-
Name
Cyclobutanemethanol
- EINECS 224-575-4
- CAS No. 4415-82-1
- Article Data46
- CAS DataBase
- Density 0.961 g/cm3
- Solubility Immiscible with water
- Melting Point 68.5 °C
- Formula C5H10O
- Boiling Point 143.499 °C at 760 mmHg
- Molecular Weight 86.1338
- Flash Point 40 °C
- Transport Information UN 1987 3/PG 3
- Appearance Clear colourless liquid
- Safety 16
- Risk Codes 10
-
Molecular Structure
- Hazard Symbols R10:;
- Synonyms (Hydroxymethyl)cyclobutane;Cyclobutylcarbinol;Cyclobutylmethyl alcohol;
- PSA 20.23000
- LogP 0.77880
Synthetic route

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| With methanol; 1,3-disulfonic acid imidazolium hydrogen sulfate at 20℃; for 0.0666667h; Green chemistry; | 98% |

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether for 1.5h; Ambient temperature; | 96% |
| With lithium aluminium tetrahydride In diethyl ether | 72% |
| With lithium aluminium tetrahydride; diethyl ether |
-
-
14924-53-9
ethyl cyclobutylcarboxylate

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether 1) rt, 24 h, 2) reflux, 1.5 h; | 53% |
| With ethanol; sodium | |
| Stage #1: ethyl cyclobutylcarboxylate With diethoxymethylane; zinc diacetate In toluene at 100℃; for 24h; Inert atmosphere; Stage #2: With methanol; potassium hydroxide chemoselective reaction; | 65 %Chromat. |

| Conditions | Yield |
|---|---|
| With sodium; toluene |
-
-
3779-29-1
diethyl cyclobutane-1,1-dicarboxylate

-
A
-
4415-73-0
[1-(hydroxymethyl)cyclobutyl]methanol

-
B
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| With ethanol; sodium | |
| With ethanol; sodium Erwaermen des Reaktionsgemisches auf 130-140grad; |
-
-
89182-08-1
(1-cyclobutenyl)methyl alcohol

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| With methanol; platinum Hydrogenation; |

| Conditions | Yield |
|---|---|
| With diethyl ether; sodium | |
| With lithium aluminium tetrahydride In diethyl ether for 2h; Heating; Yield given; |

| Conditions | Yield |
|---|---|
| (i) B2H6, THF, (ii) H2O2; Multistep reaction; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide at 80℃; for 42h; |
-
-
15963-49-2
3-(hydroxymethyl)cyclobutene

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| With hydrogen; platinum(IV) oxide In methanol |

| Conditions | Yield |
|---|---|
| With tributylphosphine; copper In tetrahydrofuran Product distribution; other reagent and solvent; | |
| With tributylphosphine; copper In tetrahydrofuran Yield given. Yields of byproduct given; | |
| With copper(l) iodide; lithium dihydronaphthylide radical; triphenylphosphine In tetrahydrofuran 1) -45 deg C, 5 min, -23 deg C, 3 h, r.t.; Yield given. Yields of byproduct given; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide; 9-borabicyclo[3.3.1]nonane dimer; dihydrogen peroxide 1.) THF, a) RT, 1.5 h, b) 110 deg C, 96 h, 2.) THF, water, 50 deg C to 60 deg C, 1 h; Yield given. Multistep reaction. Yields of byproduct given; | |
| With sodium hydroxide; 9-borabicyclo[3.3.1]nonane dimer; dihydrogen peroxide 1.) THF, a) RT, 1.5 h, b) 110 deg C, 96 h, 2.) THF, water, 50 deg C to 60 deg C 1 h; Yield given. Multistep reaction. Yields of byproduct given; |

