-
Name
4-Propoxybenzoic acid
- EINECS
- CAS No. 5438-19-7
- Article Data151
- CAS DataBase
- Density 1.133 g/cm3
- Solubility insoluble in water
- Melting Point 144-146 °C(lit.)
- Formula C10H12O3
- Boiling Point 304.9 °C at 760 mmHg
- Molecular Weight 180.203
- Flash Point 119.3 °C
- Transport Information
- Appearance White to light yellow crystal powder
- Safety 24/25
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Benzoicacid, p-propoxy- (6CI,7CI,8CI);NSC 16632;p-(n-Propoxy)benzoic acid;p-Propoxybenzoic acid;
- PSA 46.53000
- LogP 2.17360
Synthetic route
-
-
115478-59-6
methyl 4-propoxy benzoate

-
-
5438-19-7
4-propoxybenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: methyl 4-propoxy benzoate With sodium hydroxide In methanol at 60 - 65℃; for 3h; Stage #2: With hydrogenchloride In methanol; water | 98.9% |
| With alkali |

| Conditions | Yield |
|---|---|
| Stage #1: 4-propoxybenzaldehyde In water; acetonitrile at 5℃; for 0.25h; Stage #2: With potassium permanganate In water; acetonitrile at 20℃; for 2h; | 89% |
| With potassium permanganate; sodium dihydrogenphosphate; ammonium cerium (IV) nitrate In water at 20℃; for 3h; |
-
-
27914-60-9
4-allyloxybenzoic acid

-
A
-
5438-19-7
4-propoxybenzoic acid

-
B
-
99-96-7
4-hydroxy-benzoic acid

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol at 20℃; for 10h; | A 3% B 82% |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol Heating; | 80% |
| With potassium hydroxide In methanol | |
| With potassium hydroxide In methanol Reflux; | |
| In methanol |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol; water for 15h; Reflux; | 70% |
| With potassium hydroxide | |
| With potassium hydroxide; Aliquat 336 1.) 80 deg C, 24 h; 2.) ethanol, 80 deg C, 24 h; Multistep reaction; |

| Conditions | Yield |
|---|---|
| With carbon disulfide; aluminium trichloride durch Verseifen des entstandenen Amids mit NaNO2 in verd. Schwefelsaeure; |
-
-
100256-94-8
4-propoxy-benzoic acid ethyl ester

-
-
5438-19-7
4-propoxybenzoic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide | |
| With potassium hydroxide | |
| With potassium hydroxide In ethanol Hydrolysis; | |
| Stage #1: 4-propoxy-benzoic acid ethyl ester With sodium hydroxide In ethanol at 20℃; for 3h; Stage #2: With hydrogenchloride; water In ethanol pH=1; | |
| With ethanol; sodium hydroxide |

| Conditions | Yield |
|---|---|
| With potassium carbonate; butanone Erwaermen des Reaktionsprodukts mit methanol. Kalilauge; | |
| With hydrogenchloride; potassium hydroxide; sodium hydroxide In ethanol; water; dimethyl sulfoxide | |
| With potassium carbonate In acetone for 48h; Reflux; | 2.20 g |
-
-
106-94-5
propyl bromide

