-
Name
7-Hydroxyquinolinone
- EINECS 812-453-9
- CAS No. 70500-72-0
- Article Data24
- CAS DataBase
- Density 1.282g/cm3
- Solubility
- Melting Point 187-192°C
- Formula C9H7 N O2
- Boiling Point 403.7oC at 760 mmHg
- Molecular Weight 161.16
- Flash Point 198°C
- Transport Information
- Appearance Pale yellow powder
- Safety
- Risk Codes R22; R43
-
Molecular Structure
-
Hazard Symbols
 Xn,
Xn, Xi
Xi
- Synonyms 7-Hydroxy-1H-quinolin-2-one;7-Hydroxy-2(1H)-quinolinone; 7-Hydroxycarbostyril
- PSA 53.09000
- LogP 1.23370
Synthetic route
-
-
22246-18-0
7-hydroxy-3,4-dihydro-2-(1H)-quinolinone

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; water; oxygen; acetic acid at 110 - 116℃; for 18h; Reagent/catalyst; Temperature; | 94.2% |
| With palladium 10% on activated carbon In decalin at 180℃; for 8h; | 90.9% |
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In tetrahydrofuran at 66℃; for 4h; Catalytic behavior; Concentration; Solvent; Temperature; | 89% |
-
-
220364-99-8
2-oxo-1,2-dihydroquinolin-7-yl-acetate

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water for 1.5h; | 90% |

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Stage #1: C18H20BNO6 With aluminum (III) chloride; tris(pentafluorophenyl)borate In 5,5-dimethyl-1,3-cyclohexadiene at 0 - 70℃; for 2h; Stage #2: With dihydrogen peroxide; acetic acid In 5,5-dimethyl-1,3-cyclohexadiene at 0 - 20℃; for 2h; | 82.2% |

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Stage #1: C17H18BNO5 With aluminum (III) chloride; tris(pentafluorophenyl)borate In toluene at 0 - 90℃; for 2h; Stage #2: With dihydrogen peroxide; acetic acid In toluene at 0 - 20℃; for 2h; | 81.3% |
-
-
127033-74-3
(2E)-N-(3-methoxyphenyl)-3-phenylacrylamide

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With aluminium trichloride In chlorobenzene at 120℃; for 2h; | 70% |
| With aluminium trichloride In chlorobenzene | 70% |
| With aluminum (III) chloride In chlorobenzene at 0℃; for 8h; Reflux; | 67% |
-
-
93-35-6
7-hydroxy-2H-chromen-2-one

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With acetic acid In acetic acid for 3h; Substitution; Heating; | 55% |
-
-
15116-41-3
N-(3-methoxyphenyl)cinnamamide

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In chlorobenzene at 0 - 120℃; | 53% |
| With aluminum (III) chloride at 120 - 180℃; for 2.08333h; | 43% |
| With aluminum (III) chloride at 120 - 180℃; for 2h; |
-
-
536-90-3
m-Anisidine

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 95 percent 2: 70 percent / AlCl3 / chlorobenzene View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / water; acetone / 20 °C / Cooling with ice 2: aluminum (III) chloride / chlorobenzene / 0 - 120 °C View Scheme | |
| Multi-step reaction with 2 steps 1: pyridine / dichloromethane / 1 h / 5 - 20 °C 2: aluminum (III) chloride / 2.08 h / 120 - 180 °C View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / acetone; water / 0.5 h 2: aluminum (III) chloride / chlorobenzene / 120 °C / Cooling with ice View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / acetone; water / 0.5 h / 0 °C 2: aluminum (III) chloride / chlorobenzene / Reflux View Scheme |
-
-
93499-67-3
7-hydroxyquinoline 1-oxide

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 24 percent / 2 h / Heating 2: 90 percent / 1.0 N NaOH / H2O / 1.5 h View Scheme |
-
-
536-90-3
m-Anisidine

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 95 percent / aq. K2CO3 / acetone / 0.5 h / 0 °C 2: 70 percent / AlCl3 / chlorobenzene / 2 h / 120 °C View Scheme |
-
-
591-27-5
m-Hydroxyaniline

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With pyridine; AlCl3 In dichloromethane; ethyl acetate | |
| Multi-step reaction with 3 steps 1.1: sodium hydrogencarbonate / dichloromethane / 0.5 h / 20 °C / Inert atmosphere 1.2: 2 h / 20 °C / Inert atmosphere 2.1: aluminum (III) chloride / N,N-dimethyl acetamide / 4 h / 70 - 140 °C 3.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / tetrahydrofuran / 2 h / 20 - 50 °C / Inert atmosphere 3.2: 1 h / 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1: sodium hydrogencarbonate / ethyl acetate / 20 - 30 °C 2: aluminum (III) chloride / N,N-dimethyl acetamide / 130 - 140 °C 3: palladium 10% on activated carbon; water; acetic acid; oxygen / 18 h / 110 - 116 °C View Scheme |
-
-
50297-40-0
3-chloro-N-(3-hydroxyphenyl)propanamide

