-
Name
4-Nitrobenzoyl chloride
- EINECS 204-517-4
- CAS No. 122-04-3
- Article Data166
- CAS DataBase
- Density 1.453 g/cm3
- Solubility Decomposes
- Melting Point 71-74 °C(lit.)
- Formula C7H4ClNO3
- Boiling Point 277.8 °C at 760 mmHg
- Molecular Weight 185.567
- Flash Point 121.8 °C
- Transport Information UN 3261 8/PG 2
- Appearance yellow needles or powder
- Safety 26-36/37/39-45
- Risk Codes 34
-
Molecular Structure
-
Hazard Symbols
 C
C
- Synonyms p-Nitrobenzoic acid chloride;Benzoyl chloride, p-nitro-;p-Nitrobenzonyl chloride;Paranitrobenzoyl chloride;4-Nitrobenzoic acid chloride;Benzoyl chloride, 4-nitro-;
- PSA 62.89000
- LogP 2.49700
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: (4-nitrophenyl)ethanone With pyridine; disulfur dichloride In chlorobenzene at 20℃; for 2.5h; Stage #2: With sulfuryl dichloride In chlorobenzene at 20 - 132℃; for 21h; Reagent/catalyst; Temperature; | 90% |
| Stage #1: (4-nitrophenyl)ethanone With pyridine; sulfur monochloride In chlorobenzene at 20℃; Stage #2: With thionyl chloride In chlorobenzene at 20 - 132℃; | 71% |
| With pyridine; disulfur dichloride at 70 - 138℃; for 21.5h; | 83 %Spectr. |

| Conditions | Yield |
|---|---|
| With thionyl chloride In ethyl acetate for 2h; Heating; | 100% |
| With thionyl chloride for 1h; Reflux; | 100% |
| With thionyl chloride; N,N-dimethyl-formamide for 5h; Reflux; | 100% |
-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
62-23-7
4-nitro-benzoic acid

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With pyridine; triethylamine In toluene at 0 - 20℃; for 20h; | 90% |

| Conditions | Yield |
|---|---|
| 52.9% |
-
-
15922-01-7
potassium 4-nitrobenzoate

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride; N,N-dimethyl-formamide at 70 - 75℃; for 0.5h; | 98.5% |

| Conditions | Yield |
|---|---|
| In dichloromethane; N,N-dimethyl-formamide; benzene |


| Conditions | Yield |
|---|---|
| With iron(III) chloride; Benzotrichlorid In benzene at 60℃; Rate constant; Mechanism; Thermodynamic data; various concentration ratios; | |
| With trichloroisocyanuric acid In dichloromethane at 20℃; for 120h; Inert atmosphere; | |
| With trichloroisocyanuric acid In dichloromethane at 20℃; for 4h; Inert atmosphere; Irradiation; | |
| With N-chloro-succinimide; thio-xanthene-9-one In acetonitrile at 20℃; for 7h; Irradiation; |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; Nitroethane; chlorine |

| Conditions | Yield |
|---|---|
| With benzene |

| Conditions | Yield |
|---|---|
| With trichloroisocyanuric acid In dichloromethane at 20℃; for 120h; Inert atmosphere; |
-
-
19756-72-0
tert-butyl 4-nitrobenzoate

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride; water In toluene at 100℃; for 16h; Sealed tube; |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In toluene at 100℃; for 12h; |
-
-
19756-72-0
tert-butyl 4-nitrobenzoate

-
A
-
122-04-3
4-nitro-benzoyl chloride

-
B
-
75-65-0
tert-butyl alcohol

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 25℃; Rate constant; Equilibrium constant; |
-
-
52393-62-1, 92248-91-4, 99659-46-8
trans-5-methyl-2-cyclohexen-p-nitrobenzoate

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With water |
-
-
102146-13-4
(p-nitrophenyl)chlorocarbene

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With oxygen In 1,1,2-Trichloro-1,2,2-trifluoroethane at 20℃; Kinetics; Further Variations:; Temperatures; |

| Conditions | Yield |
|---|---|
| With thionyl chloride; diethyl ether |
-
-
98-88-4
benzoyl chloride

-
A
-
122-04-3
4-nitro-benzoyl chloride

-
B
-
121-90-4
m-nitrobenzoic acid chloride

| Conditions | Yield |
|---|---|
| With tetrachloromethane; phosphorus pentaoxide; dinitrogen pentoxide |
-
-
4885-02-3
Dichloromethyl methyl ether

-
-
62-23-7
4-nitro-benzoic acid

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With zinc(II) chloride |

-
-
39184-67-3
3-chloro-3-(p-nitrophenyl)diazirine

-
A
-
555-16-8
4-nitrobenzaldehdye

-
B
-
100-14-1
4-nitrobenzyl chloride

-
C
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With oxygen In 2,2,4-trimethylpentane Product distribution; Mechanism; Ambient temperature; Irradiation; |

