-
Name
D-(+)-Pantothenic acid calcium salt
- EINECS 205-278-9
- CAS No. 137-08-6
- Article Data9
- CAS DataBase
- Density 1.266 g/cm3
- Solubility water: 50 mg/mL at 25 °C, clear, nearly colorless
- Melting Point 190 °C
- Formula C18H32CaN2O10
- Boiling Point 551.5 °C at 760 mmHg
- Molecular Weight 476.537
- Flash Point 287.3 °C
- Transport Information 25kgs
- Appearance white crystalline powder
- Safety 24/25-45-36/37/39-26-22
- Risk Codes 20/21/22-37/38-41-48
-
Molecular Structure
-
Hazard Symbols
 Xn
Xn
- Synonyms b-Alanine,N-(2,4-dihydroxy-3,3-dimethyl-1-oxobutyl)-, calcium salt (2:1), (R)-;(+)-Pantothenic acid calcium salt;Calcium D-(+)-N-(a,g-dihydroxy-b,b-dimethylbutyryl)-b-alaninate;Calcium D-(+)-pantothenate;Calcium D-pantothenate;b-Alanine,N-[(2R)-2,4-dihydroxy-3,3-dimethyl-1-oxobutyl]-, calcium salt (2:1);Calpan;Calpanate;Dextro calcium pantothenate;N-(2,4-Dihydroxy-3,3-dimethylbutyryl)-b-alanine calcium;NSC 36292;Pancal;Panthoject;Pantholin;Pantothenate calcium;Pantothenicacid calcium salt;Pantothenic acid hemicalcium salt;Vitamin B5 calcium salt;D-Calcium Pantothenate;Calcium,bis(pantothenato)- (7CI);Pantothenic acid, calcium salt (2:1), D- (8CI);
- PSA 191.72000
- LogP -1.45900
Synthetic route

| Conditions | Yield |
|---|---|
| Stage #1: calcium beta-alaninate In ethanol at 75℃; for 6h; Stage #2: D-pantolactone In ethanol at 75℃; for 16h; Temperature; | 96% |

| Conditions | Yield |
|---|---|
| With sodium chloride; calcium oxide Reagent/catalyst; Milling; | 71% |
| Stage #1: (R)-Pantolacton In methanol for 1h; Heating; Large scale; Green chemistry; Stage #2: calcium beta-alaninate In methanol for 2h; Reflux; Large scale; Green chemistry; |

| Conditions | Yield |
|---|---|
| In methanol Product distribution / selectivity; |

-
-
137-08-6, 867-80-1, 6381-62-0, 6381-63-1, 15773-29-2, 64789-49-7, 122484-60-0, 131346-21-9
calcium-N-(2,4-dihydroxy-3,3-dimethylbutyryl)-β-alaninate

-
A
-
137-08-6
calcium (R)-pantothenate

-
B
-
137-08-6, 867-80-1, 6381-62-0, 6381-63-1, 15773-29-2, 64789-49-7, 122484-60-0, 131346-21-9
calcium (S)-pantothenate

| Conditions | Yield |
|---|---|
| In methanol; water at 35℃; Resolution of racemate; |

| Conditions | Yield |
|---|---|
| With oxalic acid In water at 20℃; for 2h; | 95.11% |
| With (1S)-10-camphorsulfonic acid; trifluoroacetic acid at 0℃; | |
| With Amberlite IR-120 In water Inert atmosphere; | |
| With oxalic acid In water at 20℃; for 2h; |
-
-
137-08-6
calcium (R)-pantothenate

-
-
67-63-0
isopropyl alcohol

-
-
131205-05-5
(R)-isopropyl 3-(2,4-dihydroxy-3,3-dimethylbutanamido)propanoate

| Conditions | Yield |
|---|---|
| With hydrogenchloride In water at 0 - 20℃; Reflux; | 90% |
-
-
2186-92-7
p-Anisaldehyde dimethyl acetal

-
-
137-08-6
calcium (R)-pantothenate

-
-
864239-47-4
3-{[(4R)-2-(p-methoxyphenyl)-5,5-dimethyl-1,3-dioxan-4-yl]carbonylamino}propionic acid

| Conditions | Yield |
|---|---|
| Stage #1: calcium (R)-pantothenate With sulfuric acid In N,N-dimethyl-formamide for 1h; Inert atmosphere; Stage #2: p-Anisaldehyde dimethyl acetal In N,N-dimethyl-formamide for 16h; Inert atmosphere; | 75% |
| Stage #1: calcium (R)-pantothenate With sulfuric acid In acetone at 20℃; for 2h; Stage #2: p-Anisaldehyde dimethyl acetal With toluene-4-sulfonic acid In acetone at 20℃; for 37h; | 34% |
-
-
137-08-6
calcium (R)-pantothenate

