphosphonium trifluoromethanesulfonate salt; potassium iodide In ethanol at 20℃; for 1h; | 95% |
| With phenylboronic acid In 1,2-dichloro-ethane at 120℃; for 10h; | 90% |
| With 1,1,1,2,2,2-hexamethyldisilane; tetrabutyl ammonium fluoride In tetrahydrofuran at 24 - 30℃; for 8h; | 84% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; water In 1,4-dioxane at 70℃; under 6000480 Torr; for 20h; Substitution; | 93% |

| Conditions | Yield |
|---|---|
| With copper(l) iodide; 6,7-dihydro-5H-quinolin-8-one oxime; potassium hydroxide In water at 25℃; for 24h; Inert atmosphere; | 90% |

| Conditions | Yield |
|---|---|
| In methanol at 40 - 45℃; for 4h; Autoclave; | 89.7% |

| Conditions | Yield |
|---|---|
| With N,N,N,N,-tetramethylethylenediamine In toluene at -40 - 20℃; for 1.5h; Substitution; | 86% |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride; water In acetonitrile at 20℃; Irradiation; | 86% |
| With palladium 10% on activated carbon; potassium tert-butylate at 150℃; for 48h; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In neat (no solvent) at 100℃; for 24h; Sealed tube; | 82% |

| Conditions | Yield |
|---|---|
| With ammonia In water at 240℃; for 0.5h; | 78% |

| Conditions | Yield |
|---|---|
| With potassium carbonate at 80℃; for 10h; | 75% |
| for 34h; Reflux; neat (no solvent); | 69% |
| With potassium hydroxide In neat (no solvent) at 100℃; for 24h; Sealed tube; | 21 %Spectr. |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In neat (no solvent) at 100℃; for 24h; Sealed tube; | 73% |

| Conditions | Yield |
|---|---|
| Stage #1: C9H8N2O2; dimethyl amine In water at 45℃; for 0.25h; Stage #2: With sodium hydroxide In water at 70℃; for 1 - 2h; | 67.1% |

| Conditions | Yield |
|---|---|
| With rhodium(III) chloride hydrate; potassium tert-butylate at 130℃; for 48h; Sealed tube; High pressure; | 66% |
-
-
5421-92-1
1-(4-pyridyl)pyridinium chloride hydrochloride

-
-
68-12-2, 33513-42-7
N,N-dimethyl-formamide

-
-
1122-58-3
dmap

| Conditions | Yield |
|---|---|
| Stage #1: 1-(4-pyridyl)pyridinium chloride hydrochloride; N,N-dimethyl-formamide at 150 - 155℃; for 2 - 3h; Stage #2: With sodium hydroxide In water at 20℃; pH=10 - 12; | 65.05% |
| Stage #1: 1-(4-pyridyl)pyridinium chloride hydrochloride; N,N-dimethyl-formamide at 150 - 155℃; for 2 - 3h; Stage #2: With sodium hydroxide In water at 20℃; pH=10 - 12; | 65.05% |

| Conditions | Yield |
|---|---|
| In water | 51% |

-
B
-
5028-28-4
N4,N4-dimethylpyridine-3,4-diamine

-
C
-
114474-08-7
2-amino-5-bromo-4-dimethylaminopyridine

| Conditions | Yield |
|---|---|
| With potassium amide In diethyl ether; ammonia at -78 - -33℃; for 1.5h; | A 40% B 32% C 10% |

-
-
84539-35-5
(3-bromopyridin-4-yl)-dimethyl-amine

-
A
-
1122-58-3
dmap

-
B
-
5028-28-4
N4,N4-dimethylpyridine-3,4-diamine

| Conditions | Yield |
|---|---|
| With potassium amide In diethyl ether; ammonia at -78℃; for 0.0833333h; | A 4% B 31% |

| Conditions | Yield |
|---|---|
| In 1,4-dioxane at 100℃; for 12h; | 27% |

| Conditions | Yield |
|---|---|
| With aluminum oxide at 300 - 369℃; |


| Conditions | Yield |
|---|---|
| at 180℃; |

| Conditions | Yield |
|---|---|
| With phenol at 180 - 190℃; |
-
-
55277-36-6
4-(dimethylamino)pyridinium cation