-
-
99-99-0
1-methyl-4-nitrobenzene

-
A
-
122-01-0
4-chloro-benzoyl chloride

-
B
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| at 180℃; Einleiten von Chlor; |

-
-
99-99-0
1-methyl-4-nitrobenzene

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
122-01-0
4-chloro-benzoyl chloride

-
B
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| at 180℃; Einleiten von Chlor; |

-
-
56-23-5
tetrachloromethane

-
-
98-88-4
benzoyl chloride

-
A
-
122-04-3
4-nitro-benzoyl chloride

-
B
-
121-90-4
m-nitrobenzoic acid chloride

| Conditions | Yield |
|---|---|
| at -10℃; |


| Conditions | Yield |
|---|---|
| With thionyl chloride In ethyl acetate for 2h; Heating; | 100% |
| With thionyl chloride for 1h; Reflux; | 100% |
| With thionyl chloride; N,N-dimethyl-formamide for 5h; Reflux; | 100% |
-
-
15922-01-7
potassium 4-nitrobenzoate

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride; N,N-dimethyl-formamide at 70 - 75℃; for 0.5h; | 98.5% |

| Conditions | Yield |
|---|---|
| Stage #1: (4-nitrophenyl)ethanone With pyridine; disulfur dichloride In chlorobenzene at 20℃; for 2.5h; Stage #2: With sulfuryl dichloride In chlorobenzene at 20 - 132℃; for 21h; Reagent/catalyst; Temperature; | 90% |
| Stage #1: (4-nitrophenyl)ethanone With pyridine; sulfur monochloride In chlorobenzene at 20℃; Stage #2: With thionyl chloride In chlorobenzene at 20 - 132℃; | 71% |
| With pyridine; disulfur dichloride at 70 - 138℃; for 21.5h; | 83 %Spectr. |
-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
62-23-7
4-nitro-benzoic acid

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With pyridine; triethylamine In toluene at 0 - 20℃; for 20h; | 90% |

| Conditions | Yield |
|---|---|
| 52.9% |

| Conditions | Yield |
|---|---|
| With thionyl chloride; diethyl ether |
-
-
98-88-4
benzoyl chloride

-
A
-
122-04-3
4-nitro-benzoyl chloride

-
B
-
121-90-4
m-nitrobenzoic acid chloride

| Conditions | Yield |
|---|---|
| With tetrachloromethane; phosphorus pentaoxide; dinitrogen pentoxide |

| Conditions | Yield |
|---|---|
| With hydrogenchloride; Nitroethane; chlorine |
-
-
4885-02-3
Dichloromethyl methyl ether

-
-
62-23-7
4-nitro-benzoic acid

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With zinc(II) chloride |
-
-
19756-72-0
tert-butyl 4-nitrobenzoate

-
A
-
122-04-3
4-nitro-benzoyl chloride

-
B
-
75-65-0
tert-butyl alcohol

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 25℃; Rate constant; Equilibrium constant; |
-
-
52393-62-1, 92248-91-4, 99659-46-8
trans-5-methyl-2-cyclohexen-p-nitrobenzoate

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With water |

| Conditions | Yield |
|---|---|
| With iron(III) chloride; Benzotrichlorid In benzene at 60℃; Rate constant; Mechanism; Thermodynamic data; various concentration ratios; | |
| With trichloroisocyanuric acid In dichloromethane at 20℃; for 120h; Inert atmosphere; | |
| With trichloroisocyanuric acid In dichloromethane at 20℃; for 4h; Inert atmosphere; Irradiation; | |
| With N-chloro-succinimide; thio-xanthene-9-one In acetonitrile at 20℃; for 7h; Irradiation; |
-
-
39184-67-3
3-chloro-3-(p-nitrophenyl)diazirine

-
A
-
555-16-8
4-nitrobenzaldehdye

-
B
-
100-14-1
4-nitrobenzyl chloride

-
C
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With oxygen In 2,2,4-trimethylpentane Product distribution; Mechanism; Ambient temperature; Irradiation; |


| Conditions | Yield |
|---|---|
| With benzene |
-
-
56-23-5
tetrachloromethane

-
-
98-88-4
benzoyl chloride

-
A
-
122-04-3
4-nitro-benzoyl chloride

-
B
-
121-90-4
m-nitrobenzoic acid chloride

| Conditions | Yield |
|---|---|
| at -10℃; |


-
-
99-99-0
1-methyl-4-nitrobenzene

-
-
10026-13-8, 874483-75-7
phosphorus pentachloride

-
A
-
122-01-0
4-chloro-benzoyl chloride

-
B
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| at 180℃; Einleiten von Chlor; |