-
-
67-64-1
acetone

-
-
167308-62-5
3-{[(4R)-2,2,5,5-tetramethyl-1,3-dioxan-4-yl]carbonylamino}propionic acid

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid at 20℃; for 16h; Inert atmosphere; Molecular sieve; | 73% |
| With toluene-4-sulfonic acid at 20℃; for 18h; Molecular sieve; | 55% |
| With toluene-4-sulfonic acid at 20℃; Molecular sieve; | 36.8% |
-
-
17612-91-8
1-[(2-aminoethyl)sulfanyl]ethan-1-one hydrochloride

-
-
137-08-6
calcium (R)-pantothenate

-
-
6098-36-8
(R)-S-(2-(3-(2,4-dihydroxy-3,3-dimethylbutanamido)propanamido)ethyl) ethanethioate

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; O-(benzotriazol-1-yl)-N,N,N,N-tetramethyluroniumhexafluorophosphate In N,N-dimethyl-formamide at 20℃; for 1h; | 73% |
-
-
1256166-26-3
(E)-4-(phenylsulfonyl)but-3-en-1-amine hydrochloride

-
-
137-08-6
calcium (R)-pantothenate

-
-
1245941-20-1
(R,E)-2,4-dihydroxy-3,3-dimethyl-N-(3-oxo-3-(4-(phenylsulfonyl)but-3-enylamino)propyl)-butanamide

| Conditions | Yield |
|---|---|
| With N-ethylmorpholine;; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 0 - 20℃; | 64% |
-
-
1256165-12-4
tert-butyl (E)-5-aminopent-2-enoate hydrochloride

-
-
137-08-6
calcium (R)-pantothenate

-
-
1245941-17-6
tert-butyl (R,E)-5-(3-(2,4-dihydroxy-3,3-dimethylbutanamido)propanamido)pent-2-enoate

| Conditions | Yield |
|---|---|
| With N-ethylmorpholine;; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 0 - 20℃; | 59% |
-
-
137-08-6
calcium (R)-pantothenate

-
-
77-76-9
2,2-dimethoxy-propane

-
-
167308-62-5
3-{[(4R)-2,2,5,5-tetramethyl-1,3-dioxan-4-yl]carbonylamino}propionic acid

| Conditions | Yield |
|---|---|
| With oxalic acid In acetone for 20h; Molecular sieve; Inert atmosphere; Reflux; | 52% |

| Conditions | Yield |
|---|---|
| With sulfuric acid Reflux; Inert atmosphere; | 35% |
| With sulfuric acid Reflux; |

| Conditions | Yield |
|---|---|
| With 4-methyl-morpholine; (benzotriazo-1-yloxy)tris(dimethylamino)phosphonium hexafluorophosphate In N,N-dimethyl-formamide at 20℃; | 35% |

| Conditions | Yield |
|---|---|
| With benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane | 20% |
-
-
137-08-6
calcium (R)-pantothenate

-
-
74918-57-3
2-hexyldecanoic acid chloride

| Conditions | Yield |
|---|---|
| With pyridine; dmap In tetrahydrofuran at 50℃; for 6.5h; | 11.4% |

| Conditions | Yield |
|---|---|
| With pyridine; dmap In tetrahydrofuran at 25℃; for 6.5h; | 9.5% |
-
-
51-85-4
2,2'-dithio-bis[ethylamine]

-
-
137-08-6
calcium (R)-pantothenate

-
-
138148-35-3, 141630-68-4, 16816-67-4
D-pantethine

| Conditions | Yield |
|---|---|
| (i) Et3N, oxalic acid, (ii) ClCO2Et, DMF, (iii) /BRN= 1736027/, MeOH; Multistep reaction; |

| Conditions | Yield |
|---|---|
| In water; sodium chloride at 18℃; Product distribution; Irradiation; other temperature; |
-
-
137-08-6
calcium (R)-pantothenate

-
-
108-94-1
cyclohexanone

-
-
215168-67-5
3-[((R)-3,3-Dimethyl-1,5-dioxa-spiro[5.5]undecane-2-carbonyl)-amino]-propionic acid

| Conditions | Yield |
|---|---|
| With 3 Angstroem MS; sulfuric acid; orthoformic acid triethyl ester 1.) 100 deg C, 5 min, 2. ) dioxane, 0 deg C to rt, overnight; Yield given. Multistep reaction; |
-
-
137-08-6
calcium (R)-pantothenate

