-
Name
5-Hydroxymethyluracil
- EINECS 224-636-5
- CAS No. 4433-40-3
- Article Data62
- CAS DataBase
- Density 1.401 g/cm3
- Solubility Soluble in water. (50 g/L ) at 20°C, DMSO, dimethyl formamide or 100% ethanol.
- Melting Point >300 °C
- Formula C5H6N2O3
- Boiling Point 530.4 °C at 760 mmHg
- Molecular Weight 142.114
- Flash Point 274.6 °C
- Transport Information
- Appearance White Powder
- Safety 22-24/25
- Risk Codes
-
Molecular Structure
- Hazard Symbols Xi
- Synonyms Uracil,5-(hydroxymethyl)- (6CI,7CI,8CI);5-(Hydroxymethyl)-1,3-dihydropyrimidine-2,4-dione;5-(Hydroxymethyl)-2,4(1H,3H)-pyrimidinedione;5-(Hydroxymethyl)uracil;NSC 20901;a-Hydroxythymine;
- PSA 86.47000
- LogP -0.61990
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water at 50 - 52℃; for 68h; | 100% |
| With potassium hydroxide for 0.05h; microwave irradiation; | 98% |
| With potassium hydroxide In water at 0 - 55℃; for 36h; | 98% |
-
-
3590-48-5
5-chloromethyluracil

-
-
107-21-1
ethylene glycol

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
88459-62-5
5-<(2-hydroxyethoxy)methyl>uracil

| Conditions | Yield |
|---|---|
| With pyridine In N,N-dimethyl-formamide for 24h; Title compound not separated from byproducts; | A n/a B 46% |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
1195-08-0
2,4-dioxo-1,2,3,4-tetrahydropyrimidine-5-carbaldehyde

| Conditions | Yield |
|---|---|
| With sodium persulfate In water at 85 - 90℃; for 7h; | A 5% B 41% |
| With sodium thiosulfate In water at 85 - 90℃; for 7h; | A 5% B 41% |
| With sodium persulfate In water at 85℃; for 7h; | A 0.05 mmol B 0.41 mmol |
| With Na2S2O8 buffer pH=7.0 at 70℃; for 4h; | A 20 % Chromat. B 7 % Chromat. |
| With sodium persulfate In water at 85℃; for 7h; Product distribution; Mechanism; other N-methylated thymines and uracils; | A 0.05 mmol B 0.41 mmol |
-
-
89179-86-2
5-(aminomethyl)-2,4(1H,3H)-pyrimidinedione

-
-
4433-40-3
5-hydroxymethyl uracil

| Conditions | Yield |
|---|---|
| With barium nitrite anschliessend mit wss.H2SO4; |
-
-
94820-38-9
5-(1H-[1,2,4]Triazol-3-ylsulfanylmethyl)-1H-pyrimidine-2,4-dione

-
A
-
4433-40-3
5-hydroxymethyl uracil

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84345-71-1
5-(Benzooxazol-2-ylsulfanylmethyl)-1H-pyrimidine-2,4-dione

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
75593-45-2
Benzooxazole-2-thiol anion

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84345-81-3
5-(Benzooxazol-2-ylselanylmethyl)-1H-pyrimidine-2,4-dione

-
A
-
4433-40-3
5-hydroxymethyl uracil

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84345-70-0
5-(Benzothiazol-2-ylsulfanylmethyl)-1H-pyrimidine-2,4-dione

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
45769-89-9
Benzothiazole-2-thiol anion

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84345-75-5
5-(Benzothiazol-2-ylselanylmethyl)-1H-pyrimidine-2,4-dione

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
111686-10-3
Benzothiazole-2-selenol anion

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84389-05-9
5-(9H-Purin-6-ylsulfanylmethyl)-1H-pyrimidine-2,4-dione

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
33426-51-6
6-Thiopurin Anion

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84345-64-2
5-(5-Phenyl-[1,3,4]oxadiazol-2-ylsulfanylmethyl)-1H-pyrimidine-2,4-dione

-
A
-
4433-40-3
5-hydroxymethyl uracil

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84345-68-6
5-<<<5-(2-thienyl)-1,3,4-oxadiazol-2-yl>thio>methyl>uracil

-
A
-
4433-40-3
5-hydroxymethyl uracil

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84345-69-7
5-(5-Thiophen-2-yl-2-thioxo-[1,3,4]oxadiazol-3-ylmethyl)-1H-pyrimidine-2,4-dione

