-
Name
4-Nitro-3-phenyl-L-alanine
- EINECS 213-446-8
- CAS No. 949-99-5
- Article Data77
- CAS DataBase
- Density 1.408 g/cm3
- Solubility very soluble in water
- Melting Point 236-239 °C (dec.)
- Formula C9H10N2O4
- Boiling Point 414.1 °C at 760 mmHg
- Molecular Weight 210.189
- Flash Point 204.2 °C
- Transport Information
- Appearance white to light yellow crystal powder
- Safety 26-36/37/39-22-45-16
- Risk Codes 36/37/38-22-34-11
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, C,
C, F
F
- Synonyms L-Phenylalanine,4-nitro-;Alanine,3-(p-nitrophenyl)-, L- (8CI);(S)-4-Nitrophenylalanine;3-(4-Nitrophenyl)-L-alanine;4-Nitro-L-phenylalanine;L-4-Nitrophenylalanine;L-p-Nitrophenylalanine;NSC 152925;p-Nitro-L-phenylalanine;p-Nitrophenylalanine;H-Phe(4-NO2)-OH;
- PSA 109.14000
- LogP 1.77270
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: L-phenylalanine With sulfuric acid; nitric acid at 20℃; for 5h; Stage #2: With sodium hydroxide pH=2 - 3; | 95.2% |
| Stage #1: L-phenylalanine With sulfuric acid; nitric acid In water at 20℃; Stage #2: With sodium hydroxide In water pH=2 - 3; | 95.2% |
| Stage #1: L-phenylalanine With sulfuric acid; nitric acid at 10℃; for 2.5h; Stage #2: With ammonia In water pH=5 - 6; | 94% |
-
-
17363-92-7
N-acetyl-4-nitro-L-phenylalanine

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| With Acylase I enzyme from porcine kidney; water at 24.9℃; Rate constant; | 83% |
| With hydrogenchloride |

| Conditions | Yield |
|---|---|
| With ammonium hydroxide; borane-ammonia complex In aq. buffer at 37℃; pH=9.6; Enzymatic reaction; | 57% |
| With ammonium bisulphate; EncP-R299K variant at 55℃; for 22h; pH=8.3; Enzymatic reaction; stereoselective reaction; | n/a |
-
-
1991-83-9, 2922-40-9, 56613-61-7, 949-99-5
4-nitrophenylalanine

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| With sodium hydroxide; oxygen at 30℃; for 42h; pH=7.3; aq. buffer; Enzymatic reaction; optical yield given as %ee; enantioselective reaction; | 51% |
-
-
619-89-6
p-nitrocinnamic acid

-
A
-
501120-99-6
(R)-3-amino-3-(4'-nitrophenyl)propanoic acid

-
B
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| With ammonia at 25℃; pH=9; Kinetics; aq. buffer; Enzymatic reaction; regioselective reaction; | A 8% B n/a |
-
-
101468-97-7
N-benzoyl-(4'-nitro)phenylalanine

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| With acylase-substance from pseudomonas |
-
-
81677-60-3
(L)-p-Nitrophenylalanine methyl ester

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| With water; chymotrypsin at 25℃; Rate constant; |
-
-
62561-97-1
N-acetyl-4-nitro-DL-phenylalanine

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| With phosphate buffer; acylase I from porcine kidney at 24.9℃; for 2h; |
-
-
56-40-6
glycine

-
-
100-11-8
1-bromomethyl-4-nitro-benzene

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| Stage #1: glycine With pyridine-2-carboxylic acid(2-benzoyl-phenyl)-amide; sodium methylate; nickel(II) nitrate In methanol for 0.333333h; Heating; Stage #2: 1-bromomethyl-4-nitro-benzene With sodium hydroxide; (R)-2-amino-2'-hydroxy-1,1'-binaphthyl In dichloromethane at 20℃; for 0.0666667h; Stage #3: With hydrogenchloride In methanol; water Heating; |

-
-
38335-24-9
3-(4-nitrophenyl)-2-oxopropanoic acid

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| With Celite; phenylalanine dehydrogenase N145A; potassium chloride; ammonium chloride; 1,4-dihydronicotinamide adenine dinucleotide In ethanol at 20℃; for 18h; pH=8.5; |
-
-
33305-77-0
Boc-Phe(p-NO2)-OH