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: aluminum (III) chloride / N,N-dimethyl acetamide / 4 h / 70 - 140 °C 2.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / tetrahydrofuran / 2 h / 20 - 50 °C / Inert atmosphere 2.2: 1 h / 20 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1: aluminum (III) chloride / N,N-dimethyl acetamide / 130 - 140 °C 2: palladium 10% on activated carbon; water; acetic acid; oxygen / 18 h / 110 - 116 °C View Scheme |
-
-
14737-89-4
3,4-dimethoxy-trans-cinnamic acid

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: thionyl chloride / toluene / 4 h / 70 °C 2.1: triethylamine / tetrahydrofuran / 2 h / -5 - 20 °C 3.1: tris(pentafluorophenyl)borate; aluminum (III) chloride / toluene / 2 h / 0 - 90 °C 3.2: 2 h / 0 - 20 °C View Scheme |
-
-
39856-08-1, 141236-46-6
(E)-3,4-dimethoxycinnamic chloride

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: triethylamine / tetrahydrofuran / 2 h / -5 - 20 °C 2.1: tris(pentafluorophenyl)borate; aluminum (III) chloride / toluene / 2 h / 0 - 90 °C 2.2: 2 h / 0 - 20 °C View Scheme |
-
-
20329-98-0, 20329-99-1, 90-50-6
(E)-3,4,5-trimethoxy-cinnamic acid

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: thionyl chloride / toluene / 4 h / 70 °C 2.1: pyridine / tetrahydrofuran / 2 h / -5 - 20 °C 3.1: tris(pentafluorophenyl)borate; aluminum (III) chloride / 5,5-dimethyl-1,3-cyclohexadiene / 2 h / 0 - 70 °C 3.2: 2 h / 0 - 20 °C View Scheme |
-
-
10263-19-1, 89652-61-9
(E)-3-(3,4,5-trimethoxyphenyl)acryloyl chloride

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: pyridine / tetrahydrofuran / 2 h / -5 - 20 °C 2.1: tris(pentafluorophenyl)borate; aluminum (III) chloride / 5,5-dimethyl-1,3-cyclohexadiene / 2 h / 0 - 70 °C 2.2: 2 h / 0 - 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: m-Anisidine; Cinnamoyl chloride In dichloromethane for 4h; Reflux; Stage #2: With aluminum (III) chloride In chlorobenzene for 8h; Reflux; |
-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

-
-
375358-19-3
2-chloroquinoline-7-ol

| Conditions | Yield |
|---|---|
| With trichlorophosphate In N,N-dimethyl-formamide at 70 - 75℃; Reagent/catalyst; | 94% |
| With thionyl chloride In N,N-dimethyl-formamide at 0 - 70℃; for 1h; | 80.9% |
-
-
6940-78-9
4-chlorobutyl bromide

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 40 - 50℃; for 5h; | 93.7% |
| With triethylamine In ethanol at 60 - 80℃; for 2h; Concentration; Solvent; Temperature; Reagent/catalyst; | 92.2% |
| With sodium hydroxide In ethanol for 5h; Reflux; | 88% |
-
-
108-24-7
acetic anhydride

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

-
-
220364-99-8
2-oxo-1,2-dihydroquinolin-7-yl-acetate

| Conditions | Yield |
|---|---|
| With indium(III) triflate at 0 - 30℃; for 1h; | 90% |
-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

-
-
164365-88-2
tert-butyl 4-bromobutylcarbamate

| Conditions | Yield |
|---|---|
| With potassium carbonate In water; N,N-dimethyl-formamide at 45 - 55℃; | 87.4% |
-
-
110-52-1
1,4-dibromo-butane

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

-
-
203395-59-9
7–(4-bromobutoxy)quinolin-2(1H)-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 20℃; for 3h; Reflux; | 86.5% |
| With potassium carbonate In water; N,N-dimethyl-formamide at 30 - 40℃; for 4h; | 82% |
| With potassium carbonate In acetone for 4h; Reflux; | 78% |
-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

-
-
4559-70-0
Diphenylphosphine oxide

| Conditions | Yield |
|---|---|
| With eosin; dibenzoyl peroxide In water; dimethyl sulfoxide at 20℃; for 24h; Inert atmosphere; Irradiation; regioselective reaction; | 86% |
-
-
369-34-6
3,4-difluoronitrobenzene

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; | 86% |
-
-
33977-38-7
1-bromo-4-aminobutane

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Stage #1: 4-bromo-1-aminobutane With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 0.5h; Stage #2: 7-hydroxy-1H-quinolin-2-one With sodium iodide In N,N-dimethyl-formamide at 100℃; for 0.666667h; Temperature; | 84.3% |

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In N,N-dimethyl-formamide at 80 - 90℃; | 84.1% |

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate; potassium iodide In N,N-dimethyl-formamide at 80 - 90℃; for 20h; | 83% |

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide at 130 - 135℃; Reagent/catalyst; Temperature; | 82% |
-
-
33036-62-3
4-Bromo-1-butanol

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 24h; Reflux; | 81% |
| With potassium carbonate In water; N,N-dimethyl-formamide at 40 - 50℃; | 81.4% |
-
-
29882-07-3
4-chlorobutanal dimethyl acetal