-
A
-
4433-40-3
5-hydroxymethyl uracil

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84345-65-3
5-(5-Phenyl-2-thioxo-[1,3,4]oxadiazol-3-ylmethyl)-1H-pyrimidine-2,4-dione

-
A
-
4433-40-3
5-hydroxymethyl uracil

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84345-62-0
5-<(1-phenyl-5-thioxo-2-tetrazolin-4-yl)methyl>uracil

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
53079-79-1
1-Phenyl-1H-tetrazole-5-thiol anion

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84345-57-3
5-<<(1-phenyl-1,2,3,4-tetrazol-5-yl)thio>methyl>uracil

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
53079-79-1
1-Phenyl-1H-tetrazole-5-thiol anion

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; Mechanism; Thermodynamic data; other α-substituted thymines; ΔH(excit.), ΔS(excit.), var. pH; |
-
-
84345-76-6
5-<<(1-phenyl-1,2,3,4-tetrazol-5-yl)seleno>methyl>uracil

-
A
-
4433-40-3
5-hydroxymethyl uracil

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
84345-73-3
5-<<<5-(p-toluidino)-1,3,4-selenadiazolo-2-yl>seleno>methyl>uracil

-
A
-
4433-40-3
5-hydroxymethyl uracil

| Conditions | Yield |
|---|---|
| With potassium hydroxide In water; acetonitrile at 22℃; Rate constant; |
-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
2417-22-3
5-methylbarbituric acid

-
C
-
2943-56-8
5,6-dihydro-5,6-dihydroxythymine

-
D
-
27284-91-9
N1-formyl-N2-pyruvylurea

-
E
-
13514-92-6
6-Hydroxydihydrothymine

| Conditions | Yield |
|---|---|
| With air; water Quantum yield; Irradiation; var. irradiation: γ-radiolysis; |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
2417-22-3
5-methylbarbituric acid

-
C
-
13514-92-6
6-Hydroxydihydrothymine

-
D
-
20433-38-9
5,6-dihydroxyuracil

| Conditions | Yield |
|---|---|
| With Saline; dinitrogen monoxide In water Ambient temperature; Irradiation; Further byproducts given; |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
1123-21-3
6-hydroxy-5,6-dihydrothymine

-
C
-
2943-56-8
5,6-dihydro-5,6-dihydroxythymine

-
D
-
13514-92-6
6-Hydroxydihydrothymine

| Conditions | Yield |
|---|---|
| With 1H-4(5)-nitroimidazole In water Irradiation; pH=7.0+/-0.1; Further byproducts given; |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
27284-91-9
N1-formyl-N2-pyruvylurea

-
C
-
20433-38-9
5,6-dihydroxyuracil

| Conditions | Yield |
|---|---|
| With tempol; Saline; dinitrogen monoxide In water Ambient temperature; Irradiation; |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
2417-22-3
5-methylbarbituric acid

-
C
-
1123-21-3
6-hydroxy-5,6-dihydrothymine

-
D
-
13514-92-6
6-Hydroxydihydrothymine

-
E
-
696-04-8
5,6-dihydrothymine

| Conditions | Yield |
|---|---|
| nickel(II) sulphate Product distribution; Irradiation; degradation in the presence and absence of different Ni(II) compounds; different atmospheres; |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
2417-22-3
5-methylbarbituric acid

-
C
-
1123-21-3
6-hydroxy-5,6-dihydrothymine

-
-
1124-84-1, 1431-06-7, 2943-56-8, 57908-05-1, 57908-06-2, 57968-48-6, 57968-49-7, 57968-50-0, 58000-49-0
cis-5,6-dihydroxy-5,6-dehydrothymine

-
E
-
13514-92-6
6-Hydroxydihydrothymine

-
F
-
696-04-8
5,6-dihydrothymine

| Conditions | Yield |
|---|---|
| With ammonium ferric sulfate In water Product distribution; γ-radiolysis, absence of NH4Fe(SO4)2, in presence of NH4Fe(SO4)2 and t-butyl alcohol; |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
2417-22-3
5-methylbarbituric acid

-
C
-
27284-91-9
N1-formyl-N2-pyruvylurea

-
D
-
13514-92-6
6-Hydroxydihydrothymine

-
E
-
20433-38-9
5,6-dihydroxyuracil

-
F
-
696-04-8
5,6-dihydrothymine

| Conditions | Yield |
|---|---|
| With Saline In water Product distribution; Mechanism; Ambient temperature; Irradiation; promotion effect of addition of 2,2,6,6-tetramethylpiperidine-1-oxyls, influence of various saturation gases; |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
1123-21-3
6-hydroxy-5,6-dihydrothymine