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| With hydrogenchloride In methanol at 0 - 20℃; for 5h; | |
| With trifluoroacetic acid In dichloromethane |

| Conditions | Yield |
|---|---|
| With carbon dioxide; Taxus chinensis phenylalanine aminomutase; ammonia In water pH=10; Kinetics; Enzymatic reaction; optical yield given as %ee; enantioselective reaction; | |
| With ammonium carbamate at 30℃; for 168h; pH=9.1; Time; | n/a |
-
-
1991-83-9, 2922-40-9, 56613-61-7, 949-99-5
4-nitrophenylalanine

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| With chiral stationary phase including isopropyl-functionalized CF6 In methanol; acetic acid; triethylamine; acetonitrile at 0℃; Purification / work up; Resolution of racemate; | |
| With dichloro bis(acetonitrile) palladium(II); (S)-(1,1'-binaphthalene)-2,2'-diylbis(diphenylphosphine) In aq. phosphate buffer; dichloromethane at 25℃; pH=7; Equilibrium constant; Reagent/catalyst; Solvent; Temperature; Resolution of racemate; enantioselective reaction; | |
| With (S)-2-hydroxy-2'-(3-(N-phenylcarbamoylamino)benzyl)-1,1'-binaphthyl-3-carboxaldehyde In dimethyl sulfoxide Resolution of racemate; stereoselective reaction; | A n/a B n/a |

| Conditions | Yield |
|---|---|
| With CmII Enzymatic reaction; | |
| With Streptomyces venezuelae CmlI; oxygen In aq. buffer at 4℃; pH=9; Kinetics; Enzymatic reaction; |
-
-
17224-90-7
(S)-2-(((benzyloxy)carbonyl)amino)-3-(4-nitrophenyl)propanoic acid

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| With ethylenediaminetetraacetic acid; phenylmethylsulphonyl fluoride; water In aq. phosphate buffer at 30℃; Kinetics; Reagent/catalyst; Microbiological reaction; Enzymatic reaction; |
-
-
62561-97-1
N-acetyl-4-nitro-DL-phenylalanine

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
89615-73-6
N-acetyl-4-nitro-D-phenylalanine

| Conditions | Yield |
|---|---|
| With lipase AS 'Amano' at 35℃; for 24h; pH=6.5; Enzymatic reaction; enantioselective reaction; | A n/a B n/a |
-
-
1991-83-9, 2922-40-9, 56613-61-7, 949-99-5
4-nitrophenylalanine

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
89615-73-6
N-acetyl-4-nitro-D-phenylalanine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: triethylamine / water; N,N-dimethyl-formamide / 20 °C 2: lipase AS 'Amano' / 24 h / 35 °C / pH 6.5 / Enzymatic reaction View Scheme |
-
-
882-06-4
4-nitro-trans-cinnamic acid

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| With ammonium hydroxide at 30℃; for 0.33h; pH=9.5; Reagent/catalyst; Enzymatic reaction; | A n/a B n/a |
| With ammonium hydroxide at 30℃; for 22h; pH=9.5; Enzymatic reaction; | A n/a B n/a |
| With ammonia at 30℃; for 17h; pH=10; Time; | A n/a B n/a |
-
-
1627101-69-2
(R)-2-amino-3[(R)-2H]-3(4-nitrophenyl)propanoic acid hydrochloride

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 2: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme |
-
-
122-04-3
4-nitro-benzoyl chloride

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: lithium aluminium deuteride / diethylene glycol dimethyl ether; tert-butyl alcohol / 0.5 h / -78 °C 2: sodium acetate / acetic anhydride / 100 °C 3: methanol / 0.5 h / 20 °C 4: hydrogen; (-)-1,2-bis-[(2R,5R)-2,5-diethylphospholano]benzene(cyclooctadiene)rhodium(I) tetrafluoroborate / methanol / 1.5 h / 5171.62 Torr 5: hydrogenchloride / water; acetone / 7 h / Reflux 6: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 7: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme | |
| Multi-step reaction with 8 steps 1: lithium aluminium deuteride / diethylene glycol dimethyl ether; tert-butyl alcohol / 0.5 h / -78 °C 2: sodium acetate / acetic anhydride / 100 °C 3: methanol / 0.5 h / 20 °C 4: hydrogen; (+)-1,2-bis[(2S,5S)-2,5-diethylphospholano]benzene(cyclooctadiene)rhodium(I) tetrafluoroborate / methanol / 1.5 h / 5171.62 Torr 5: lithium hydroxide / methanol; water; tetrahydrofuran / 2 h / 20 °C 6: cobalt(II) chloride hexahydrate; N-acetyl amino acid racemase G291D/F323Y; D-acylase / aq. buffer / 23 h / 37 °C / pH 8.0 / Enzymatic reaction 7: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 8: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme |
-
-
94781-50-7
α-deuterio-4-nitro-benzaldehyde