-
-
51108-24-8
cyclobutyl brosylate

-
A
-
4415-82-1
cyclobutanemethanol

-
B
-
138-36-3
p-bromobenzene sulfonic acid

| Conditions | Yield |
|---|---|
| With sodium methylate In ethanol; water at 45℃; Rate constant; | |
| With sodium acetate In formic acid; acetic acid at 45℃; Rate constant; |

| Conditions | Yield |
|---|---|
| With tert.-butyl lithium 1.) Et2O, pentane, -78 deg C, 5 min; 2.) warming; Yield given. Multistep reaction. Yields of byproduct given; | |
| With tert.-butyl lithium In diethyl ether; pentane at -78℃; Product distribution; Effect of various additives or warming.; |


| Conditions | Yield |
|---|---|
| With silver(I) nitrite |
-
-
64-17-5
ethanol

-
-
3779-29-1
diethyl cyclobutane-1,1-dicarboxylate

-
A
-
4415-73-0
[1-(hydroxymethyl)cyclobutyl]methanol

-
B
-
4415-82-1
cyclobutanemethanol

-
-
7664-93-9
sulfuric acid

-
A
-
38834-90-1
cyclobutylmethyl cyclobutanecarboxylate

-
B
-
4415-82-1
cyclobutanemethanol

-
-
5454-82-0
cyclobutylmethylamine hydrochloride

-
A
-
1120-56-5
methylidenecyclobutane

-
B
-
96-41-3
Cyclopentanol

-
C
-
4415-82-1
cyclobutanemethanol

-
D
-
142-29-0
cyclopentene

| Conditions | Yield |
|---|---|
| man erhitzt die Loesung des erhaltenen Nitrils; |

-
-
40628-41-9
methyl cyclobut-1-enecarboxylate

-
A
-
4415-82-1
cyclobutanemethanol

-
B
-
89182-08-1
(1-cyclobutenyl)methyl alcohol

| Conditions | Yield |
|---|---|
| With lithium aluminium tetrahydride In diethyl ether at 25℃; for 0.25h; Reduction; Title compound not separated from byproducts.; |
-
-
23519-90-6
1-cyclobutenecarboxylic acid

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 100 percent / diethyl ether / 0.5 h / 0 °C 2: lithium aluminum hydride / diethyl ether / 0.25 h / 25 °C View Scheme | |
| Multi-step reaction with 2 steps 1: 81 percent / diethyl ether / 48 h / 20 °C 2: lithium aluminum hydride / diethyl ether / 0.25 h / 25 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) KOH / 1.) EtOH, H2O; 2.) reflux 2: thionyl chloride 3: lithium aluminum hydride / diethyl ether / 2 h / Heating View Scheme |
-
-
81469-29-6
1-chloro-1-(hydroxymethyl)cyclobutane

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: methanol. KOH-solution 2: platinum; methanol / Hydrogenation View Scheme |
-
-
35207-71-7
3-chlorocyclobutane-1-carboxylic acid

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: (i) Et2O, (ii) LiAlH4 2: KOtBu / dimethylsulfoxide 3: H2 / PtO2 / methanol View Scheme |
-
-
51816-01-4
cyclobutane-1,1-dicarboxylic acid dichloride

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: SO2Cl2, (PhCO)2O2 / 3 h / Heating 2: H2O 3: 12 h / 180 °C 4: (i) Et2O, (ii) LiAlH4 5: KOtBu / dimethylsulfoxide 6: H2 / PtO2 / methanol View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: SOCl2 / 3 h / Heating 2: SO2Cl2, (PhCO)2O2 / 3 h / Heating 3: H2O 4: 12 h / 180 °C 5: (i) Et2O, (ii) LiAlH4 6: KOtBu / dimethylsulfoxide 7: H2 / PtO2 / methanol View Scheme |
-
-
89639-43-0
3-chloro-1,1-cyclobutanedicarboxylic acid

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 12 h / 180 °C 2: (i) Et2O, (ii) LiAlH4 3: KOtBu / dimethylsulfoxide 4: H2 / PtO2 / methanol View Scheme |
-
-
89582-02-5
3-Chlor-1,1-cyclobutandicarbonsaeure-dichlorid

