-
Name
4-Hydroxy-2-butanone
- EINECS 209-693-6
- CAS No. 590-90-9
- Article Data131
- CAS DataBase
- Density 0.987 g/cm3
- Solubility miscible with water, ethanol and ether
- Melting Point 15°C (estimate)
- Formula C4H8O2
- Boiling Point 156.3 °C at 760 mmHg
- Molecular Weight 88.1063
- Flash Point 55.5 °C
- Transport Information UN 1993
- Appearance Colorless liquid
- Safety 16
- Risk Codes 10
-
Molecular Structure
- Hazard Symbols R10:;
- Synonyms 1-Hydroxybutan-3-one;2-Hydroxyethyl methyl ketone;3-Ketobutan-1-ol;3-Ketobutanol;3-Oxo-1-butanol;3-Oxobutanol;4-Butanol-2-one;4-Hydroxy-2-butanone;Methylolacetone;Monomethylolacetone;NSC 41219;
- PSA 37.30000
- LogP -0.04220
Synthetic route

| Conditions | Yield |
|---|---|
| With silver hexafluoroantimonate; disodium chloro[1,3-bis(2,6-diisopropyl-4-sodiumsulfonatophenyl)imidazol-2-ylidene]gold(I); water at 60℃; for 2h; | 99% |
| With 2CHF3O3S*C29H35NO6P2Pt; water; sodium dodecyl-sulfate at 80℃; Micellar solution; | 11 %Chromat. |
| With cis-dichlorido[1-(3-sodiumsulfonatepropyl)-3-(2,4,6-trimethylphenyl)imidazol-2-ylidene](dimethylsulfoxide)platinum(II); water at 80℃; for 24h; Inert atmosphere; Schlenk technique; | 23 %Spectr. |

| Conditions | Yield |
|---|---|
| With oxalic acid; silica gel In dichloromethane Ambient temperature; | 98% |
| With cerium(III) chloride; sodium iodide In acetonitrile for 21h; Ambient temperature; | 91% |
| With oxalic acid; silica gel In dichloromethane at 20℃; | 87% |

| Conditions | Yield |
|---|---|
| With bis(η5-cyclopentadienyl)hafnium dihydride In isopropyl alcohol at 80℃; for 8h; Product distribution; other dicarbonyl compounds, α,β-unsaturated carbonyl compounds; other temperature; other group IVA metallocene complexes; | 95% |
| With bis(η5-cyclopentadienyl)hafnium dihydride In isopropyl alcohol at 80℃; for 8h; | 95% |

| Conditions | Yield |
|---|---|
| With D-Prolin at 20℃; for 24h; Reagent/catalyst; Inert atmosphere; | 92.3% |
| ambersep 900 OH at 50℃; for 23.5h; Product distribution / selectivity; | 13% |
| Amberlyst A 26 OH at 50℃; for 23.5h; Product distribution / selectivity; | 10% |
-
-
72064-43-8
tert-butyldimethylsilyl 4-phenylbutyl ether

-
-
590-90-9
1-Hydroxy-3-butanone

| Conditions | Yield |
|---|---|
| With sodium periodate In tetrahydrofuran; water at 25℃; | 92% |
-
-
120591-36-8
4-(tert-butyldimethylsilyloxy)butan-2-one

-
-
590-90-9
1-Hydroxy-3-butanone

| Conditions | Yield |
|---|---|
| With sodium periodate In tetrahydrofuran; water at 25℃; | 92% |
-
-
18826-95-4, 107-88-0
1.3-butanediol

-
-
590-90-9
1-Hydroxy-3-butanone

| Conditions | Yield |
|---|---|
| With sodium bromate; sodium hydrogensulfite In water; acetonitrile at 20℃; for 4.25h; Oxidation; | 90% |
| With 3,3-dimethyldioxirane In acetone | 90% |
| With sodium bromate; sodium hydrogensulfite In acetonitrile for 2h; Heating; | 90% |

| Conditions | Yield |
|---|---|
| With magnesium hydroxide; water In methanol at 50℃; for 10h; Reagent/catalyst; Temperature; Sealed tube; | 82% |
-
-
598-32-3
2-hydroxy-3-butene