-
-
114444-46-1
C14H13NO

-
A
-
1122-58-3
dmap

-
B
-
124225-44-1
1-methyl-4-(phenylacetyl)pyridinium cation

| Conditions | Yield |
|---|---|
| In water at 25℃; Equilibrium constant; |

-
-
130671-19-1
1-(2-Cyano-ethyl)-4-dimethylamino-pyridinium; bromide

-
A
-
1122-58-3
dmap

-
B
-
107-13-1
acrylonitrile

| Conditions | Yield |
|---|---|
| With potassium hydroxide; potassium chloride In water at 25℃; Rate constant; var. basic media; pH dependence of the velocity const.; |

| Conditions | Yield |
|---|---|
| With potassium chloride; hydroxide In water at 25℃; Equilibrium constant; |
-
-
135041-86-0
4-Dimethylamino-1-[2-(4-nitro-phenyl)-ethyl]-pyridinium; bromide

-
A
-
1122-58-3
dmap

-
B
-
100-13-0
4-nitrostyrene

| Conditions | Yield |
|---|---|
| With potassium hydroxide; potassium chloride at 25℃; Rate constant; |
-
-
96318-91-1
N-phenylbenzimidoyl-4-dimethylaminopyridinium chloride

-
A
-
93-98-1
N-phenyl benzoyl amide

-
B
-
1122-58-3
dmap

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium chloride In 1,4-dioxane; water at 30℃; | |
| With sodium hydroxide; sodium chloride In 1,4-dioxane; water at 25℃; Rate constant; |
-
-
101476-38-4
4-Dimethylamino-1-{[(Z)-phenylimino]-m-tolyl-methyl}-pyridinium; chloride

-
A
-
1122-58-3
dmap

-
B
-
23099-05-0
3-methyl-N-phenylbenzamide

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium chloride In 1,4-dioxane; water at 30℃; | |
| With sodium hydroxide; sodium chloride In 1,4-dioxane; water at 25℃; Rate constant; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium chloride In 1,4-dioxane; water at 30℃; | |
| With sodium hydroxide; sodium chloride In 1,4-dioxane; water at 25℃; Rate constant; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium chloride In 1,4-dioxane; water at 30℃; | |
| With sodium hydroxide; sodium chloride In 1,4-dioxane; water at 25℃; Rate constant; |
-
-
1122-58-3
dmap

-
-
709-49-9
1-iodo-2,4-dinitrobenzene

-
-
111055-10-8
4-Dimethylamino-1-(2,4-dinitro-phenyl)-pyridinium; iodide

| Conditions | Yield |
|---|---|
| In diethyl ether Ambient temperature; | 100% |
-
-
1122-58-3
dmap

-
-
88-88-0
2,4,6-trinitrochlorobenzene

-
-
111055-14-2
4-Dimethylamino-1-(2,4,6-trinitro-phenyl)-pyridinium; chloride

| Conditions | Yield |
|---|---|
| In diethyl ether Ambient temperature; | 100% |
-
-
1122-58-3
dmap

-
-
4975-09-1
2,4-bis(trifluoromethylsulfonyl)chlorobenzene

-
-
111055-13-1
1-(2,4-Bis-trifluoromethanesulfonyl-phenyl)-4-dimethylamino-pyridinium; chloride

| Conditions | Yield |
|---|---|
| In diethyl ether Ambient temperature; | 100% |
-
-
1122-58-3
dmap

-
-
970-88-7
2,4-dinitrophenyl benzenesulfonate

-
-
111055-12-0
Benzenesulfonate4-dimethylamino-1-(2,4-dinitro-phenyl)-pyridinium;

| Conditions | Yield |
|---|---|
| In diethyl ether Ambient temperature; | 100% |
-
-
1122-58-3
dmap

-
-
71153-92-9
N-Pentafluorophenyl(C-pentafluorophenyl)imidoyl chloride

| Conditions | Yield |
|---|---|
| In acetonitrile at 25℃; Rate constant; Mechanism; | 100% |

| Conditions | Yield |
|---|---|
| In benzene for 0.166667h; Ambient temperature; | 100% |
| In acetone for 0.5h; Heating; | 90% |
-
-
1122-58-3
dmap

