-
Name
5-Methylpyridin-3-amine
- EINECS 624-420-4
- CAS No. 3430-19-1
- Article Data11
- CAS DataBase
- Density 1.068 g/cm3
- Solubility
- Melting Point 59-63 °C
- Formula C6H8N2
- Boiling Point 261.1 °C at 760 mmHg
- Molecular Weight 108.143
- Flash Point 135.6 °C
- Transport Information
- Appearance COLORLESS TO YELLOW SOLID
- Safety 26-36/37/39
- Risk Codes 22-37/38-41-20/21/22
-
Molecular Structure
-
Hazard Symbols
 T,
T,  Xi,
Xi,  Xn
Xn
- Synonyms 3-Picoline,5-amino- (7CI,8CI);(5-Methylpyridin-3-yl)amine;3-Amino-5-methylpyridine;5-Methyl-3-pyridinamine;
- PSA 38.91000
- LogP 1.55340
Synthetic route

| Conditions | Yield |
|---|---|
| With potassium hydroxide; bromine 1.) RT, 1 h, 2.) 70 deg C, 15 min; | 67% |
| With sodium hydroxide; bromine In tetrahydrofuran at 5 - 70℃; Hoffmann rearrangement; |
-
-
18344-51-9
2-amino-3-methyl-5-nitropyridine

-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| With hydrazine In water at 110 - 120℃; for 5h; | 45% |
| Multi-step reaction with 3 steps 1: calcium chloride; aqueous sodium nitrite solution; aqueous hydrochloric acid / Erhitzen des Reaktionsprodukts mit Wasser 2: phosphoryl chloride 3: palladium/charcoal; sodium acetate; acetic acid / Hydrogenation View Scheme | |
| Multi-step reaction with 2 steps 1: calcium chloride; aqueous sodium nitrite solution; aqueous hydrochloric acid / Erhitzen des Reaktionsprodukts mit Wasser 2: palladium/charcoal; sodium acetate; acetic acid / Hydrogenation View Scheme |

| Conditions | Yield |
|---|---|
| With hydrazine In water at 110 - 120℃; for 5h; | 41% |
-
-
22280-56-4
2-chloro-3-methyl-5-nitropyridine

-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; sodium acetate; acetic acid Hydrogenation; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 27 percent / potassium permanganate / H2O / 1.) from RT to 43 deg C, 5 h, 2.) 45 deg C, overnight 2: thionyl chloride / Heating 3: ammonia gas / 1,2-dichloro-ethane / 0.5 h / -30 °C 4: 67 percent / bromine, aq. potassium hydroxide / 1.) RT, 1 h, 2.) 70 deg C, 15 min View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: thionyl chloride / Heating 2: ammonia gas / 1,2-dichloro-ethane / 0.5 h / -30 °C 3: 67 percent / bromine, aq. potassium hydroxide / 1.) RT, 1 h, 2.) 70 deg C, 15 min View Scheme |
-
-
884494-95-5
5-methyl-nicotinic acid chloride

-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: ammonia gas / 1,2-dichloro-ethane / 0.5 h / -30 °C 2: 67 percent / bromine, aq. potassium hydroxide / 1.) RT, 1 h, 2.) 70 deg C, 15 min View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: H2SO4; HNO3 2: calcium chloride; aqueous sodium nitrite solution; aqueous hydrochloric acid / Erhitzen des Reaktionsprodukts mit Wasser 3: phosphoryl chloride 4: palladium/charcoal; sodium acetate; acetic acid / Hydrogenation View Scheme | |
| Multi-step reaction with 3 steps 1: H2SO4; HNO3 2: calcium chloride; aqueous sodium nitrite solution; aqueous hydrochloric acid / Erhitzen des Reaktionsprodukts mit Wasser 3: palladium/charcoal; sodium acetate; acetic acid / Hydrogenation View Scheme |
-
-
21901-34-8
2-hydroxy-3-methyl-5-nitropyridine