-
A
-
107-89-1
acetaldol

-
B
-
590-90-9
1-Hydroxy-3-butanone

-
C
-
513-86-0, 52217-02-4
3-hydroxy-2-butanon

-
D
-
78-94-4
methyl vinyl ketone

-
E
-
123-73-9
crotonaldehyde

| Conditions | Yield |
|---|---|
| With hydrogenchloride; lithium tetrachloropalladate(II); p-benzoquinone In water at 25℃; for 0.5h; Rate constant; Mechanism; Kinetics; equilibrium constants; | A 8% B 10% C 79% D 0.7% E 2.3% |

-
-
598-32-3
2-hydroxy-3-butene

-
A
-
107-89-1
acetaldol

-
B
-
590-90-9
1-Hydroxy-3-butanone

-
C
-
513-86-0, 52217-02-4
3-hydroxy-2-butanon

-
D
-
123-73-9
crotonaldehyde

| Conditions | Yield |
|---|---|
| hydrogenchloride; lithium tetrachloropalladate(II); p-benzoquinone In water at 25℃; for 0.5h; Further byproducts given; | A 8% B 10% C 79% D 2.3% |


| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethyl acetate under 760 Torr; Ambient temperature; | 71% |
-
-
627-27-0
homoalylic alcohol

-
A
-
20191-91-7
2-isopropoxytetrahydrofuran

-
B
-
590-90-9
1-Hydroxy-3-butanone

-
C
-
71780-57-9
tetrahydrofuran-2'-yl ether

| Conditions | Yield |
|---|---|
| With oxygen; copper dichloride; Pd(MeCN)2Cl(NO2) 1.) 55 deg C, 2 h; 2.) 30 deg C, 3 h; | A n/a B 70% C n/a |

-
-
627-27-0
homoalylic alcohol

-
-
67-63-0
isopropyl alcohol

-
A
-
20191-91-7
2-isopropoxytetrahydrofuran

-
B
-
590-90-9
1-Hydroxy-3-butanone

-
C
-
71780-57-9
tetrahydrofuran-2'-yl ether

| Conditions | Yield |
|---|---|
| With oxygen; copper dichloride; Pd(MeCN)2Cl(NO2) 1.) 55 deg C, 2 h; 2.) 30 deg C, 3 h; | A n/a B 70% C n/a |


| Conditions | Yield |
|---|---|
| With crosslinked poly-4-vinylpyridine hydrobromide In acetonitrile at 50℃; for 21.5h; electric current, 60mA, 10-30V; Yield given; | A n/a B 59% |
-
-
6117-91-5
(E/Z)-2-buten-1-ol

-
A
-
107-89-1
acetaldol

-
B
-
590-90-9
1-Hydroxy-3-butanone

-
C
-
78-94-4
methyl vinyl ketone

-
D
-
5077-67-8
1-Hydroxy-2-butanone

-
E
-
123-73-9
crotonaldehyde

| Conditions | Yield |
|---|---|
| With hydrogenchloride; lithium tetrachloropalladate(II); p-benzoquinone In water at 25℃; for 0.5h; Rate constant; Mechanism; Kinetics; equilibrium constants; | A 39% B 49% C 0.9% D 2.6% E 8.5% |

-
-
6117-91-5
(E/Z)-2-buten-1-ol

-
A
-
107-89-1
acetaldol

-
B
-
590-90-9
1-Hydroxy-3-butanone

-
C
-
5077-67-8
1-Hydroxy-2-butanone

-
D
-
123-73-9
crotonaldehyde

| Conditions | Yield |
|---|---|
| hydrogenchloride; lithium tetrachloropalladate(II); p-benzoquinone In water at 25℃; for 0.5h; Further byproducts given; | A 39% B 49% C 2.6% D 8.5% |


| Conditions | Yield |
|---|---|
| With lithium hypochlorite In water Ambient temperature; | A 36% B 3% |
-
-
78-18-2
1-hydroxycyclohexyl 1-hydroperoxycyclohexyl peroxide