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: H2O 2: 12 h / 180 °C 3: (i) Et2O, (ii) LiAlH4 4: KOtBu / dimethylsulfoxide 5: H2 / PtO2 / methanol View Scheme |
-
-
15963-47-0
3-chlorocyclobutanemethanol

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: KOtBu / dimethylsulfoxide 2: H2 / PtO2 / methanol View Scheme |
-
-
4415-82-1
cyclobutanemethanol

-
-
124-63-0
methanesulfonyl chloride

-
-
63659-30-3
cyclobutylmethyl methanesulfonate

| Conditions | Yield |
|---|---|
| With triethylamine In chloroform at 20℃; for 2h; | 100% |
| With dmap; triethylamine In dichloromethane at 0 - 20℃; for 3h; | 66% |
| With triethylamine In dichloromethane at -10℃; for 0.333333h; |

| Conditions | Yield |
|---|---|
| With pyridinium chlorochromate In dichloromethane for 1h; Ambient temperature; | 99% |
| Stage #1: cyclobutanemethanol With 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; sodium hydrogencarbonate; potassium bromide In dichloromethane; water at -5℃; for 0.333333h; Stage #2: With sodium hypochlorite In dichloromethane; water at -5 - 0℃; for 0.5h; Stage #3: With potassium dihydrogenphosphate In dichloromethane; water at 20℃; for 0.5h; | 70.4% |
| Stage #1: cyclobutanemethanol With oxalyl dichloride; dimethyl sulfoxide In dichloromethane at -78℃; for 1h; Stage #2: With triethylamine In dichloromethane at -78℃; | 63% |

| Conditions | Yield |
|---|---|
| With 2,4,6-tripropyl-1,3,5,2,4,6-trioxatriphosphinane-2,4,6-trioxide; N-ethyl-N,N-diisopropylamine In tetrahydrofuran; ethyl acetate; toluene at 20℃; for 4h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In dichloromethane | 99% |
-
-
876-27-7
4-chlorophenyl acetate

-
-
4415-82-1
cyclobutanemethanol

-
-
2158-14-7
4-acetamidobenzenesulfonyl azide

| Conditions | Yield |
|---|---|
| Stage #1: 4-chlorophenyl acetate; cyclobutanemethanol With dmap; 1-ethyl-(3-(3-dimethylamino)propyl)-carbodiimide hydrochloride; N-ethyl-N,N-diisopropylamine In dichloromethane for 20h; Stage #2: 4-acetamidobenzenesulfonyl azide With 1,8-diazabicyclo[5.4.0]undec-7-ene In acetonitrile at 0℃; for 16h; | 99% |

-
-
25726-04-9
phenylglyoxylyl chloride

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| With 1H-imidazole In dichloromethane at 0 - 20℃; Inert atmosphere; | 98% |

| Conditions | Yield |
|---|---|
| With tert.-butylhydroperoxide In dimethyl sulfoxide at 25 - 35℃; for 40h; Irradiation; Green chemistry; | 98% |
| With tert.-butylhydroperoxide at 110℃; for 12h; | 74% |
-
-
4415-82-1
cyclobutanemethanol

-
-
98-59-9
p-toluenesulfonyl chloride

-
-
13295-53-9
cyclobutylmethyl 4-methylbenzenesulfonate

| Conditions | Yield |
|---|---|
| With dmap In dichloromethane at 0 - 25℃; | 97% |
| In pyridine at 0℃; for 24h; | 92% |
| In pyridine at -15℃; | 92% |
-
-
4415-82-1
cyclobutanemethanol

-
-
16000-39-8
1-cyano-2-methoxynaphthalene

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In 1,4-dioxane at 20℃; for 16h; Inert atmosphere; Sealed tube; Glovebox; | 96% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid at 120℃; for 16h; Sealed tube; | 95% |

| Conditions | Yield |
|---|---|
| With 1,3-disulfonic acid imidazolium hydrogen sulfate In neat (no solvent) at 20℃; for 0.05h; Green chemistry; | 94% |

| Conditions | Yield |
|---|---|
| With caesium carbonate In dimethyl sulfoxide at 80℃; for 5h; | 94% |