-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: phosphoryl chloride 2: palladium/charcoal; sodium acetate; acetic acid / Hydrogenation View Scheme |
-
-
23132-21-0
2-bromo-3-methyl-5-nitropyridine

-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In methanol |

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); ammonia; 2'-(di-tert-butylphosphanyl)-N,N-dimethyl-[1,1'-biphenyl]-2-amine; sodium t-butanolate In 1,4-dioxane for 24h; Reagent/catalyst; | 86 %Chromat. |
-
-
22353-33-9
3-chloro-5-nitropyridine

-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| With palladium on activated charcoal; hydrogen In methanol |
-
-
3430-19-1
5-methylpyridin-3-amine

-
-
24424-99-5
di-tert-butyl dicarbonate

-
-
631910-23-1
tert-butyl N-(5-methylpyridin-3-yl)carbamate

| Conditions | Yield |
|---|---|
| Stage #1: 5-methylpyridin-3-amine With sodium hexamethyldisilazane In tetrahydrofuran at 20℃; for 0.25h; Stage #2: di-tert-butyl dicarbonate In tetrahydrofuran at 20℃; | 88% |
| With sodium hexamethyldisilazane In tetrahydrofuran at 20℃; | 88% |
| Stage #1: 5-methylpyridin-3-amine With sodium hexamethyldisilazane In tetrahydrofuran at 20℃; for 0.25h; Stage #2: di-tert-butyl dicarbonate In tetrahydrofuran at 20℃; | 88% |
-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| With triethylamine; N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate In N,N-dimethyl-formamide at 20℃; | 86% |

| Conditions | Yield |
|---|---|
| Stage #1: N-Benzyloxycarbonyl-L-proline With chloroformic acid ethyl ester; triethylamine In tetrahydrofuran at 0℃; for 0.5h; Inert atmosphere; Stage #2: 5-methylpyridin-3-amine In tetrahydrofuran at 0 - 70℃; for 25h; Inert atmosphere; | 81% |

| Conditions | Yield |
|---|---|
| With sodium hydride In N,N-dimethyl-formamide at 20℃; Inert atmosphere; | 79% |
-
-
3430-19-1
5-methylpyridin-3-amine

-
-
530-62-1
1,1'-carbonyldiimidazole

-
-
1387638-53-0
1,3-bis(5-methylpyridin-3-yl)urea

| Conditions | Yield |
|---|---|
| In toluene at 20 - 80℃; | 75% |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In toluene at 100℃; for 10h; | 75% |
| In toluene at 100℃; for 10h; | 75% |
-
-
3430-19-1
5-methylpyridin-3-amine

-
-
1885-14-9
phenyl chloroformate

-
-
438190-95-5
(5-methyl-pyridin-3-yl)-carbamic acid phenyl ester

| Conditions | Yield |
|---|---|
| With pyridine In tetrahydrofuran | 71% |
| With pyridine In tetrahydrofuran at 0℃; | 71% |

| Conditions | Yield |
|---|---|
| With N,N,N'N'-tetramethyl-1,3-propanediamine; boric acid In 1,3,5-trimethyl-benzene at 164℃; for 7h; Dean-Stark; Inert atmosphere; Green chemistry; regiospecific reaction; | 70% |
-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| With acetic acid at 120℃; for 12h; | 64.5% |
| In acetic acid for 18h; Reflux; |
-
-
3430-19-1
5-methylpyridin-3-amine

-
-
1027256-79-6
3-{1-[(6-chloropyrazin-2-yl)amino]ethyl}benzoic acid

-
-
1027253-93-5
3-{1-[(6-chloropyrazin-2-yl)amino]ethyl}-N-(5-methylpyridin-3-yl)benzamide

| Conditions | Yield |
|---|---|
| With 4-pyrrolidin-1-ylpyridine; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; triethylamine In dichloromethane at 20℃; for 17h; | 61% |
-
-
3430-19-1
5-methylpyridin-3-amine


| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; carbonic acid dimethyl ester In tetrahydrofuran; dichloromethane at 20℃; | 53.9% |
-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| Stage #1: 4-(((4-(tert-butyl)phenyl)sulfonamido)methyl)benzoic acid With oxalyl dichloride; N,N-dimethyl-formamide In tetrahydrofuran at 20 - 66℃; for 3h; Stage #2: 5-methylpyridin-3-amine With pyridine at 20℃; for 16h; | 50% |
-
-
3430-19-1
5-methylpyridin-3-amine