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: sodium acetate / acetic anhydride / 100 °C 2: methanol / 0.5 h / 20 °C 3: hydrogen; (-)-1,2-bis-[(2R,5R)-2,5-diethylphospholano]benzene(cyclooctadiene)rhodium(I) tetrafluoroborate / methanol / 1.5 h / 5171.62 Torr 4: hydrogenchloride / water; acetone / 7 h / Reflux 5: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 6: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme | |
| Multi-step reaction with 7 steps 1: sodium acetate / acetic anhydride / 100 °C 2: methanol / 0.5 h / 20 °C 3: hydrogen; (+)-1,2-bis[(2S,5S)-2,5-diethylphospholano]benzene(cyclooctadiene)rhodium(I) tetrafluoroborate / methanol / 1.5 h / 5171.62 Torr 4: lithium hydroxide / methanol; water; tetrahydrofuran / 2 h / 20 °C 5: cobalt(II) chloride hexahydrate; N-acetyl amino acid racemase G291D/F323Y; D-acylase / aq. buffer / 23 h / 37 °C / pH 8.0 / Enzymatic reaction 6: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 7: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme |
-
-
1627096-62-1
[4-2H]-4(4-nitrobenzylidene)-2-methyloxazol-5(4H)-one

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: methanol / 0.5 h / 20 °C 2: hydrogen; (-)-1,2-bis-[(2R,5R)-2,5-diethylphospholano]benzene(cyclooctadiene)rhodium(I) tetrafluoroborate / methanol / 1.5 h / 5171.62 Torr 3: hydrogenchloride / water; acetone / 7 h / Reflux 4: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 5: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme | |
| Multi-step reaction with 6 steps 1: methanol / 0.5 h / 20 °C 2: hydrogen; (+)-1,2-bis[(2S,5S)-2,5-diethylphospholano]benzene(cyclooctadiene)rhodium(I) tetrafluoroborate / methanol / 1.5 h / 5171.62 Torr 3: lithium hydroxide / methanol; water; tetrahydrofuran / 2 h / 20 °C 4: cobalt(II) chloride hexahydrate; N-acetyl amino acid racemase G291D/F323Y; D-acylase / aq. buffer / 23 h / 37 °C / pH 8.0 / Enzymatic reaction 5: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 6: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme |
-
-
1627098-43-4
[4-2H]-methyl 2-acetamido-3(4-nitrophenyl)acrylate

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: hydrogen; (-)-1,2-bis-[(2R,5R)-2,5-diethylphospholano]benzene(cyclooctadiene)rhodium(I) tetrafluoroborate / methanol / 1.5 h / 5171.62 Torr 2: hydrogenchloride / water; acetone / 7 h / Reflux 3: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 4: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme | |
| Multi-step reaction with 5 steps 1: hydrogen; (+)-1,2-bis[(2S,5S)-2,5-diethylphospholano]benzene(cyclooctadiene)rhodium(I) tetrafluoroborate / methanol / 1.5 h / 5171.62 Torr 2: lithium hydroxide / methanol; water; tetrahydrofuran / 2 h / 20 °C 3: cobalt(II) chloride hexahydrate; N-acetyl amino acid racemase G291D/F323Y; D-acylase / aq. buffer / 23 h / 37 °C / pH 8.0 / Enzymatic reaction 4: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 5: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme |
-
-
1627099-40-4
(R)-methyl 2-acetamido-3[2H]-3(4-nitrophenyl)propanoate

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: hydrogenchloride / water; acetone / 7 h / Reflux 2: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 3: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme |
-
-
1627099-66-4
(S)-methyl 2-acetamido-3-[2H]-3(4-nitrophenyl)propanoate

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: lithium hydroxide / methanol; water; tetrahydrofuran / 2 h / 20 °C 2: cobalt(II) chloride hexahydrate; N-acetyl amino acid racemase G291D/F323Y; D-acylase / aq. buffer / 23 h / 37 °C / pH 8.0 / Enzymatic reaction 3: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 4: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme |
-
-
1627100-30-4
(R)-2-amino-3[(S)-2H]-3(4-nitrophenyl)propanoic acid hydrochloride

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 2: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme |
-
-
1627100-31-5
(S)-2-acetamido-3-[2H]-3(4-nitrophenyl)propanoic acid

