-
Name
Diethyl succinate
- EINECS 204-612-0
- CAS No. 123-25-1
- Article Data208
- CAS DataBase
- Density 1.039 g/cm3
- Solubility
- Melting Point -20 °C
- Formula C8H14O4
- Boiling Point 218.4 °C at 760 mmHg
- Molecular Weight 174.197
- Flash Point 99.5 °C
- Transport Information
- Appearance clear liquid
- Safety 26-36-24/25-22
- Risk Codes 36/37/38
-
Molecular Structure
-
Hazard Symbols
 Xi
Xi
- Synonyms Butanedioicacid, diethyl ester (9CI);Succinic acid, diethyl ester (6CI,8CI);Butandioicacid diethyl ester;Diethyl butanedioate;
- PSA 52.60000
- LogP 0.89280
Synthetic route

| Conditions | Yield |
|---|---|
| With hexacarbonyl molybdenum; phenylsilane In tetrahydrofuran for 4.5h; Heating; | 100% |
| With 1,2,2,3,4,4-hexamethylphosphetane 1-oxide; phenylsilane; water In toluene at 80℃; for 24h; Catalytic behavior; Solvent; Temperature; Reagent/catalyst; Schlenk technique; Inert atmosphere; | 99% |
| With acetic acid; zinc for 0.5h; Ambient temperature; sonication; | 98% |

| Conditions | Yield |
|---|---|
| With 5% Pd/C; hydrogen In ethanol at 20℃; for 0.5h; chemoselective reaction; | 100% |
| With hydrogen In methanol at 24.84℃; under 760.051 Torr; | |
| With silica gel supported sulfonic acid In acetonitrile at 20℃; Reagent/catalyst; Electrochemical reaction; | 75 %Spectr. |

| Conditions | Yield |
|---|---|
| With hydrogen; palladium on activated charcoal In ethyl acetate at 80℃; under 2585.81 Torr; for 0.05h; microwave irradiation; | 99% |
| With magnesium chloride electrolysis: large surface area gold wire electrode; 150 W cm-2 ultrasound intensity; Yield given; | |
| With water; zinc In tetrahydrofuran; methanol for 3h; Heating; | 100 % Spectr. |

| Conditions | Yield |
|---|---|
| With acetic acid; zinc for 2h; Ambient temperature; sonication; | 98% |
| With Decaborane; palladium on activated charcoal In methanol at 25℃; for 0.3h; Reduction; | 98% |
| With 1,3-dimethyl-2-phenyl-2,3-dihydro-1H-7-methylbenzo-[d]imidazole; [Zr(2,6-bis(5-methyl-3-phenyl-1H-pyrrol-2-yl)pyridine)2] In benzene-d6 for 8h; UV-irradiation; | 98% |

| Conditions | Yield |
|---|---|
| With alumina methanesulfonic acid at 80℃; for 0.133333h; Microwave irradiation; | 97% |
| With Fe(SO4)3 * xH2O In toluene for 3h; Heating; | 95% |
| With N-dodecanoyl-N-methyl-1-glucamine In water at 45℃; for 6h; Green chemistry; | 87% |

| Conditions | Yield |
|---|---|
| With chloro-trimethyl-silane at 40℃; for 5h; | 96% |
| With chloro-trimethyl-silane at 20 - 40℃; for 5h; Inert atmosphere; | 96% |
-
-
144150-81-2
1,2-bis(ethoxycarbonyl)vinyl toluene-p-sulfonate

-
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With sodium tetrahydroborate; nickel dichloride In methanol ice-cooling; | 92% |
-
-
141-05-9
Diethyl maleate

-
A
-
24766-10-7
diethyl 2-n-butylsuccinate

-
B
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With aluminium trichloride; lithium di-n-butylcuprate In diethyl ether at -65 - -40℃; for 1h; | A 1.8% B 92% |
| With lithium di-n-butylcuprate In diethyl ether at -65 - -40℃; for 1h; Product distribution; in the presence of AlCl3 as well; other solvents; | A 1% B 57% |
-
-
1114-31-4
diethyl meso-2,3-dibromosuccinate

-
A
-
623-91-6
diethyl Fumarate

-
B
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium tetrahydroborate; 1,2-di(thiophen-2-yl)ditelluride In ethanol; water at 5℃; | A 90% B n/a |

| Conditions | Yield |
|---|---|
| Stage #1: C12H24O4Si With oxalyl dichloride; N,N-dimethyl-formamide Stage #2: ethanol With pyridine | 83% |

| Conditions | Yield |
|---|---|
| With tetrabutyl ammonium fluoride | 83% |
| With tetrabutyl ammonium fluoride In tetrahydrofuran at 0℃; |
-
-
64-17-5
ethanol