-
-
52412-07-4
2,4-dibromo-butyryl bromide

| Conditions | Yield |
|---|---|
| Stage #1: 5-methylpyridin-3-amine; 2,4-dibromo-butyryl bromide In dichloromethane at 0 - 20℃; for 3h; Stage #2: With sodium hydroxide In dichloromethane; water at 20℃; for 3h; | 49.2% |
-
-
3430-19-1
5-methylpyridin-3-amine


| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In tetrahydrofuran; dichloromethane; toluene at 20℃; for 0.5h; | 46.3% |
-
-
3430-19-1
5-methylpyridin-3-amine

-
-
32315-10-9
bis(trichloromethyl) carbonate

| Conditions | Yield |
|---|---|
| Stage #1: 5-methylpyridin-3-amine; bis(trichloromethyl) carbonate; (4S)-7-(2-methylpyridin-4-yl)-2,3,4,5-tetrahydro-1,4-methanopyrido[2,3-b][1,4]diazepine In tetrahydrofuran at 30℃; for 1h; Stage #2: 5-methylpyridin-3-amine With triethylamine at 30 - 65℃; for 16h; | 45% |
| Stage #1: bis(trichloromethyl) carbonate; (4S)-7-(2-methylpyridin-4-yl)-2,3,4,5-tetrahydro-1,4-methanopyrido[2,3-b][1,4]diazepine In tetrahydrofuran at 30℃; for 1h; Stage #2: 5-methylpyridin-3-amine With triethylamine In tetrahydrofuran at 65℃; for 16h; | 45% |

-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| Stage #1: 5-methylpyridin-3-amine With lithium hexamethyldisilazane In tetrahydrofuran at 20℃; for 0.166667h; Stage #2: 1-(5-(4,6-dichloropyrimidin-2-yl)-2-methylpiperidin-1-yl)ethan-1-one In tetrahydrofuran at 20℃; for 16h; | 45% |
-
-
3430-19-1
5-methylpyridin-3-amine

-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
1027255-35-1
(S)-N-[1-(3-aminophenyl)ethyl]-6-chloropyrazin-2-amine

-
-
1027253-94-6
N-(3-{(1S)-1-[(6-chloropyrazin-2-yl)amino]ethyl}phenyl)-N'-(5-methylpyridin-3-yl)urea

| Conditions | Yield |
|---|---|
| Stage #1: bis(trichloromethyl) carbonate; (S)-N-[1-(3-aminophenyl)ethyl]-6-chloropyrazin-2-amine With sodium hydrogencarbonate In dichloromethane; water for 3h; Stage #2: 5-methylpyridin-3-amine In dichloromethane; water for 17h; | 42% |

-
-
3430-19-1
5-methylpyridin-3-amine


| Conditions | Yield |
|---|---|
| With pyridine; N-ethyl-N,N-diisopropylamine In dichloromethane at 20℃; for 0.5h; | 39.6% |
-
-
3430-19-1
5-methylpyridin-3-amine

-
-
591-50-4
iodobenzene

-
-
73183-34-3
bis(pinacol)diborane

-
-
10477-94-8
3-methyl-5-phenylpyridine

| Conditions | Yield |
|---|---|
| Stage #1: 5-methylpyridin-3-amine; bis(pinacol)diborane With tert.-butylnitrite In acetonitrile at 80℃; for 2h; Stage #2: iodobenzene With tetrakis(triphenylphosphine) palladium(0); potassium carbonate In N,N-dimethyl-formamide at 100℃; for 12h; Suzuki-Miyaura Coupling; Inert atmosphere; | 35% |