-
A
-
597-31-9
2,2-dimethyl-3-hydroxypropionaldehyde

-
B
-
124-38-9
carbon dioxide

-
C
-
36321-40-1
calcium beta-alaninate

| Conditions | Yield |
|---|---|
| With sodium hydroxide; potassium permanganate; sodium perchlorate In water at 25℃; for 8h; Kinetics; Further Variations:; Reagents; Temperatures; |


| Conditions | Yield |
|---|---|
| In water byproducts: CaSO4; addn. of 0.01 mol ZnSO4 soln. to a soln. of 0.01 mol Ca-pantothenate in water; filtration of CaSO4; evaporation to dryness in a water bath;; extraction by ethanol with heating in a water bath; evaporation to dryness; washing with ethanol and ether; elem. anal.;; |

| Conditions | Yield |
|---|---|
| In water byproducts: CaSO4; addn. of 0.01 mol CuSO4 soln. to a soln. of 0.01 mol Ca-pantothenate in water; filtration of CaSO4; evaporation to dryness in a water bath;; extraction by ethanol with heating in a water bath; evaporation to dryness; washing with ethanol and ether; elem. anal.;; |

-
-
137-08-6
calcium (R)-pantothenate

| Conditions | Yield |
|---|---|
| In water byproducts: CaSO4; addn. of 0.01 mol NiSO4 soln. to a soln. of 0.01 mol Ca-pantothenate in water; filtration of CaSO4; evaporation to dryness in a water bath;; extraction by ethanol with heating in a water bath; evaporation to dryness; washing with ethanol and ether; elem. anal.;; |

| Conditions | Yield |
|---|---|
| In water addn. of a satd. aq. soln. of 0.02 mol CoCl2 soln. to a soln. of 0.01 mol Ca-pantothenate in water; addn. of acetone; pptn.; separation;; washing with acetone and ether; elem. anal.;; |

| Conditions | Yield |
|---|---|
| In water addn. of a satd. aq. soln. of 0.02 mol NiCl2 soln. to a soln. of 0.01 mol Ca-pantothenate in water; addn. of acetone; pptn.; separation;; washing with acetone and ether; elem. anal.;; |
-
-
1256165-14-6
(E)-5-amino-1-phenylpent-2-en-1-one trifluoroacetate salt

-
-
137-08-6
calcium (R)-pantothenate

-
-
1245941-18-7
(R,E)-2,4-dihydroxy-3,3-dimethyl-N-(3-oxo-3-(5-oxo-5-phenylpent-3-enylamino)propyl)-butanamide

| Conditions | Yield |
|---|---|
| With N-ethylmorpholine;; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 0 - 20℃; | 0.263 g |
-
-
1256166-02-5
(E)-4-(methylsulfonyl)but-3-en-1-amine hydrochloride

-
-
137-08-6
calcium (R)-pantothenate

-
-
1245941-19-8
(R,E)-2,4-dihydroxy-3,3-dimethyl-N-(3-(4-(methylsulfonyl)but-3-enylamino)-3-oxopropyl)-butanamide

| Conditions | Yield |
|---|---|
| With N-ethylmorpholine;; benzotriazol-1-ol; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride In N,N-dimethyl-formamide at 0 - 20℃; | 0.174 g |
-
-
137-08-6
calcium (R)-pantothenate

-
-
1401067-68-2
(4R)-N-(3-((4-((tert-butyldimethylsilyl)oxy)-4-(2-nitrophenyl)butyl)amine)-3-oxo-propyl)-2,2,5,5-tetramethyl-1,3-dioxane-4-carboxamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: oxalic acid / acetone / 20 h / Molecular sieve; Inert atmosphere; Reflux 2: dicyclohexyl-carbodiimide; 4-hydroxybenzotriazole / N,N-dimethyl-formamide / 19 h / 5 - 20 °C / Inert atmosphere View Scheme |
-
-
137-08-6
calcium (R)-pantothenate