-
A
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
B
-
56613-61-7
(R)-4-nitro-phenylalanine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: cobalt(II) chloride hexahydrate; N-acetyl amino acid racemase G291D/F323Y; D-acylase / aq. buffer / 23 h / 37 °C / pH 8.0 / Enzymatic reaction 2: E. coli BL21(DE3) whole cells expressing Anabaena variabilis phenylalanine ammonia lyase / aq. buffer / 48 h / 30 °C / pH 8.3 / Enzymatic reaction 3: ammonium hydroxide / 22 h / 30 °C / pH 9.5 / Enzymatic reaction View Scheme |
-
-
65615-89-6
methyl (S)-2-((tert-butoxycarbonyl)amino)-3-(4-nitrophenyl)propanoate

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: lithium hydroxide / tetrahydrofuran; water / 20 °C 2: trifluoroacetic acid / dichloromethane View Scheme |

| Conditions | Yield |
|---|---|
| With borane-ammonia complex Enzymatic reaction; |
-
-
67-56-1
methanol

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
17193-40-7
(S)-4-nitrophenylalanine methyl ester hydrochloride

| Conditions | Yield |
|---|---|
| With thionyl chloride at 20℃; | 100% |
| With thionyl chloride at 5 - 45℃; | 100% |
| With thionyl chloride Ambient temperature; | 99% |
-
-
13139-17-8
N-(Benzyloxycarbonyloxy)succinimide

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
17224-90-7
(S)-2-(((benzyloxy)carbonyl)amino)-3-(4-nitrophenyl)propanoic acid

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In water; acetonitrile at 20℃; | 100% |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
501-53-1
benzyl chloroformate

-
-
17224-90-7
(S)-2-(((benzyloxy)carbonyl)amino)-3-(4-nitrophenyl)propanoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium carbonate In water at 6 - 9℃; for 1h; | 99.5% |
| With sodium hydroxide; sodium carbonate In water at 6 - 9℃; for 1h; | 99.5% |
| Stage #1: 4-nitro-3-phenyl-L-alanine; benzyl chloroformate With sodium hydroxide; sodium carbonate In water at 6 - 9℃; for 1h; Stage #2: With hydrogenchloride In water pH=2; | 99.5% |
-
-
67-56-1
methanol

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
81677-60-3
(L)-p-Nitrophenylalanine methyl ester

| Conditions | Yield |
|---|---|
| With thionyl chloride at 0℃; for 2h; Inert atmosphere; Reflux; | 98.1% |
| With thionyl chloride at 0℃; for 1h; Reflux; | 91% |
| With thionyl chloride at 0℃; for 1h; Reflux; | 91% |

| Conditions | Yield |
|---|---|
| In benzene at 120℃; for 12h; | 98% |
| In benzene for 10h; Heating; Yield given; |

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
4659-45-4
2,6-Dichlorobenzoyl chloride

-
-
401906-14-7
Nα-(2,6-dichlorobenzoyl)-4-nitro-L-phenylalanine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water; acetone at 10 - 15℃; for 2h; pH=~ 14; | 97% |
| Stage #1: 4-nitro-3-phenyl-L-alanine; 2,6-Dichlorobenzoyl chloride With sodium hydroxide In water; acetone at 15℃; pH=>= 13; Cooling in ice; Stage #2: With hydrogenchloride In water; acetone | 95% |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
33173-27-2
(S)-3-(4-nitrophenyl)-2-hydroxypropionic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid; sodium nitrite In water; acetone at -5 - 20℃; for 18h; | 96% |
| With sulfuric acid; sodium nitrite In water; acetone at -5 - 20℃; for 18h; | 96% |
| With sulfuric acid; water; sodium nitrite at 0 - 20℃; | 77% |
| With sulfuric acid; sodium nitrite at 0 - 20℃; | 55% |
| With sulfuric acid; sodium nitrite |
-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
33305-77-0
Boc-Phe(p-NO2)-OH

| Conditions | Yield |
|---|---|
| With sodium hydroxide In 1,4-dioxane; water for 3h; Ambient temperature; | 96% |
| In 1,4-dioxane; water at 0 - 20℃; for 24h; Substitution; | 92% |
| In tetrahydrofuran; sodium hydroxide | 88% |