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran; mineral oil at 20 - 50℃; for 2.5h; | 91% |
-
-
57125-20-9
ditungsten hexa-tert-butoxide

-
-
4415-82-1
cyclobutanemethanol

-
-
120853-44-3
W4(OCH2-c-Bu)12(HOCH2-c-Bu)

| Conditions | Yield |
|---|---|
| In hexane byproducts: t-C4H9OH; under N2, adding cyclobutylmethanol dropwise to soln. of W2(O-t-Bu)6, room temp., stirring, 2 h; evapn. of volatile components in vac., dissolving in 1,2-dimethoxyethane, cooling to -20°C, 16 h, filtration at 0°C (2nd crop from filtrate); elem. anal.; | 90.2% |

| Conditions | Yield |
|---|---|
| Stage #1: cyclobutanemethanol With sodium hydride In N,N-dimethyl-formamide at 20℃; for 1h; Stage #2: 3-chloropyrazin-2-amine In N,N-dimethyl-formamide at 100℃; for 20h; | 89% |
-
-
674-82-8
4-methyleneoxetan-2-one

-
-
4415-82-1
cyclobutanemethanol

-
-
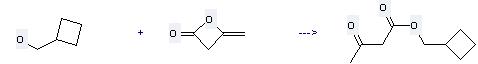
103785-41-7
cyclobutylmethyl acetoacetate

| Conditions | Yield |
|---|---|
| With sodium hydride at 70 - 80℃; for 2h; | 88% |
-
-
4415-82-1
cyclobutanemethanol

-
-
474295-85-7
methyl (3S)-3-{[(tert-butoxy)carbonyl]amino}-3-(4-hydroxyphenyl)propanoate

| Conditions | Yield |
|---|---|
| With triphenylphosphine; diethylazodicarboxylate In tetrahydrofuran at 20 - 35℃; Mitsunobu Displacement; Inert atmosphere; | 87% |
-
-
4415-82-1
cyclobutanemethanol


| Conditions | Yield |
|---|---|
| With caesium carbonate In tetrahydrofuran at 50℃; for 2h; | 86% |
-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| With formic acid; isocyanate de chlorosulfonyle; triethylamine In dichloromethane at 0 - 25℃; Schlenk technique; Inert atmosphere; | 86% |
-
-
944804-88-0
tert-butyl N-(4-bromo-1,3-thiazol-2-yl)carbamate

-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| With di-isopropyl azodicarboxylate; triphenylphosphine In tetrahydrofuran at 0 - 40℃; for 1.5h; Inert atmosphere; | 85% |

| Conditions | Yield |
|---|---|
| With 2-Hydroxymethylpyridine; dodecacarbonyl-triangulo-triruthenium at 170℃; for 4h; | 84% |


| Conditions | Yield |
|---|---|
| With 2,2,6,6-tetramethyl-piperidine; bis(1,5-cyclooctadiene)nickel (0); acetone; [2-((diphenylphospino)methyl)-2-methyl-1,3-propanediyl]bis[diphenylphosphine] In toluene at 130℃; for 20h; Inert atmosphere; | 84% |
-
-
4415-82-1
cyclobutanemethanol

-
-
848666-52-4
cyclobutylmethoxyacetic acid

| Conditions | Yield |
|---|---|
| Stage #1: cyclobutanemethanol With sodium hydride In tetrahydrofuran at -15 - 25℃; for 1h; Stage #2: sodium monochloroacetic acid In dimethyl sulfoxide at 20℃; for 20h; Stage #3: With hydrogenchloride In water | 83% |
| Stage #1: cyclobutanemethanol With sodium hydride In tetrahydrofuran at -15 - 25℃; for 1h; pH=2; Stage #2: sodium monochloroacetic acid In tetrahydrofuran; dimethyl sulfoxide at 20℃; for 2h; | 83% |
-
-
4415-82-1
cyclobutanemethanol