-
-
78-94-4
methyl vinyl ketone

-
A
-
73154-83-3
2,12-Tridecanedione

-
B
-
590-90-9
1-Hydroxy-3-butanone

-
C
-
333-60-8
2-oxodecanoic acid

-
D
-
108-94-1
cyclohexanone

| Conditions | Yield |
|---|---|
| With titanium(III) chloride; acetic acid for 3h; Ambient temperature; Yields of byproduct given. Title compound not separated from byproducts; | A 26% B n/a C n/a D n/a |
| With titanium(III) chloride; acetic acid for 3h; Ambient temperature; Yield given. Title compound not separated from byproducts; | A 26% B n/a C n/a D n/a |


| Conditions | Yield |
|---|---|
| at 120 - 180℃; |

| Conditions | Yield |
|---|---|
| With mercury(II) sulfate at 70 - 90℃; | |
| With mercury(II) sulfate at 70 - 90℃; |

| Conditions | Yield |
|---|---|
| With sulfuric acid at 25℃; | |
| With calcium hydroxide; formaldehyd at 25℃; | |
| With sulfuric acid | |
| With potassium carbonate |
-
-
50-00-0
formaldehyd

-
-
67-64-1
acetone

-
A
-
73255-29-5
3-(hydroxymethyl)but-3-en-2-one

-
B
-
590-90-9
1-Hydroxy-3-butanone

| Conditions | Yield |
|---|---|
| With potassium hydroxide | |
| With potassium carbonate at 30 - 35℃; |


| Conditions | Yield |
|---|---|
| With mercury(II) diacetate; acetic acid |
-
-
13492-60-9
4-hydroxy-3-phenylsulfanyl-butan-2-one

-
-
590-90-9
1-Hydroxy-3-butanone

| Conditions | Yield |
|---|---|
| With hydrogen; nickel In 1,4-dioxane |

| Conditions | Yield |
|---|---|
| With ozone-containing oxygen; dimethylsulfide 1.) methanol, -78 deg C, 2.) -78 deg C -> room temperature; Yield given. Multistep reaction; |
-
-
58408-33-6
4,5-dihydro-1-(3-oxobutyl)-4-phenyl-1H-1,2,3,4-tetrazole-5-thione

-
A
-
590-90-9
1-Hydroxy-3-butanone

-
B
-
53079-79-1
1-Phenyl-1H-tetrazole-5-thiol anion

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |

| Conditions | Yield |
|---|---|
| With oxygen; cobalt(II) bis(salicylidene-γ-iminopropyl)methylamine |

| Conditions | Yield |
|---|---|
| at 30℃; for 43h; Kluyveromyces lactis IFO 1267, potassium phosphate buffer; | A 38 mmol B n/a |
| at 30℃; for 43h; Kluyveromyces lactis IFO 1267, potassium phosphate buffer; |
-
-
18826-95-4, 107-88-0
1.3-butanediol

-
A
-
107-89-1
acetaldol

-
B
-
590-90-9
1-Hydroxy-3-butanone

-
C
-
75-07-0
acetaldehyde

-
D
-
78-94-4
methyl vinyl ketone

-
E
-
78-93-3
butanone

-
F
-
78-92-2, 15892-23-6
iso-butanol

| Conditions | Yield |
|---|---|
| L-1930 Product distribution; various catalysts, temp.; |

-
-
18826-95-4, 107-88-0
1.3-butanediol

-
A
-
590-90-9
1-Hydroxy-3-butanone

-
B
-
67-64-1
acetone

-
C
-
78-93-3
butanone

| Conditions | Yield |
|---|---|
| MK-I at 120℃; under 760 Torr; Product distribution; Thermodynamic data; 150 - 220 deg C, different catalysts, E(excit.); | A 29.46 % Chromat. B 0.6 % Chromat. C 3.3 % Chromat. |


| Conditions | Yield |
|---|---|
| With thionyl chloride; N,N-dimethyl-formamide In dichloromethane at 20℃; for 14h; Inert atmosphere; | 100% |
| With hydrogenchloride; water at 0℃; | |
| With thionyl chloride; benzene at 75℃; | |
| With thionyl chloride In dichloromethane at 20℃; | 1.06 g |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 2h; | 100% |
| With pyridine | |
| With pyridine |
-
-
13154-24-0
triisopropylsilyl chloride