-
-
3430-19-1
5-methylpyridin-3-amine

-
-
26638-43-7
2-methoxycarbonylbenzenesulfonyl chloride

-
-
931126-93-1
methyl 2-{[(5-methylpyridin-3-yl)amino]sulfonyl}benzoate

| Conditions | Yield |
|---|---|
| With triethylamine In dichloromethane at 20℃; for 1h; | 34% |
-
-
3430-19-1
5-methylpyridin-3-amine

-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
1303585-09-2
(S)-6-(3-(3-fluoropyrrolidin-1-yl)phenyl)-2,2-dimethyl-N-(5-methylpyridin-3-yl)-2H-pyrido[3,2-b][1,4]oxazine-4(3H)-carboxamide

| Conditions | Yield |
|---|---|
| Stage #1: bis(trichloromethyl) carbonate; (S)-6-(3-(3-fluoropyrrolidin-1-yl)phenyl)-2,2-dimethyl-3,4-dihydro-2H-pyrido[3,2-b][1,4]oxazine With triethylamine In tetrahydrofuran at 20℃; for 1.5h; Stage #2: 5-methylpyridin-3-amine In tetrahydrofuran at 60℃; for 18h; | 31% |

-
-
3430-19-1
5-methylpyridin-3-amine

-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
1303588-28-4
7-(3-(trifluoromethyl)phenyl)-1,2,3,4-tetrahydro-1,8-naphthyridine

-
-
1303583-59-6
N-(5-methylpyridin-3-yl)-7-(3-(trifluoromethyl)phenyl)-3,4-dihydro-1,8-naphthyridine-1(2H)-carboxamide

| Conditions | Yield |
|---|---|
| Stage #1: bis(trichloromethyl) carbonate; 7-(3-(trifluoromethyl)phenyl)-1,2,3,4-tetrahydro-1,8-naphthyridine With triethylamine In tetrahydrofuran at 30℃; for 0.5h; Inert atmosphere; Stage #2: 5-methylpyridin-3-amine In tetrahydrofuran at 60℃; for 18h; Inert atmosphere; | 27% |

-
-
3430-19-1
5-methylpyridin-3-amine

-
A
-
126325-51-7
3-amino-2,6-dibromo-5-methylpyridine

-
B
-
34552-14-2
3-amino-2-bromo-5-methylpyridine

| Conditions | Yield |
|---|---|
| With hydrogen bromide; dihydrogen peroxide at 70℃; for 1h; Product distribution; | A 26% B 6% C 0.5% |
| With hydrogen bromide; dihydrogen peroxide at 70℃; for 1h; | A 26% B 6% C 0.5% |

-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| With tris-(dibenzylideneacetone)dipalladium(0); dicyclohexyl-(2′,4′,6′-triisopropyl-3,6-dimethoxy-[1,1′-biphenyl]-2-yl)phosphine; caesium carbonate In 1,4-dioxane at 100℃; for 16h; Inert atmosphere; | 22% |
-
-
3430-19-1
5-methylpyridin-3-amine

-
-
32315-10-9
bis(trichloromethyl) carbonate

-
-
1303588-19-3
2,2-dimethyl-6-(3-(pyrrolidin-1-ylmethyl)phenyl)-3,4-dihydro-2H-pyrido[3,2-b][1,4]oxazine

-
-
1303584-47-5
2,2-dimethyl-N-(5-methylpyridin-3-yl)-6-(3-(pyrrolidin-1-ylmethyl)phenyl)-2H-pyrido[3,2-b][1,4]oxazine-4(3H)-carboxamide

| Conditions | Yield |
|---|---|
| Stage #1: bis(trichloromethyl) carbonate; 2,2-dimethyl-6-(3-(pyrrolidin-1-ylmethyl)phenyl)-3,4-dihydro-2H-pyrido[3,2-b][1,4]oxazine With triethylamine In tetrahydrofuran at 20℃; for 0.5h; Stage #2: 5-methylpyridin-3-amine In tetrahydrofuran at 60℃; | 20% |