-
-
70-34-8
2,4-Dinitrofluorobenzene

-
-
111055-09-5
4-Dimethylamino-1-(2,4-dinitro-phenyl)-pyridinium; fluoride

| Conditions | Yield |
|---|---|
| In diethyl ether Ambient temperature; | 100% |
-
-
1122-58-3
dmap

-
-
87228-28-2
N-(pentafluorophenyl)-benzimidoyl chloride

-
-
120703-73-3
4-Dimethylamino-1-{[(Z)-pentafluorophenylimino]-phenyl-methyl}-pyridinium; chloride

| Conditions | Yield |
|---|---|
| In acetonitrile at 25℃; Rate constant; Mechanism; presence of tetraethylammonium salts; | 100% |
-
-
1122-58-3
dmap

-
-
120703-72-2
2,3,4,5,6-Pentafluoro-N-phenyl-benzimidoyl chloride

-
-
120703-76-6
4-Dimethylamino-1-{pentafluorophenyl-[(Z)-phenylimino]-methyl}-pyridinium; chloride

| Conditions | Yield |
|---|---|
| In acetonitrile at 25℃; Rate constant; Mechanism; presence of tetraethylammonium chloride; | 100% |
-
-
1122-58-3
dmap

-
-
120703-70-0
4-Methyl-N-(2,3,4,5,6-pentafluoro-phenyl)-benzimidoyl chloride

-
-
120703-74-4
4-Dimethylamino-1-{[(Z)-pentafluorophenylimino]-p-tolyl-methyl}-pyridinium; chloride

| Conditions | Yield |
|---|---|
| In acetonitrile at 25℃; Rate constant; Mechanism; | 100% |
-
-
1122-58-3
dmap

-
-
120703-71-1
4-Nitro-N-(2,3,4,5,6-pentafluoro-phenyl)-benzimidoyl chloride

-
-
120703-75-5
4-Dimethylamino-1-{(4-nitro-phenyl)-[(Z)-pentafluorophenylimino]-methyl}-pyridinium; chloride

| Conditions | Yield |
|---|---|
| In acetonitrile at 25℃; Rate constant; Mechanism; | 100% |
-
-
1122-58-3
dmap

-
-
97-00-7
1-chloro-2,4-dinitro-benzene

-
-
110465-52-6
4-Dimethylamino-1-(2,4-dinitro-phenyl)-pyridinium; chloride

| Conditions | Yield |
|---|---|
| In diethyl ether Ambient temperature; | 100% |

| Conditions | Yield |
|---|---|
| In ethyl acetate Ambient temperature; | 100% |
| In ethyl acetate | 100% |


| Conditions | Yield |
|---|---|
| In 1,2-dichloro-benzene at 80℃; for 3h; | 100% |
| In 1,2-dichloro-benzene at 60℃; | 87% |
| In 1,2-dichloro-benzene at 80 - 90℃; |

| Conditions | Yield |
|---|---|
| In 1,2-dichloro-benzene at 120℃; for 3h; | 100% |
| In 1,2-dichloro-benzene Heating; | |
| Heating; |

| Conditions | Yield |
|---|---|
| In acetonitrile for 15h; Heating; | 100% |
| In acetone for 9h; Heating; | 50% |
| In ethyl acetate at 70℃; for 48h; |

| Conditions | Yield |
|---|---|
| In methanol for 24h; Heating; | 100% |
-
-
864950-69-6
methanesulfonic acid 3-(2-phenylethynyl-phenyl)-prop-2-ynyl ester

-
-
1122-58-3
dmap

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 24h; | 100% |

| Conditions | Yield |
|---|---|
| In water; dimethyl sulfoxide at 25℃; Kinetics; | 100% |

-
-
1122-58-3
dmap

-
-
27607-77-8
trimethylsilyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| In chlorobenzene for 24h; Heating; | 100% |