-
-
1401067-69-3
(4R)-N-(3-((4-(2-aminophenyl)-4-((tert-butyldimethylsilyl)oxy)-butyl)amino)-3-oxopropyl)-2,2,5,5-tetramethyl-1,3-dioxane-4-carboxamide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: oxalic acid / acetone / 20 h / Molecular sieve; Inert atmosphere; Reflux 2: dicyclohexyl-carbodiimide; 4-hydroxybenzotriazole / N,N-dimethyl-formamide / 19 h / 5 - 20 °C / Inert atmosphere 3: 10% Pd/C; hydrogen / ethyl acetate / 20 h / 20 °C / Inert atmosphere View Scheme |
Calcium pantothenate Chemical Properties
IUPAC Name: calcium 3-[(2,4-dihydroxy-3,3-dimethylbutanoyl)amino]propanoate
Empirical Formula: C18H32CaN2O10
Molecular Weight: 476.5321g/mol
EINECS: 205-278-9
Structure of Calcium pantothenate (CAS NO.137-08-6):
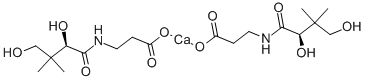
Melting Point: 190 °C
Flash Point: 287.3 °C
Enthalpy of Vaporization: 95.61 kJ/mol
Boiling Point: 551.5 °C at 760 mmHg
Vapour Pressure: 1.84E-14 mmHg at 25°C
Solubility: H2O: 50 mg/mL at 25 °C, clear, nearly colorless
Stability: Stable, but may be moisture or air sensitive. Incompatible with strong acids, strong bases.
Product Categories: Biochemistry;Ca (Calcium) Compounds;Classes of Metal Compounds;Typical Metal Compounds;Vitamins;Nutritional Supplements;Nutritional fortification substances
Canonical SMILES: CC(C)(CO)C(C(=O)NCCC(=O)[O-])O.CC(C)(CO)C(C(=O)NCCC(=O)[O-])O.[Ca+2]
InChI: InChI=1S/2C9H17NO5.Ca/c2*1-9(2,5-11)7(14)8(15)10-4-3-6(12)13;/h2*7,11,14H,3-5H2,1-2H3,(H,10,15)(H,12,13);/q;;+2/p-2
InChIKey: FAPWYRCQGJNNSJ-UHFFFAOYSA-L
Calcium pantothenate Uses
Calcium Pantothenate is used as extender of Nutrition in animal feed.
Calcium pantothenate Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 920mg/kg (920mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES | Proceedings of the Society for Experimental Biology and Medicine. Vol. 45, Pg. 311, 1940. |
| mouse | LD50 | intravenous | 910mg/kg (910mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES | Proceedings of the Society for Experimental Biology and Medicine. Vol. 45, Pg. 311, 1940. |
| mouse | LD50 | oral | 10gm/kg (10000mg/kg) | Drugs in Japan Vol. 6, Pg. 599, 1982. | |
| mouse | LD50 | subcutaneous | 2700mg/kg (2700mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES | Proceedings of the Society for Experimental Biology and Medicine. Vol. 45, Pg. 311, 1940. |
| rat | LD50 | intraperitoneal | 820mg/kg (820mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES | Proceedings of the Society for Experimental Biology and Medicine. Vol. 45, Pg. 311, 1940. |
| rat | LD50 | intravenous | 830mg/kg (830mg/kg) | Drugs in Japan Vol. 6, Pg. 599, 1982. | |
| rat | LD50 | oral | > 10gm/kg (10000mg/kg) | Drugs in Japan Vol. 6, Pg. 599, 1982. | |
| rat | LD50 | subcutaneous | 3400mg/kg (3400mg/kg) | BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY) LUNGS, THORAX, OR RESPIRATION: OTHER CHANGES | Proceedings of the Society for Experimental Biology and Medicine. Vol. 45, Pg. 311, 1940 |
RTECS RU4375000
Calcium pantothenate Safety Profile
Hazard Codes:  Xn
Xn
Risk Statements: 20/21/22-37/38-41-48
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed.
R37/38:Irritating to respiratory system and skin.
R41:Risk of serious damage to the eyes.
R48:Danger of serious damage to health by prolonged exposure.
Safety Statements: 24/25-45-36/37/39-26-22
S24/25:Avoid contact with skin and eyes.
S45:In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
S36/37/39:Wear suitable protective clothing, gloves and eye/face protection.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S22:Do not breathe dust.
WGK Germany: 1
RTECS: RU4375000
F: 3-10
Calcium pantothenate Specification
Calcium pantothenate , its cas register number is 137-08-6. It also can be called Bis(pantothenato)calcium ; Ca-HOPA ;
Calcii pantothenas ; Calcium D-pantothenate ; Calcium d-pantothenate ; Calcium, bis(pantothenato)- ; D-Calcium pantothenate calcium chloride double salt ; Dextro calcium pantothenate ; N-(2,4-Dihydroxy-3,3-dimethylbutyryl)-beta-alanine calcium ; Pancal ; Panthoject ; Pantotenato calcico ; Pantothenate calcium ; Pantothenate de calcium ; Pantothenic acid, calcium salt ; Vitamin B5, calcium salt ; beta-Alanine, N-(2,4-dihydroxy-3,3-dimethyl-1-oxobutyl)-, calcium salt (2:1), (R)- . Calcium pantothenate (CAS NO.137-08-6) is a white crystalline powder.
Related Products
- Calcium
- Calcium valproate
- Calcium (1)-bis(2-hydroxy-4-methylvalerate)
- Calcium (2S)-2-[(4-chlorobenzoyl)amino]-3-(1H-indol-3-yl)propanoate
- Calcium (S)-3-methyl-2-oxovalerate
- Calcium 2,4-dichlorophenolate
- Calcium 2-ethylhexanoate
- Calcium 2-hydroxypropanoate
- Calcium 2-methoxyethoxide
- Calcium 2-oxoglutarate
- 13708-63-9
- 137088-51-8
- 13709-30-3
- 13709-31-4
- 13709-38-1
- 13709-42-7
- 13709-46-1
- 13709-47-2
- 13709-49-4
- 13709-52-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View