-
-
99-99-0
1-methyl-4-nitrobenzene

-
A
-
122-01-0
4-chloro-benzoyl chloride

-
B
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| at 180℃; Einleiten von Chlor; |

-
-
102146-13-4
(p-nitrophenyl)chlorocarbene

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With oxygen In 1,1,2-Trichloro-1,2,2-trifluoroethane at 20℃; Kinetics; Further Variations:; Temperatures; |

| Conditions | Yield |
|---|---|
| In dichloromethane; N,N-dimethyl-formamide; benzene |


| Conditions | Yield |
|---|---|
| With trichloroisocyanuric acid In dichloromethane at 20℃; for 120h; Inert atmosphere; |
-
-
19756-72-0
tert-butyl 4-nitrobenzoate

-
-
122-04-3
4-nitro-benzoyl chloride

| Conditions | Yield |
|---|---|
| With thionyl chloride; water In toluene at 100℃; for 16h; Sealed tube; |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); 4,5-bis(diphenylphos4,5-bis(diphenylphosphino)-9,9-dimethylxanthenephino)-9,9-dimethylxanthene In toluene at 100℃; for 12h; |

| Conditions | Yield |
|---|---|
| With triethylamine In benzene | 100% |
| With triethylamine In chloroform | 78% |

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 2.5h; | 100% |
| With pyridine Reflux; | 95% |
| With pyridine at 20℃; Inert atmosphere; Reflux; | 91% |

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 20℃; for 96h; Inert atmosphere; | 100% |
| With dmap In dichloromethane at 20℃; for 24h; | 92% |
| With dmap; triethylamine In dichloromethane at 0 - 20℃; for 1h; Inert atmosphere; | 82% |
-
-
122-04-3
4-nitro-benzoyl chloride

-
-
108-46-3
recorcinol

-
-
187264-29-5
3-(4-nitrobenzoyloxy)phenyl 4-nitrobenzoate

| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran at 20℃; for 24h; | 100% |
| With tetralin at 160℃; | |
| With pyridine at 100℃; | |
| With pyridine In toluene Heating; |
-
-
487-79-6
(-)-kainic acid

-
-
122-04-3
4-nitro-benzoyl chloride

-
-
101575-64-8
[(3S)-2t-carboxy-4c-isopropenyl-1-(4-nitro-benzoyl)-pyrrolidin-3r-yl]-acetic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 0 - 20℃; for 20h; Inert atmosphere; | 100% |
| With water; sodium carbonate |
-
-
3143-02-0
3-hydroxymethyl-3-methyloxethane

-
-
122-04-3
4-nitro-benzoyl chloride

-
-
140635-77-4
(3-methyloxetan-3-yl)methyl 4-nitrobenzoate

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 20℃; for 0.333333h; | 100% |
-
-
6234-01-1
L-glutamic acid 5-tert-butyl 1-methyl ester hydrochloride

-
-
122-04-3
4-nitro-benzoyl chloride

-
-
95485-03-3
α-methyl γ-tert-butyl N-(p-nitrobenzoyl)-L-glutamate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane for 18h; | 100% |

| Conditions | Yield |
|---|---|
| With bis(η3-allyl-μ-chloropalladium(II)) In N,N,N,N,N,N-hexamethylphosphoric triamide at 20℃; for 0.166667h; Mechanism; | 100% |
| With bis(η3-allyl-μ-chloropalladium(II)) In N,N,N,N,N,N-hexamethylphosphoric triamide at 20℃; for 0.166667h; | 100% |
| With benzoylchlorobis(triphenylphosphine)palladium(II) In chloroform at 65℃; for 24h; | 95% |
| With N,N,N,N,N,N-hexamethylphosphoric triamide; poly-γ-(diphenylphosphino)propylsiloxane palladium(0) at 65℃; for 20h; | 83% |
-
-
122-04-3
4-nitro-benzoyl chloride

-
-
140934-56-1
1-(2-oxazolinyl)indole

| Conditions | Yield |
|---|---|
| In benzene for 12h; Heating; | 100% |
-
-
122-04-3
4-nitro-benzoyl chloride

-
-
73891-06-2
2,3,6-tridesoxy-3-(trifluoroacetamido)-L-ribo-hex-1-enitol

-
-
73891-07-3
2,3,6-tridesoxy-4-O-p-nitrobenzoyl-3-(trifluoroacetamido)-L-ribo-hex-1-enitol

| Conditions | Yield |
|---|---|
| In pyridine at 20℃; for 16h; | 100% |
-
-
122-04-3
4-nitro-benzoyl chloride