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Stage #1: 7-hydroxy-1H-quinolin-2-one With potassium carbonate In N,N-dimethyl-formamide at 25 - 35℃; for 0.5h; Stage #2: 4-chlorobutanal dimethyl acetal In N,N-dimethyl-formamide at 70 - 75℃; for 10h; | 80% |
| With tetrabutylammomium bromide; potassium carbonate In dimethyl sulfoxide at 20 - 70℃; for 20h; |
-
-
928-51-8
4-Chloro-1-butanol

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| Stage #1: 7-hydroxy-1H-quinolin-2-one With potassium carbonate In N,N-dimethyl-formamide at 25 - 35℃; for 0.5h; Stage #2: 4-Chloro-1-butanol In N,N-dimethyl-formamide at 70 - 75℃; | 80% |
-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

-
-
109-64-8
1,3-dibromo-propane

-
-
1076199-59-1
7–(3-bromopropoxy)quinolin-2(1H)-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 4h; Reflux; | 79% |
| With potassium carbonate In ethanol Reflux; | 55% |
| With potassium carbonate In acetonitrile at 50℃; for 20h; |
-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With phosphorus tribromide In N,N-dimethyl-formamide at 70 - 75℃; | 79% |
-
-
4897-84-1
Methyl 4-bromobutyrate

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

-
-
87364-43-0
4-(2-Oxo-1,2-dihydro-quinolin-7-yloxy)-butyric acid methyl ester

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In isopropyl alcohol for 4h; Heating; | 78% |
-
-
6139-83-9
4-chloro-1,1-diethoxybutane

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With tetrabutylammomium bromide; sodium carbonate In dimethyl sulfoxide at 100 - 110℃; for 3h; Reagent/catalyst; Solvent; Temperature; Large scale; | 78% |
-
-
16234-14-3
2,4-dichlorothieno[3,2-d]pyrimidine

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In water; acetonitrile at 70℃; | 78% |
| With potassium carbonate In water; acetonitrile Reflux; | 78% |

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With N,N-dimethyl acetamide; potassium carbonate; 4-methyl-2-pentanone at 116 - 118℃; for 20h; Reagent/catalyst; | 77.9% |
-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

-
-
106-93-4
ethylene dibromide

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 4h; Reflux; | 77% |
| With potassium hydroxide In methanol at 65℃; for 4h; Inert atmosphere; | 16.2% |
-
-
111-24-0
1,5-dibromo-pentane

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 4h; Reflux; | 76% |
| With potassium carbonate In acetonitrile at 50℃; for 20h; |
-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

-
-
403-29-2
2-bromo-4'-fluoroacetophenone

-
-
220365-01-5
7-<2-(4-fluorophenyl)-2-oxoethoxy>quinolin-2(1H)-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 24.5h; Ambient temperature; | 75% |
-
-
606-55-3
1-ethyl-2-methylquinolinium iodide

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With piperidine In ethanol Condensation; Heating; | 75% |
-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

-
-
106-95-6
allyl bromide

-
-
177419-02-2
7-(prop-2-en-1-yloxy)-1,2-dihydroquinolin-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone at 60℃; for 16h; | 74% |
| With potassium carbonate In acetone at 60℃; for 16h; | 74% |
-
-
629-03-8
1 ,6-dibromohexane

-
-
70500-72-0
7-hydroxy-1H-quinolin-2-one

| Conditions | Yield |
|---|---|
| With potassium carbonate In acetone for 4h; Reflux; | 74% |
7-Hydroxyquinolinone Chemical Properties
Molecular structure of 7-Hydroxyquinolinone (CAS NO.70500-72-0) is:
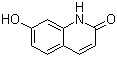
Product Name: 7-Hydroxyquinolinone
CAS Registry Number: 70500-72-0
Systematic Name: 7-hydroxyquinolin-2(1H)-one
Empirical Formula: C9H7NO2
Molecular Weight: 161.1574
Melting Point: 187-192°C
Index of Refraction: 1.639
Molar Refractivity: 43.391 cm3
Molar Volume: 120.517 cm3
Surface Tension: 55.384 dyne/cm
Density: 1.337 g/cm3
Flash Point: 211.944 °C
Enthalpy of Vaporization: 70.809 kJ/mol
Boiling Point: 426.837 °C at 760 mmHg
Product Categories: Aromatics Compounds;Quinoline;Aromatics;Intermediates & Fine Chemicals;Pharmaceuticals
7-Hydroxyquinolinone Specification
7-Hydroxyquinolinone , its cas register number is 70500-72-0. It also can be called 7-Hydroxy-2-(1H)-quinolinone ; 7-Hydroxycarbostyril ; 2,7-Dihydroxyquinoline ; 7-Hydroxy-2-Quinolinone ; 7-Hydroxy-1h-Quinolin-2-One ; 7-Hydroxyquinoline-(1h)-2-One ; 3,4-Dihydro-7-hydroxyquinoline-2(1H)-one .It is a off-white solid.
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View