-
-
590-90-9
1-Hydroxy-3-butanone

-
-
98264-29-0
4-((triisopropylsilyl)oxy)butan-2-one

| Conditions | Yield |
|---|---|
| With 1H-imidazole at 20℃; for 19.5h; Inert atmosphere; | 100% |
| With 1H-imidazole | |
| With 1H-imidazole In dichloromethane at 20℃; |
-
-
590-90-9
1-Hydroxy-3-butanone

-
-
18162-48-6
tert-butyldimethylsilyl chloride

-
-
120591-36-8
4-(tert-butyldimethylsilyloxy)butan-2-one

| Conditions | Yield |
|---|---|
| With 1H-imidazole In dichloromethane at 20℃; for 18h; | 100% |
| With dmap; triethylamine In dichloromethane at 20℃; for 2h; | 98% |
| With 1H-imidazole In dichloromethane for 1.5h; Inert atmosphere; | 94% |
-
-
590-90-9
1-Hydroxy-3-butanone

| Conditions | Yield |
|---|---|
| With N-nitro-2,4,6-trimethylpyridinium tetrafluoroborate In acetonitrile at 0℃; for 2h; | 100% |
-
-
590-90-9
1-Hydroxy-3-butanone

-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
97250-25-4
tert-Butyldiphenylsilyl β-hydroxybutanone

| Conditions | Yield |
|---|---|
| With 1H-imidazole In dichloromethane at 20℃; for 16h; Cooling with ice; | 100% |
-
-
110-87-2
3,4-dihydro-2H-pyran

-
-
590-90-9
1-Hydroxy-3-butanone

-
-
20705-59-3
4-(tetrahydropyran-2-yloxy)butan-2-one

| Conditions | Yield |
|---|---|
| With pyridinium p-toluenesulfonate In dichloromethane at 20℃; for 4h; | 99% |
| With toluene-4-sulfonic acid In dichloromethane at 0℃; for 1h; | 92% |
| Stage #1: 3,4-dihydro-2H-pyran; 1-Hydroxy-3-butanone With toluene-4-sulfonic acid In dichloromethane; water at 0℃; for 2h; Stage #2: With sodium hydrogencarbonate for 0.5h; | 83% |
-
-
590-90-9
1-Hydroxy-3-butanone


| Conditions | Yield |
|---|---|
| (+)-(1R)-(1,2,2,3,3,7,7,8,8,12,12,13,13,17,17,18,18-Heptadecamethyl-10,20-diaza-octahydroporphinato)nickel-perchlorat In toluene byproducts: methanol; mixt. refluxed for 2 h while methanol removed by mol. sieve; solvent evapd., residue dissolved in hexane/acetonitrile, washed, filtered, solvent evapd., residue dried at 60°C, 0.01 Torr for 24 h; | 98.7% |

| Conditions | Yield |
|---|---|
| With <((R)-(1,1'-binaphthyl-2,2'-diyl)bis(diphenylphosphine))RuCl2>2NEt3; hydrogen In methanol at 50℃; under 38000 Torr; for 24h; | 98% |
| With hydrogen; dichloro(benzene)ruthenium(II) dimer; (R)-2,2'-bis(diphenylphosphanyl)-1,1'-binaphthyl In ethanol at 30℃; under 76000.1 Torr; for 40h; Yields of byproduct given. Title compound not separated from byproducts; | 96% |
| With methanol; phosphate buffer at 30℃; for 6.5h; Candida boidinii KK912 (IFO 10574); | 60% |
-
-
590-90-9
1-Hydroxy-3-butanone

-
-
18826-95-4, 107-88-0
1.3-butanediol

| Conditions | Yield |
|---|---|
| With hydrogen; sodium hydroxide In water at 25℃; under 7500.75 Torr; for 1h; Pressure; Reagent/catalyst; Solvent; Temperature; Autoclave; | 98% |
| With Cp*Ir(6,6'-dionato-2,2'-bipyridine)(H2O); isopropyl alcohol In water at 82℃; for 6h; Inert atmosphere; Schlenk technique; chemoselective reaction; | 91% |
| With potassium borohydride In tetrahydrofuran; water at 45℃; for 4h; Reagent/catalyst; Temperature; Cooling with ice; | 83% |

| Conditions | Yield |
|---|---|
| With bis(cyclopentadienyl)dihydrozirconium In tetrahydrofuran for 5h; Ambient temperature; | 98% |
-
-
590-90-9
1-Hydroxy-3-butanone