-
-
1122-58-3
dmap

-
-
27607-77-8
trimethylsilyl trifluoromethanesulfonate

| Conditions | Yield |
|---|---|
| In chlorobenzene for 48h; Heating; | 100% |

4-Dimethylaminopyridine Specification
The 4-Dimethylaminopyridine, with the CAS registry number 1122-58-3,is also known as N,N-Dimethylpyridin-4-amine; DMAP. It belongs to the product categories of Nucleotides;Amines;Aromatics. This chemical's molecular formula is C7H10N2 and molecular weight is 122.17.Its EINECS number is 214-353-5. What's more,Its systematic name is 4-(Dimethylamino)pyridine .It is a White solid which is stable,incompatible with acids,oxidizing agents. It is a highly efficient catalyst for acylation reactions.Its analogues are used in kinetic resolution experiments of mainly secondary alcohols and Evans auxilar type amides.
Physical properties about 4-Dimethylaminopyridine are:
(1)ACD/LogP: 1.34; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -1.15; (4)ACD/LogD (pH7.4): -0.63; (5)ACD/BCF (pH 5.5): 1.00; (6)ACD/BCF (pH 7.4): 1.00; (7)ACD/KOC (pH 5.5): 1.00; (8)ACD/KOC (pH 7.4): 1.37; (9)#H bond acceptors: 2; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 1; (12)Index of Refraction: 1.554; (13)Molar Refractivity: 38.658 cm3; (14)Molar Volume: 120.648 cm3;(15)Surface Tension: 40.0180015563965 dyne/cm; (16)Density: 1.013 g/cm3; (17)Flash Point: 71.687 °C; (18)Enthalpy of Vaporization: 43.112 kJ/mol; (19)Boiling Point: 194.922 °C at 760 mmHg; (20)Vapour Pressure: 0.430999994277954 mmHg at 25°C.
You can still convert the following datas into molecular structure:
(1)SMILES:n1ccc(N(C)C)cc1;
(2)Std. InChI:InChI=1S/C7H10N2/c1-9(2)7-3-5-8-6-4-7/h3-6H,1-2H3;
(3)Std. InChIKey:VHYFNPMBLIVWCW-UHFFFAOYSA-N.
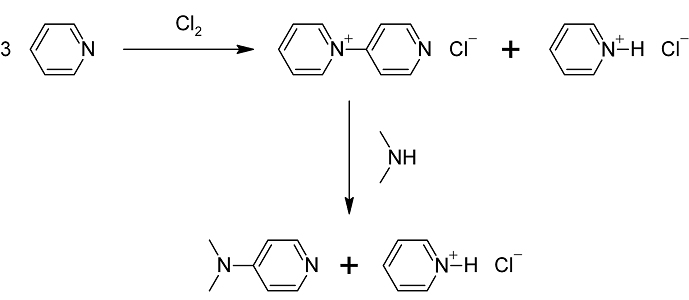
Preparation of 4-Dimethylaminopyridine :
4-Dimethylaminopyridine can be prepared in a two-step procedure from pyridine, which is first oxidized to 4-pyridylpyridinium cation. This cation then reacts with dimethylamine:
Safety Information of 4-Dimethylaminopyridine:
The 4-Dimethylaminopyridine is toxic by inhalation, in contact with skin and if swallowed and Irritating to eyes, respiratory system and skin. It is harmful by inhalation. And it may cause harm to the unborn child. There is limited evidence of a carcinogenic effect. In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) .When you use it wear suitable protective clothing, gloves and eye/face protection. Avoid exposure - obtain special instructions before use. After contact with skin, wash immediately with plenty of soap-suds. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. And take off immediately all contaminated clothing.
The toxicity data of 4-Dimethylaminopyridine as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intravenous | 56mg/kg (56mg/kg) | U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. Vol. NX#04228, | |
| mouse | LDLo | oral | 470mg/kg (470mg/kg) | Archives of Environmental Contamination and Toxicology. Vol. 14, Pg. 111, 1985. | |
| rabbit | LD50 | skin | 13mg/kg (13mg/kg) | National Technical Information Service. Vol. OTS0536502, | |
| rat | LD50 | oral | 250mg/kg (250mg/kg) | National Technical Information Service. Vol. OTS0536502, |
Related Products
- 4-Dimethylaminopyridine
- 4-Dimethylaminopyridine N-oxide
- 112258-59-0
- 1122-59-4
- 112259-66-2
- 1122-60-7
- 1122-61-8
- 1122-62-9
- 112-26-5
- 112266-62-3
- 1122-69-6
- 112270-06-1
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View