-
-
3430-19-1
5-methylpyridin-3-amine

| Conditions | Yield |
|---|---|
| With 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphinane-2,4,6-trioxide; triethylamine In ethyl acetate at 20℃; | 20% |

| Conditions | Yield |
|---|---|
| With sodium hydrogencarbonate In dichloromethane; water at 0℃; for 2h; | 19% |
5-Methylpyridin-3-amine Chemical Properties
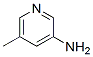
Molecule structure of 3-Amino-5-Methylpyridine (CAS NO.3430-19-1) :
IUPAC Name: (2S)-2-amino-3-(3-chloro-4-hydroxyphenyl)propanoic acid
Molecular Weight: 215.6336 g/mol
Molecular Formula: C9H10ClNO3
Density: 1.068 g/cm3
Melting Point: 59-63 °C
Boiling Point: 261.1 °C at 760 mmHg
Flash Point: 135.6 °C
Molar Volume: 101.2 cm3
Polarizability: 13.24*10-24 cm3
Surface Tension: 46.9 dyne/cm
Enthalpy of Vaporization: 49.88 kJ/mol
Vapour Pressure: 0.0118 mmHg at 25 °C
XLogP3: -1.8
H-Bond Donor: 3
H-Bond Acceptor: 4
Rotatable Bond Count: 3
Tautomer Count: 3
Exact Mass: 215.034921
MonoIsotopic Mass: 215.034921
Topological Polar Surface Area: 83.6
Heavy Atom Count: 14
Complexity: 212
Defined Atom StereoCenter Count: 1
Canonical SMILES: C1=CC(=C(C=C1CC(C(=O)O)N)Cl)O
Isomeric SMILES: C1=CC(=C(C=C1C[C@@H](C(=O)O)N)Cl)O
InChI: InChI=1S/C9H10ClNO3/c10-6-3-5(1-2-8(6)12)4-7(11)9(13)14/h1-3,7,12H,4,11H2,(H,13,14)/t7-/m0/s1
InChIKey: ACWBBAGYTKWBCD-ZETCQYMHSA-N
Product Categories of 3-Amino-5-Methylpyridine (CAS NO.3430-19-1) : VARIOUSAMINE; Heterocyclic Compounds; Amino; Intermediate; Pyridine; Boronic Acid; Heterocyclic Building Blocks; Pyridines; Building Blocks
5-Methylpyridin-3-amine Safety Profile
Hazard Codes:  T,
T,  Xi,
Xi,  Xn
Xn
Risk Statements: 22-37/38-41-20/21/22
R22: Harmful if swallowed
R37/38: Irritating to respiratory system and skin
R41:Risk of serious damage to the eyes
R20/21/22: Harmful by inhalation, in contact with skin and if swallowed
Safety Statements: 26-36/37/39-36
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice
S36/37/39: Wear suitable protective clothing, gloves and eye/face protection
S36: Wear suitable protective clothing
Hazard Note: Toxic
5-Methylpyridin-3-amine Specification
3-Amino-5-Methylpyridine (CAS NO.3430-19-1) is also called 3-Amino-5-picoline ; 5-Methyl-pyridin-3-ylamine ; 3-Amino-5-methylpyridine97% ; 3-Pyridinamine,5-methyl-(9CI) ; 5-Methyl-3-pyridylamine hydrochloride salt ; 5-Methyl-3-pyridinamine ; 5-Amino-3-methylpyridine .
Related Products
- 5-Methylpyridin-3-amine
- 3430-21-5
- 3430-22-6
- 3430-23-7
- 34302-37-9
- 3430-25-9
- 3430-26-0
- 34302-69-7
- 3430-27-1
- 3430-29-3
- 3430-31-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View