-
-
16782-08-4
sel de potassium de l'acide p-hydroxybenzoique

-
-
5438-19-7
4-propoxybenzoic acid

| Conditions | Yield |
|---|---|
| In ethanol Heating; |

| Conditions | Yield |
|---|---|
| In chloroform-d1 Equilibrium constant; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: NaOEt / 3 h / Heating 2: KOH / ethanol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: conc. H2SO4 / Heating 2: NaOEt / 3 h / Heating 3: KOH / ethanol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 1.) NaH / 1.) THF, 0 deg C, 2.) reflux, 4 h View Scheme | |
| Multi-step reaction with 2 steps 1: sodium methylate 2: alkali View Scheme | |
| Multi-step reaction with 2 steps 1: potassium hydroxide / methanol / Reflux 2: potassium hydroxide / ethanol; water / 2 h / Reflux View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium ethylate; ethanol 2: ethanolic KOH-solution View Scheme | |
| Multi-step reaction with 2 steps 1: sodium ethylate; ethanol 2: aq. NaOH solution View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / N,N-dimethyl-formamide 2: sodium hydroxide; ethanol View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: 4-hydroxy-benzoic acid; 1-halidopropane With potassium hydroxide In methanol Reflux; Stage #2: With water; potassium hydroxide In methanol Reflux; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol | |
| With potassium hydroxide In methanol Reflux; | |
| With potassium hydroxide In ethanol for 8h; Reflux; | |
| In methanol |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol Reflux; | |
| With potassium hydroxide In methanol | |
| Multi-step reaction with 3 steps 1: sulfuric acid / 1 h / Reflux 2: potassium hydroxide / methanol / Reflux 3: potassium hydroxide / ethanol; water / 2 h / Reflux View Scheme |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol Reflux; | |
| With potassium hydroxide In methanol |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol Reflux; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol Reflux; |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In methanol |

| Conditions | Yield |
|---|---|
| With dmap; sodium carbonate In N,N-dimethyl-formamide for 12h; Reflux; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: potassium carbonate / N,N-dimethyl-formamide / 80 °C 2: potassium permanganate; sodium dihydrogenphosphate; ammonium cerium (IV) nitrate / water / 3 h / 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: potassium carbonate / N,N-dimethyl-formamide / 8 h / 80 °C 2.1: sodium phosphate / acetonitrile; water / 0.25 h / 5 °C 2.2: 2 h / 20 °C View Scheme |
-
-
5438-19-7
4-propoxybenzoic acid

| Conditions | Yield |
|---|---|
| With N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate In dichloromethane for 16h; | 92% |

| Conditions | Yield |
|---|---|
| With iron(III)-acetylacetonate; di-tert-butyl peroxide at 120℃; for 24h; Schlenk technique; | 91% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-propoxybenzoic acid; meta-hydroxybenzaldehyde With dmap In N,N-dimethyl-formamide at 20℃; for 0.25h; Stage #2: With dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 20℃; | 90.1% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol acid-compd. and 1 equiv. of NaOH were stirred in MeOH at room temp. for 30 min, 1/3 equiv. of metal-nitrate was added in MeOH, 1 d, stirring forfurther 12 h; ppt. was filtered off and washed with MeOH, elem. anal.; | 90% |

-
-
5438-19-7
4-propoxybenzoic acid

-
-
123-08-0
4-hydroxy-benzaldehyde

-
-
56800-28-3
4-formylphenyl 4-propyloxybenzoate

| Conditions | Yield |
|---|---|
| Stage #1: 4-propoxybenzoic acid; 4-hydroxy-benzaldehyde With dmap In N,N-dimethyl-formamide at 20℃; for 0.25h; Stage #2: With dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 20℃; | 87.1% |
| In chloroform | |
| With dicyclohexyl-carbodiimide In dichloromethane at 20℃; |
-
-
5438-19-7
4-propoxybenzoic acid

-
-
848478-61-5
1-(4-hydroxyphenyl)-3-hydroxyoctan-1-one

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 23h; | 87% |
-
-
102-71-6
triethanolamine

-
-
5438-19-7
4-propoxybenzoic acid

-
-
1440507-27-6
tris[2-(4-propyloxybenzoyloxy)ethyl]amine

| Conditions | Yield |
|---|---|
| Stage #1: triethanolamine; 4-propoxybenzoic acid In chloroform for 0.166667h; Stage #2: With dmap; dicyclohexyl-carbodiimide In chloroform for 12h; | 85% |
-
-
118712-54-2
glycidyl p-toluenesulfonate