-
C
-
2943-56-8
5,6-dihydro-5,6-dihydroxythymine

-
D
-
13514-92-6
6-Hydroxydihydrothymine

-
E
-
696-04-8
5,6-dihydrothymine

| Conditions | Yield |
|---|---|
| With (60)Co γ-ray irradiation; 1H-4(5)-nitroimidazole In water Mechanism; Irradiation; var. nitro compounds, pH=7.0+/-0.1; |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
1123-21-3
6-hydroxy-5,6-dihydrothymine

-
C
-
13514-92-6
6-Hydroxydihydrothymine

-
D
-
696-04-8
5,6-dihydrothymine

| Conditions | Yield |
|---|---|
| With 1H-4(5)-nitroimidazole In water Irradiation; pH=7.0+/-0.1; Further byproducts given; |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
2943-56-8
5,6-dihydro-5,6-dihydroxythymine

-
C
-
27284-91-9
N1-formyl-N2-pyruvylurea

-
D
-
13514-92-6
6-Hydroxydihydrothymine

-
E
-
696-04-8
5,6-dihydrothymine

| Conditions | Yield |
|---|---|
| With water Quantum yield; Irradiation; var. irradiation: γ-radiolysis; |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
94705-75-6, 95180-99-7, 142237-27-2
1-(6'-hydroxy-5',6'-dihydrothymin-5'-yl)thymine (5a) and 1-(5'-hydroxy-5',6'-dihydrothymin-6'-yl)thymine

-
D
-
696-04-8
5,6-dihydrothymine

| Conditions | Yield |
|---|---|
| With sodium chloride In water for 5h; galvanostatic electrolysis, Pt electrodes; Further byproducts given; | A 4.0 % Chromat. B 66.0 % Chromat. C 15.8 % Chromat. D 9.1 % Chromat. |

-
-
65-71-4
thymin

-
A
-
4433-40-3
5-hydroxymethyl uracil

-
B
-
13514-92-6
6-Hydroxydihydrothymine

-
C
-
20433-38-9
5,6-dihydroxyuracil

-
D
-
696-04-8
5,6-dihydrothymine

| Conditions | Yield |
|---|---|
| With Saline In water Ambient temperature; Irradiation; Further byproducts given; |


| Conditions | Yield |
|---|---|
| With thionyl chloride In 1,4-dioxane for 4h; Heating / reflux; | 100% |
| With hydrogenchloride at 20 - 35℃; | 99% |
| With hydrogenchloride at 20℃; for 2h; | 71% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 50℃; for 48h; | 94% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 4h; Heating; | 93.5% |

| Conditions | Yield |
|---|---|
| With dipotassium peroxodisulfate; silver nitrate In water at 40℃; for 0.583333h; | 93% |
| With dipotassium peroxodisulfate; silver nitrate In water | 84% |
| With dipotassium peroxodisulfate; silver nitrate In water at 40 - 45℃; | 83% |
-
-
4433-40-3
5-hydroxymethyl uracil

-
-
76-83-5
trityl chloride

-
-
53910-86-4
5-(triphenylmethoxymethyl)uracil

| Conditions | Yield |
|---|---|
| With pyridine at 95 - 100℃; for 3h; | 93% |

| Conditions | Yield |
|---|---|
| With trifluoroacetic acid at 130℃; for 0.333333h; Sealed tube; | 84% |
| With trifluoroacetic acid at 140℃; for 14h; | 82% |
-
-
4433-40-3
5-hydroxymethyl uracil

-
-
58479-61-1
tert-butylchlorodiphenylsilane

-
-
1234711-50-2
5-(tert-butyldiphenylsiloxy)methyl-1H,3H-pyrimidine-2,4-dione

| Conditions | Yield |
|---|---|
| With 1H-imidazole In tetrahydrofuran at 20℃; | 84% |
| With 1H-imidazole In N,N-dimethyl-formamide at 20℃; for 24h; Inert atmosphere; | 81% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride at 100℃; for 3h; | 83% |
-
-
4433-40-3
5-hydroxymethyl uracil

-
-
1006-59-3
2,6-diethylphenol

-
-
84876-24-4
5-(3,5-diethyl-4-hydroxybenzyl)uracil

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 5h; Heating; | 82% |
-
-
4433-40-3
5-hydroxymethyl uracil