-
-
91923-30-7
methyl cyclohexanethionocarboxylate

| Conditions | Yield |
|---|---|
| With sodium hydride at 75 - 80℃; for 0.5h; | 83% |

| Conditions | Yield |
|---|---|
| With caesium carbonate In N,N-dimethyl-formamide at 60℃; for 12h; Inert atmosphere; | 81.5% |
-
-
4415-82-1
cyclobutanemethanol

| Conditions | Yield |
|---|---|
| With C22H36IrNP(1+)*CF3O3S(1-); potassium hydroxide In toluene at 120℃; for 40h; | 81% |

-
-
4415-82-1
cyclobutanemethanol


| Conditions | Yield |
|---|---|
| With tetrakis(actonitrile)copper(I) hexafluorophosphate In dichloromethane at 25℃; for 2h; Inert atmosphere; Sealed tube; Microwave irradiation; | 81% |


| Conditions | Yield |
|---|---|
| With t-butylnitrite; C24H20N2O4Ru; sodium t-butanolate In tert-butyl alcohol at 130℃; for 24h; Schlenk technique; Inert atmosphere; | 81% |
Cyclobutanemethanol Specification
The Cyclobutanemethanol, with the CAS registry number 4415-82-1, is also known as Hydroxymethylcyclobutane. It belongs to the product categories of API Intermediates; Cyclobutanes & Cyclobutenes; Simple 4-Membered Ring Compounds; Cycloalkanes; Alcohols; C2 to C6; Oxygen Compounds. Its EINECS registry number is 224-575-4. This chemical's molecular formula is C5H10O and molecular weight is 86.13. What's more, both its IUPAC name and systematic name are the same which is called Cyclobutylmethanol. When you are dealing with this chemical, you should be very careful. This chemical is flammable. Therefore, you should keep away from sources of ignition.
Physical properties about Cyclobutanemethanol are: (1)ACD/LogP: 0.737; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0.74; (4)ACD/LogD (pH 7.4): 0.74; (5)ACD/BCF (pH 5.5): 2.14; (6)ACD/BCF (pH 7.4): 2.14; (7)ACD/KOC (pH 5.5): 60.00; (8)ACD/KOC (pH 7.4): 60.00; (9)#H bond acceptors: 1; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 20.23 Å2; (13)Index of Refraction: 1.463; (14)Molar Refractivity: 24.676 cm3; (15)Molar Volume: 89.549 cm3; (16)Polarizability: 9.782×10-24cm3; (17)Surface Tension: 37.253 dyne/cm; (18)Density: 0.962 g/cm3; (19)Flash Point: 40 °C; (20)Enthalpy of Vaporization: 44.341 kJ/mol; (21)Boiling Point: 143.499 °C at 760 mmHg; (22)Vapour Pressure: 2.137 mmHg at 25 °C.
Preparation of Cyclobutanemethanol: this chemical can be prepared by cyclobutanecarboxylic acid. This reaction needs reagent LiAlH4 and solvent diethyl ether at ambient temperature. The reaction time is 1.5 hours. The yield is 96 %.

Uses of Cyclobutanemethanol: (1) it is used as pharmaceutical intermediates; (2) it is used to produce other chemicals. For example, it can react with diketene to get cyclobutylmethyl acetoacetate. The reaction occurs with reagent NaH in oil at temperature of 70-80 °C. The reaction time is 2 hours. The yield is 88 %.
You can still convert the following datas into molecular structure:
(1) SMILES: OCC1CCC1
(2) InChI: InChI=1S/C5H10O/c6-4-5-2-1-3-5/h5-6H,1-4H2
(3) InChIKey: WPOPOPFNZYPKAV-UHFFFAOYSA-N
Related Products
- Cyclobutanemethanol
- Cyclobutanemethanol,1-[(methylamino)methyl]-
- 4415-83-2
- 4415-87-6
- 441715-01-1
- 441717-40-4
- 441774-09-0
- 4417-81-6
- 441798-33-0
- 4418-26-2
- 4418-61-5
- 4418-66-0
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View