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 20℃; for 24h; Inert atmosphere; Schlenk technique; Darkness; | 94.7% |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
58816-66-3
(S)-2-amino-3-(4-nitrophenyl)propionic acid ethyl ester hydrochloride

| Conditions | Yield |
|---|---|
| Stage #1: ethanol With thionyl chloride at 0 - 20℃; for 0.75h; Stage #2: 4-nitro-3-phenyl-L-alanine Heating / reflux; | 92% |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
49607-21-8
(2S)-2-amino-3-(2,4-dinitrophenyl)propanoic acid

| Conditions | Yield |
|---|---|
| With sulfuric acid; uronium nitrate at 0 - 60℃; for 5h; Temperature; Reagent/catalyst; Time; regioselective reaction; | 87.8% |

| Conditions | Yield |
|---|---|
| With barium sulfate; hydrogen; palladium | 87% |
| With rosenmund catalyst; hydrogen In water at 20℃; for 3h; | 85% |
| With methanol; aluminum oxide; palladium/alumina Hydrogenation; | |
| With hydrogen; palladium on barium sulfate In water | |
| With hydrogen; Pd-BaSO4 In water |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
82911-69-1
N-(9H-fluoren-2-ylmethoxycarbonyloxy)succinimide

-
-
95753-55-2
N-α-FMOC-4-nitro-L-phenylalanine

| Conditions | Yield |
|---|---|
| Acylation; | 87% |
| With sodium hydroxide In water; acetonitrile at 20℃; for 3h; | 67% |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
75315-63-8
N-(benzyloxycarbonyl)succinimide

-
-
17224-90-7
(S)-2-(((benzyloxy)carbonyl)amino)-3-(4-nitrophenyl)propanoic acid

| Conditions | Yield |
|---|---|
| With triethylamine In 1,4-dioxane; water | 87% |
-
-
28920-43-6
(fluorenylmethoxy)carbonyl chloride

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
95753-55-2
N-α-FMOC-4-nitro-L-phenylalanine

| Conditions | Yield |
|---|---|
| With sodium carbonate In 1,4-dioxane 0 deg C, then Rt 2 h.; | 86% |
| With sodium carbonate In 1,4-dioxane at 0℃; for 3h; | 64% |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
89288-22-2
(S)-(-)-2-amino-3-(4-nitrophenyl)propan-1-ol

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; iodine In tetrahydrofuran at 70℃; for 18h; | 86% |
| With lithium borohydride; chloro-trimethyl-silane In tetrahydrofuran at 0 - 20℃; | 68% |
| With sodium tetrahydroborate; iodine In tetrahydrofuran at 60℃; for 20h; | 67% |
-
-
5292-43-3
bromoacetic acid tert-butyl ester

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
1619224-40-6
2-(bis-2-tert-butoxycarbonylmethylamino)-3-(4-nitrophenyl)propanoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitro-3-phenyl-L-alanine With potassium carbonate In acetonitrile for 0.5h; Inert atmosphere; Stage #2: bromoacetic acid tert-butyl ester In acetonitrile for 18h; Inert atmosphere; Reflux; | 85% |
-
-
34404-33-6
N-tertiarybutoxycarbonyl-D-alanine N-hydroxysuccinimide ester

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine In N,N-dimethyl-formamide for 4h; Ambient temperature; | 80% |
-
-
64-17-5
ethanol

-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
34276-53-4
ethyl (S)-2-amino-3-(4-nitrophenyl)propionate

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 3h; Heating; | 80% |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
126513-13-1
p-[N-(2-methyl-4-oxo-3,4-dihydroquinazolin-6-ylmethyl)-N-(prop-2-ynyl)amino]benzoyl azide

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In N,N-dimethyl-formamide for 20h; | 80% |

| Conditions | Yield |
|---|---|
| In water at 80℃; for 24h; | 76% |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
71159-98-3
2-Formyl-21,23-dihydro-5,10,15,20-tetraphenylporphyrin

| Conditions | Yield |
|---|---|
| Stage #1: 4-nitro-3-phenyl-L-alanine; 2-Formyl-21,23-dihydro-5,10,15,20-tetraphenylporphyrin With sodium cyanoborohydride In tetrahydrofuran; methanol for 24h; Reflux; Stage #2: Heating; | 75% |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
108-24-7
acetic anhydride

-
-
17363-92-7
N-acetyl-4-nitro-L-phenylalanine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In acetone for 1h; | 74% |
| In water |

| Conditions | Yield |
|---|---|
| Stage #1: ethylene glycol With hydrogenchloride at 20℃; Stage #2: 4-nitro-3-phenyl-L-alanine at 80 - 85℃; for 2h; | 73.9% |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
1222073-44-0
2-bromo-4-nitro-L-phenylalanine

| Conditions | Yield |
|---|---|
| With tribromo-isocyanuric acid; sulfuric acid optical yield given as %ee; | 73% |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
98-88-4
benzoyl chloride