-
-
5438-19-7
4-propoxybenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-propoxybenzoic acid With potassium hydroxide In methanol; isopropyl alcohol at 20℃; for 1h; Stage #2: glycidyl p-toluenesulfonate In N,N-dimethyl-formamide at 70℃; for 7h; | 83% |
-
-
5438-19-7
4-propoxybenzoic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 16h; | 82% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 16h; | 82% |
-
-
5438-19-7
4-propoxybenzoic acid

-
-
848478-63-7
1-(4-hydroxyphenyl)-3-chlorooctan-1-one

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; | 81% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol acid-compd. and 1 equiv. of NaOH were stirred in MeOH at room temp. for 30 min, 1/3 equiv. of metal-nitrate was added in MeOH, 1 d, stirring forfurther 12 h; ppt. was filtered off and washed with MeOH, elem. anal.; | 81% |

-
-
5438-19-7
4-propoxybenzoic acid

-
-
1440548-48-0
(E)-hex-1-enyl 4-propoxybenzoate

| Conditions | Yield |
|---|---|
| With dmap; oxygen; copper(I) bromide In acetonitrile at 60℃; for 24h; Chan-Lam Coupling; Molecular sieve; stereospecific reaction; | 81% |

| Conditions | Yield |
|---|---|
| Stage #1: 5-methyl-2-hydroxyacetophenone; 4-propoxybenzoic acid With pyridine at 20℃; for 0.166667h; Stage #2: With trichlorophosphate at 0 - 10℃; for 3h; | 81% |

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 15h; | 81% |
-
-
5438-19-7
4-propoxybenzoic acid

-
-
115914-43-7
bis{1,9-(4-hydroxyphenyl)nonane}

| Conditions | Yield |
|---|---|
| With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In dichloromethane Steglich Esterification; | 80.4% |
-
-
914784-11-5
3-(4-hydroxyphenyl)-5-pentylisoxazole

-
-
5438-19-7
4-propoxybenzoic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane | 79% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-propoxybenzoic acid With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In tetrahydrofuran at 20℃; for 0.25h; Stage #2: 2-(3-hydroxyphenyl)-1,3-benzoxazole In tetrahydrofuran at 20℃; for 3h; | 79% |

| Conditions | Yield |
|---|---|
| Stage #1: 4-propoxybenzoic acid; 1-chloro-3-hydroxypropane With dmap In N,N-dimethyl-formamide at 20℃; for 0.166667h; Steglich Esterification; Stage #2: With dicyclohexyl-carbodiimide In N,N-dimethyl-formamide at 20℃; for 6h; Steglich Esterification; | 76.2% |
-
-
443682-63-1
(R)-4-hydroxy-1-(3-ethyl-1-methylpentyloxycarbonyl)benzene

-
-
5438-19-7
4-propoxybenzoic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 20℃; for 12h; | 76% |
-
-
5438-19-7
4-propoxybenzoic acid

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane at 25℃; for 168h; | 74% |
-
-
5438-19-7
4-propoxybenzoic acid

-
-
77103-47-0
ethyl (4-hydroxybenzoyl)acetate

-
-
1462352-06-2
ethyl 3-[4-(4'-propoxy)benzoyloxyphenyl]-3-oxo-propanoate

| Conditions | Yield |
|---|---|
| With dmap; dicyclohexyl-carbodiimide In dichloromethane for 25h; | 73% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In methanol acid-compd. and 1 equiv. of NaOH were stirred in MeOH at room temp. for 30 min, 1/3 equiv. of metal-nitrate was added in MeOH, 1 d, stirring forfurther 12 h; ppt. was filtered off and washed with MeOH, elem. anal.; | 72% |