-
-
101024-28-6
methyl 5-O-benzoyl-3-O-benzyl-β-D-ribofuranoside

-
-
101024-29-7
methyl 5-O-benzoyl-3-O-benzyl-2-O-(p-nitrobenzoyl)-β-D-ribofuranoside

| Conditions | Yield |
|---|---|
| With pyridine for 18h; Ambient temperature; | 100% |
-
-
122-04-3
4-nitro-benzoyl chloride

-
-
101024-26-4
methyl 5-O-benzoyl-2-O-benzyl-β-D-ribofuranoside

-
-
101024-27-5
methyl 5-O-benzoyl-2-O-benzyl-3-O-(p-nitrobenzoyl)-β-D-ribofuranoside

| Conditions | Yield |
|---|---|
| With pyridine for 18h; Ambient temperature; | 100% |
4-Nitrobenzoyl chloride Consensus Reports
4-Nitrobenzoyl chloride Specification
The IUPAC name of this chemical is 4-Nitrobenzoyl chloride. With the CAS registry number 122-04-3 and EINECS registry number 204-517-4, it is also named as p-nitrobenzonyl chloride. In addition, the molecular formula is C7H4ClNO3. What's more, it is a kind of yellow needles or powder and sensitive with moisture. Besides, it is incompatible with water, alcohols, strong oxidizing agents and strong bases.
Physical properties about this chemical are: (1)ACD/LogP: 2.17; (2)ACD/LogD (pH 5.5): 2.17; (3)ACD/LogD (pH 7.4): 2.17; (4)ACD/BCF (pH 5.5): 26.28; (5)ACD/BCF (pH 7.4): 26.28; (6)ACD/KOC (pH 5.5): 361.21; (7)ACD/KOC (pH 7.4): 361.21; (8)#H bond acceptors: 4; (9)#Freely Rotating Bonds: 2; (10)Polar Surface Area: 62.89 Å2; (11)Index of Refraction: 1.589; (12)Molar Refractivity: 43.04 cm3; (13)Molar Volume: 127.6 cm3; (14)Polarizability: 17.06 ×10-24cm3; (15)Surface Tension: 54.2 dyne/cm; (16)Density: 1.453 g/cm3; (17)Flash Point: 121.8 °C; (18)Enthalpy of Vaporization: 51.64 kJ/mol; (19)Boiling Point: 277.8 °C at 760 mmHg; (20)Vapour Pressure: 0.00442 mmHg at 25°C.
Preparation of 4-Nitrobenzoyl chloride: At first, add p-nitrobenzoic acid and phosphorus pentylchloride into the oil bath with heating. After the reaction, you can recycle trichlorooxyphosphorus through vacuum distillation. And then via vacuum distillation you can obtain the crude of this product. Next, melt the crude into liquld and then add carbon tetrachloride. You get the desired product through filtration, cooling and crystallization.
![]()
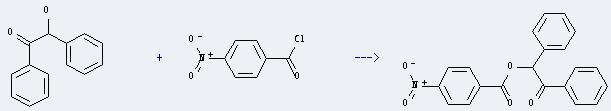
Uses of 4-Nitrobenzoyl chloride: it can react with 2-hydroxy-1,2-diphenyl-ethanone to get a-(4-nitro-benzoyloxy)-deoxybenzoin. This reaction will need reagent pyridine. The reaction time is 15 minutes by heating. The yield is about 62%.
When you are using this chemical, please be cautious about it as the following:
This chemical can cause burns. During using it, wear suitable protective clothing, gloves and eye/face protection. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. And in case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.).
You can still convert the following datas into molecular structure:
(1)SMILES: O=[N+]([O-])c1ccc(C(Cl)=O)cc1
(2)Std.InChI: InChI=1S/C7H4ClNO3/c8-7(10)5-1-3-6(4-2-5)9(11)12/h1-4H
(3)Std.InChIKey: SKDHHIUENRGTHK-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | oral | 3440mg/kg (3440mg/kg) | BEHAVIORAL: MUSCLE WEAKNESS BEHAVIORAL: ATAXIA BEHAVIORAL: MUSCLE CONTRACTION OR SPASTICITY) | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 28(9), Pg. 47, 1984. |
| rabbit | LD50 | oral | 4750mg/kg (4750mg/kg) | BEHAVIORAL: MUSCLE WEAKNESS BEHAVIORAL: ATAXIA BEHAVIORAL: MUSCLE CONTRACTION OR SPASTICITY) | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 28(9), Pg. 47, 1984. |
| rat | LD50 | oral | 5600mg/kg (5600mg/kg) | BEHAVIORAL: MUSCLE WEAKNESS BEHAVIORAL: ATAXIA BEHAVIORAL: MUSCLE CONTRACTION OR SPASTICITY) | Gigiena Truda i Professional'nye Zabolevaniya. Labor Hygiene and Occupational Diseases. Vol. 28(9), Pg. 47, 1984. |
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View