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
446876-96-6
4-((p-toluenesulfonyl)oxy)-2-butanone

| Conditions | Yield |
|---|---|
| With pyridine In dichloromethane for 4h; Cooling with ice; | 98% |
| With pyridine In dichloromethane at 0 - 20℃; | 75% |
| With pyridine | 75% |
-
-
590-90-9
1-Hydroxy-3-butanone

-
-
24621-61-2
1,3-butanediol

| Conditions | Yield |
|---|---|
| With Ru2Cl4-(S)-(2,2'-bis(diphenylphosphino)-1,1'-binaphthyl)2*NEt3; hydrogen In methanol at 50℃; under 38000 Torr; for 24h; | 97% |
| at 30℃; for 48h; Candida parapsilosis IFO 1396; | 60% |
| With sodium perborate; (+)-β-chlorodiisopinocampheylborane 1.) THF, 0 deg C, 12 h; 2.) THF, EE, H2O, 0 deg C, 30 min; Yield given. Multistep reaction; |

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 0 - 20℃; for 3h; | 96% |
-
-
590-90-9
1-Hydroxy-3-butanone

-
-
598-21-0
2-Bromoacetyl bromide

-
-
89641-46-3
bromo-acetic acid-(3-oxo-butyl ester)

| Conditions | Yield |
|---|---|
| With pyridine at 0℃; | 96% |
-
-
75-77-4
chloro-trimethyl-silane

-
-
590-90-9
1-Hydroxy-3-butanone

-
-
81852-67-7
4-((trimethylsilyl)oxy)-3-butanone

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 0 - 20℃; | 95% |
| With triethylamine In diethyl ether | 94% |
| With triethylamine In dichloromethane at 25℃; for 2h; | 80% |

| Conditions | Yield |
|---|---|
| With bis(cyclopentadienyl)dihydrozirconium In tetrahydrofuran for 5h; Ambient temperature; | 95% |
| With scandium tris(trifluoromethanesulfonate) In tetrahydrofuran at 20℃; for 2.5h; Reduction; Tishchenko; |

| Conditions | Yield |
|---|---|
| With iodine In dimethyl sulfoxide at 20℃; for 8h; Solvent; Reagent/catalyst; Inert atmosphere; | 94% |
| With iodine In dimethyl sulfoxide at 20℃; for 8h; Reagent/catalyst; Solvent; Time; | 94% |
| With tert.-butylnitrite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical In dimethyl sulfoxide at 20℃; for 12h; Catalytic behavior; Reagent/catalyst; Solvent; Time; Inert atmosphere; | 88% |
-
-
288-32-4
1H-imidazole

-
-
590-90-9
1-Hydroxy-3-butanone

-
-
7757-82-6
sodium sulfate

-
-
58479-61-1
tert-butylchlorodiphenylsilane

| Conditions | Yield |
|---|---|
| In (2S)-N-methyl-1-phenylpropan-2-amine hydrate; dichloromethane | 94% |


| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine at 150℃; for 1h; Inert atmosphere; Neat (no solvent); | 94% |
| With N-ethyl-N,N-diisopropylamine at 150℃; for 2h; | 22.2% |
| With N-ethyl-N,N-diisopropylamine at 150℃; for 2h; Inert atmosphere; | |
| With N-ethyl-N,N-diisopropylamine at 150℃; for 2h; Inert atmosphere; | |
| With N-ethyl-N,N-diisopropylamine at 150℃; for 2h; Inert atmosphere; |
-
-
116-11-0
2-Methoxypropene

-
-
590-90-9
1-Hydroxy-3-butanone

-
-
82495-49-6
4-(1-Methoxy-1-methyl-ethoxy)-butan-2-one

| Conditions | Yield |
|---|---|
| With trichlorophosphate | 93% |
| With toluene-4-sulfonic acid In tetrahydrofuran |
-
-
590-90-9
1-Hydroxy-3-butanone