-
-
5438-19-7
4-propoxybenzoic acid

-
-
551-93-9
2-aminoacetophenone

-
-
1319210-02-0
N-(2-acetylphenyl)-4-propoxybenzamide

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran at 70℃; for 3h; | 72% |
-
-
5438-19-7
4-propoxybenzoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 4-propoxybenzoic acid With benzotriazol-1-ol; triethylamine In dichloromethane; 1,2-dichloro-ethane at 0 - 5℃; Stage #2: (E)-7-hydroxy-3-(3-(4-hydroxyphenyl)acryloyl)–2H-chromen-2-one In dichloromethane at 0 - 5℃; for 0.5h; Stage #3: In dichloromethane at 20℃; for 20h; | 72% |
-
-
5438-19-7
4-propoxybenzoic acid

-
-
1017029-32-1
N-(2-aminoethyl)benzofuran-2-carboxamide

-
-
1397262-86-0
N-(2-(4-propoxybenzamido)ethyl)benzofuran-2-carboxamide

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 20℃; for 20h; Inert atmosphere; | 71% |
4-Propoxybenzoic acid Specification
The Benzoic acid,4-propoxy-, with the CAS registry number 5438-19-7, has the systmatic name of 4-propoxybenzoic acid. And the molecular formula of this chemical is C10H12O3. It is a kind of white to light yellow crystal powder, and belongs to the following product categories: Aromatic Carboxylic Acids, Amides, Anilides, Anhydrides & Salts; Benzoic acid; Organic acids; Acids & Esters; Anisoles, Alkyloxy Compounds & Phenylacetates; Benzoic Acids (Building Blocks for Liquid Crystals); Building Blocks for Liquid Crystals; Functional Materials; Building blockCarbonyl Compounds; C10; Carboxylic Acids; Liquid Crystals; Organic Electronics and Photonics. What's more, it is used as the intermediates of liquid crystals.
The physical properties of Benzoic acid,4-propoxy- are as following: (1)ACD/LogP: 3.02; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 1.97; (4)ACD/LogD (pH 7.4): 0.31; (5)ACD/BCF (pH 5.5): 10.35; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 93.1; (8)ACD/KOC (pH 7.4): 2.02; (9)#H bond acceptors: 3; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 4; (12)Polar Surface Area: 35.53 Å2; (13)Index of Refraction: 1.53; (14)Molar Refractivity: 49.12 cm3; (15)Molar Volume: 158.9 cm3; (16)Polarizability: 19.47×10-24cm3; (17)Surface Tension: 42.3 dyne/cm; (18)Density: 1.133 g/cm3; (19)Flash Point: 119.3 °C; (20)Enthalpy of Vaporization: 57.58 kJ/mol; (21)Boiling Point: 304.9 °C at 760 mmHg; (22)Vapour Pressure: 0.000372 mmHg at 25°C.
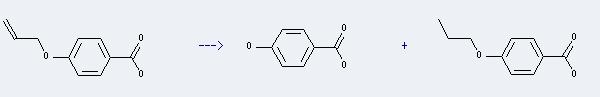
Preparation of Benzoic acid,4-propoxy-: This chemical can be prepared by 4-allyloxy-benzoic acid, and the other product is 4-hydroxy-benzoic acid. The reaction will need reagent KOH, and the solvent methanol. The reaction time is 10 hours with temperature of 20°C, and the yield is about 82%.
You should be cautious while dealing with this chemical. It irritates eyes, respiratory system and skin. Therefore, you had better take avoid contact with skin and eyes.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)c1ccc(OCCC)cc1
(2)InChI: InChI=1/C10H12O3/c1-2-7-13-9-5-3-8(4-6-9)10(11)12/h3-6H,2,7H2,1H3,(H,11,12)
(3)InChIKey: GDFUWFOCYZZGQU-UHFFFAOYAE
Related Products
- 4-Propoxybenzoic acid
- 543-82-8
- 5438-36-8
- 54384-06-4
- 5438-41-5
- 5438-68-6
- 5438-70-0
- 54389-65-0
- 54389-67-2
- 54389-98-9
- 543906-09-8
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View