-
-
69935-12-2
(S)-4-nitro-N-benzoyl-phenylalanine

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 0 - 23℃; | 68% |
| With sodium hydroxide | |
| Stage #1: 4-nitro-3-phenyl-L-alanine; benzoyl chloride With sodium hydroxide at 20℃; for 2h; Cooling with ice; Stage #2: With hydrogenchloride In water pH=5 - 6; |
-
-
949-99-5
4-nitro-3-phenyl-L-alanine

-
-
80994-44-1
{[(tert-butoxycarbonyl)oxy]amino}(phenyl)acetonitrile

-
-
33305-77-0
Boc-Phe(p-NO2)-OH

| Conditions | Yield |
|---|---|
| With triethylamine In 1,4-dioxane; water Ambient temperature; | 67.2% |

| Conditions | Yield |
|---|---|
| In water at 80℃; for 24h; | 67% |
4-Nitro-3-phenyl-L-alanine Chemical Properties
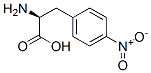
IUPAC Name: (2S)-2-Amino-3-(4-nitrophenyl)propanoic acid
Molecular Formula: C9H10N2O4
Molecular Weight: 210.186700 g/mol
Appearance: white to light yellow crystal powde
Melting Point: 222-223 °C
EINECS: 213-446-8
Density: 1.408g/cm3
Flash Point: 204.2 °C
storage temp.: Store at RT.
Solubility in water: Very soluble
Boiling Point: 414.1 °C at 760mmHg
Product Categories of 4-Nitro-3-phenyl-L-alanine (949-99-5): Amino Acids;Phenylalanine analogs and other aromatic alpha amino acids;Amino ACIDS SERIES;chiral;Phenylalanine [Phe, F]
4-Nitro-3-phenyl-L-alanine Toxicity Data With Reference
Toxicity Information of 4-Nitro-3-phenyl-L-alanine (949-99-5):
1.mouse LD50 intravenous 180mg/kg (180mg/kg)
U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. Vol. NX#03822
4-Nitro-3-phenyl-L-alanine Safety Profile
Safety Information of 4-Nitro-3-phenyl-L-alanine (949-99-5):
Hazard Codes: Xn  ,C
,C  ,F
,F 
Risk Statements: 36/37/38-22-34-11
36/37/38:Irritating to eyes, respiratory system and skin
22:Harmful if swallowed
34:Causes burns
11:Highly Flammable
Safety Statements: 26-36/37/39-22-45-16
26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
36/37/39:Wear suitable protective clothing, gloves and eye/face protection
22:Do not breathe dust
45:In case of accident or if you feel unwell, seek medical advice immediately (show label where possible)
16:Keep away from sources of ignition - No smoking
RIDADR: 2811
RTECS: AY7400000
Hazard Note: Harmful
4-Nitro-3-phenyl-L-alanine Specification
4-Nitro-3-phenyl-L-alanine , its cas register number is 949-99-5. It also can be called P-Nitro-phenylalanine ; P-Nitro-L-phenylalanine ; P-Nitro-2-phenylalanine ; (s)-4-Nitrophenylalanine ; (s)-4-Nitrophenylalanine hydrate ; (s)-2-Amino-3-(4-nitrophenyl)propanoic acid ; Rarechem bk pt 0073 ; and 4-Nitro-L-Phenylalanine Monohydrate .
First aid measures of 4-Nitro-3-phenyl-L-alanine (949-99-5) are as following. If product was ingested ,seek medical assistance immediately. When inhalation, move victim to fresh air. Apply artificial respiration if victim is not breathing. Do not use mouth-to-mouth method if victim ingested or inhaled the substance; induce artificial respiration with the aid of a pocket mask equipped with a one-way valve or other proper respiratory medical device. Administer oxygen if breathing is difficult. While on skin, remove and isolate contaminated clothing and shoes. Immediately flush with running water for at least 20 minutes. For minor skin contact, avoid spreading material on unaffected skin. When in eyes, immediately flush with running water for at least 20 minutes.
Related Products
- 4-Nitro-3-phenyl-L-alanine
- 95-00-1
- 95008-83-6
- 95008-91-6
- 95009-28-2
- 95010-17-6
- 95-01-2
- 95013-42-6
- 950265-42-6
- 95-03-4
- 95034-01-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View