-
-
350848-02-1
2-[(4-methoxybenzyl)oxy]-3-nitropyridine

-
-
85846-38-4
4-((4-methoxybenzyl)oxy)butan-2-one

| Conditions | Yield |
|---|---|
| With trimethylsilyl trifluoromethanesulfonate In dichloromethane at 20℃; for 0.0833333h; | 93% |
-
-
590-90-9
1-Hydroxy-3-butanone

-
-
107-30-2
chloromethyl methyl ether

-
-
26822-73-1
4-(methoxymethoxy)butan-2-one

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane for 17h; Reflux; Inert atmosphere; | 93% |

| Conditions | Yield |
|---|---|
| With iodine In dimethyl sulfoxide at 20℃; for 8h; Inert atmosphere; | 93% |
| With iodine In dimethyl sulfoxide at 20℃; for 8h; | 93% |
| With tert.-butylnitrite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical In dimethyl sulfoxide at 20℃; for 9h; Inert atmosphere; Green chemistry; | 86% |
| With tert.-butylnitrite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical In dimethyl sulfoxide at 20℃; for 9h; Inert atmosphere; | 86% |

| Conditions | Yield |
|---|---|
| With pyridine; p-toluenesulfonic acid monohydrate In dichloromethane at 20℃; | 92% |
| With cobalt(II) chloride In acetonitrile Ambient temperature; | 81% |
| With hydrogenchloride |
-
-
590-90-9
1-Hydroxy-3-butanone

-
-
67373-56-2
chlorodimethyl(1,1,2-trimethylpropyl)silane

-
-
140139-14-6
4-dimethylthexylsilyloxy-2-butanone

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane | 92% |
4-Hydroxy-2-butanone Specification
The IUPAC name of 2-Butanone, 4-hydroxy- is 4-hydroxybutan-2-one. With the CAS registry number 590-90-9 and EINECS 209-693-6, it is also named as 3-Ketobutan-1-ol. The product's categories are Straight Chain Compounds; Ketone. It is colorless liquid which is miscible with water, ethanol and ether and flammable. Additionally, this chemical should be sealed in the container and stored in the cool and dry place.
The other characteristics of this product can be summarized as: (1)ACD/LogP: -0.79; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -0.78; (4)ACD/LogD (pH 7.4): -0.78; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 8.92; (8)ACD/KOC (pH 7.4): 8.92; (9)#H bond acceptors: 2; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 3; (12)Polar Surface Area: 26.3 Å2; (13)Index of Refraction: 1.411; (14)Molar Refractivity: 22.14 cm3; (15)Molar Volume: 89.1 cm3; (16)Polarizability: 8.77×10-24 cm3; (17)Surface Tension: 32.5 dyne/cm; (18)Density: 0.987 g/cm3; (19)Flash Point: 55.5 °C; (20)Enthalpy of Vaporization: 45.77 kJ/mol; (21)Boiling Point: 156.3 °C at 760 mmHg; (22)Vapour Pressure: 1.05 mmHg at 25°C.
Preparation of 2-Butanone, 4-hydroxy-: It can be obtained by (+-)-butane-1,3-diol. This reaction needs reagents N-hydroxyphthalimide, O2, Co(acac)2 and solvent acetonitrile by heating. The reaction time is 12 hours. The yield is 61 % Chromat.
.gif)
Uses of 2-Butanone, 4-hydroxy-: It is used as pharmaceutical intermediate and in organic synthesis. For example, it can react with 3,4-dihydro-2H-pyran to get 4-tetrahydropyran-2-yloxy-butan-2-one. This reaction needs reagent p-toluenesulfonic acid and solvent CH2Cl2 at temperature of 0 °C. The reaction time is 1 hours. The yield is 92%.
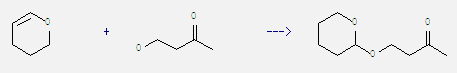
People can use the following data to convert to the molecule structure.
1. SMILES:O=C(C)CCO
2. InChI:InChI=1/C4H8O2/c1-4(6)2-3-5/h5H,2-3H2,1H3
3. InChIKey:LVSQXDHWDCMMRJ-UHFFFAOYAN
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View