-
-
98-88-4
benzoyl chloride

-
-
378750-51-7
bis-N,O-benzoyl-5-(hydroxymethyl)uracil

| Conditions | Yield |
|---|---|
| In pyridine; acetonitrile | 82% |
-
-
4433-40-3
5-hydroxymethyl uracil

-
-
105-36-2
ethyl bromoacetate

-
-
944249-69-8
ethyl 2-(5-hydroxymethyl-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)acetate

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 16h; | 82% |
| Stage #1: 5-hydroxymethyl uracil With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 1h; Inert atmosphere; Stage #2: ethyl bromoacetate In N,N-dimethyl-formamide Inert atmosphere; | 30% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 80℃; for 4h; | 82% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride a) r.t., 15 min, b) 100 deg C, 3h; | 81% |
-
-
4433-40-3
5-hydroxymethyl uracil

-
-
107-21-1
ethylene glycol

-
-
88459-62-5
5-<(2-hydroxyethoxy)methyl>uracil

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 0.0833333h; Heating; | 81% |
-
-
4433-40-3
5-hydroxymethyl uracil

| Conditions | Yield |
|---|---|
| With pyridine; potassium phosphate; tris-(dibenzylideneacetone)dipalladium(0); (1R,2R)-diamino-(1N,2N)-bis(1'-diphenylphosphino-2-naphthoyl)cyclohexane at 0℃; for 7h; Inert atmosphere; enantioselective reaction; | 79.6% |

| Conditions | Yield |
|---|---|
| With hydrogenchloride for 1h; Heating; | 79% |
| With acid | |
| With hydrogenchloride Reflux; |
-
-
4433-40-3
5-hydroxymethyl uracil

-
-
13035-61-5
1,2,3,5-tetraacetylribose

-
-
285549-57-7
2′,3′,5′-tri-O-acetyl-5-hydroxymethyluridine

| Conditions | Yield |
|---|---|
| With benzenesulfonamide; trimethylsilyl trifluoromethanesulfonate In acetonitrile at 75℃; for 2h; Substitution; | 79% |
5-Hydroxymethyluracil Chemical Properties
IUPAC Name: 5-(Hydroxymethyl)-1H-pyrimidine-2,4-dione
Following is the structure of 2,4(1H,3H)-Pyrimidinedione,5-(hydroxymethyl)- (CAS NO.4433-40-3):
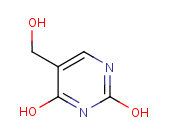
Empirical Formula: C5H6N2O3
Molecular Weight: 142.1127 g/mol
EINECS: 224-636-5
Index of Refraction: 1.528
Molar Refractivity: 31.23 cm3
Molar Volume: 101.3 cm3
Density: 1.401 g/cm3
Flash Point: 274.6 °C
Melting point: >300 °C
Surface Tension: 49.1 dyne/cm
Enthalpy of Vaporization: 84.8 kJ/mol
Boiling Point: 530.4 °C at 760 mmHg
Vapour Pressure: 4.44E-12 mmHg at 25°C
Product Categories of 2,4(1H,3H)-Pyrimidinedione,5-(hydroxymethyl)- (CAS NO.4433-40-3): PYRIMIDINE; Nucleotides and Nucleosides; Nucleic acids; 5-FOA; Bases & Related Reagents; Nucleotides; Building Blocks; Heterocyclic Building Blocks; Pyrimidines
Appearance of 2,4(1H,3H)-Pyrimidinedione,5-(hydroxymethyl)- (CAS NO.4433-40-3): White powder
Canonical SMILES: C1=C(C(=O)NC(=O)N1)CO
InChI: InChI=1S/C5H6N2O3/c8-2-3-1-6-5(10)7-4(3)9/h1,8H,2H2,(H2,6,7,9,10)
InChIKey: JDBGXEHEIRGOBU-UHFFFAOYSA-N
5-Hydroxymethyluracil Safety Profile
Hazard Codes:  Xi
Xi
Safety Statements: 22-24/25
S22:Do not breathe dust.
S24/25:Avoid contact with skin and eyes.
WGK Germany: 3
RTECS: YR0513000
Hazard Note: Irritant
HazardClass: IRRITANT
5-Hydroxymethyluracil Specification
2,4(1H,3H)-Pyrimidinedione,5-(hydroxymethyl)- , its cas register number is 4433-40-3. It also can be called 4-Methyl-5-oxyuracil ; 5-(Hydroxymethyl)uracil ; 5-Oxymethyluracil ; 5-Hydroxymethyluracil ; and Thymine, alpha-hydroxy- .
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View