-
-
539-88-8
4-oxopentanoic acid ethyl ester

-
A
-
95-92-1
oxalic acid diethyl ester

-
B
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| Stage #1: 4-oxopentanoic acid ethyl ester With acetic acid at 120℃; for 6h; Stage #2: ethanol for 6h; Reflux; | A 8.7% B 80% |

-
-
110-15-6
succinic acid

-
-
21882-77-9, 53472-13-2
(2,2-diethoxyvinylidene)triphenylphosphorane

-
A
-
1099-45-2
ethyl (triphenylphosphoranylidene)acetate

-
B
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| In tetrahydrofuran for 3h; Ambient temperature; | A n/a B 76% |


| Conditions | Yield |
|---|---|
| sulfuric acid In ethanol Heating; | 73% |
| With mineral acid |
-
-
930-68-7
cyclohexenone

-
-
626-62-0
1-iodocyclohexane

-
-
153094-32-7
O,O'-bistributyltin benzopinacolate

-
-
623-91-6
diethyl Fumarate

-
A
-
21086-38-4
3-(hydroxydiphenylmethyl)cyclohexan-1-one

-
B
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| In benzene at 80℃; for 8h; | A 45% B 72% |


| Conditions | Yield |
|---|---|
| With N,N,N,N,N,N-hexamethylphosphoric triamide; samarium diiodide; Trimethylacetic acid In tetrahydrofuran at 20 - 22℃; for 2h; | 71% |

| Conditions | Yield |
|---|---|
| With N,N-diethyl-N-bromoamine In tetrahydrofuran; diethyl ether at 20 - 25℃; Reagent/catalyst; Cooling; Inert atmosphere; | 71% |

-
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In water; acetonitrile at 40℃; for 16h; Sealed tube; Schlenk technique; Inert atmosphere; Irradiation; | 69% |
-
-
141-05-9
Diethyl maleate

-
A
-
24766-10-7
diethyl 2-n-butylsuccinate

-
B
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| In diethyl ether at -65 - -40℃; for 1h; | A 9% B 66% |

-
-
408332-88-7
Diethyl tartrate

-
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With phosphorus; hydrogen iodide In water; toluene at 80℃; for 1.5h; Inert atmosphere; | 65% |
-
-
105-36-2
ethyl bromoacetate

-
-
121745-78-6
O-tert-butyl O-ethyl ketene acetal

-
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With 2,4,6-trimethyl-pyridine; dilauryl peroxide In cyclohexane for 3h; Heating; | 64% |
-
-
10170-69-1
dimanganese decacarbonyl

-
-
105-36-2
ethyl bromoacetate

-
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| In chloroform-d1 Irradiation (UV/VIS); (N2 or Ar); 250-W sun lamp, 3 h; colorless solution is filtered, (1)H-NMR; | 64% |
-
-
61844-36-8
5-diazo-4-oxo-valeric acid ethyl ester

-
-
3220-49-3
phenyl copper

-
A
-
20416-11-9
Ethyl 4-oxo-5-phenyl-pentanoate

-
B
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| for 1h; Ambient temperature; | A 62% B n/a |

-
-
109-97-7
pyrrole

-
-
61844-36-8
5-diazo-4-oxo-valeric acid ethyl ester

-
A
-
105886-77-9
ethyl 4-oxo-5-(2-pyrrolyl)-pentanoate

-
B
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With copper In benzene for 0.25h; | A 61% B n/a |


| Conditions | Yield |
|---|---|
| With 1,2-diamino-benzene at 140℃; for 4h; | 61% |
-
-
3859-41-4
1,3-cyclopentadione

-
-
64-17-5
ethanol

-
A
-
1070-34-4
butanedioic acid, monoethyl ester

-
B
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With potassium peroxomonosulfate at 20℃; for 18h; | A 25% B 61% |


| Conditions | Yield |
|---|---|
| With carbonic acid dimethyl ester at 180℃; under 10343.2 Torr; for 5h; Reagent/catalyst; Temperature; Inert atmosphere; | 60.2% |
-
-
623-91-6
diethyl Fumarate

-
A
-
1187-74-2
diethyl 2-acetylmethyl-1,4-butanedioate

-
C
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With sulfur dioxide; N-ethyl-N,N-diisopropylamine In N,N-dimethyl acetamide at 80℃; for 1.25h; | A 6.2% B 10% C 58% |

-
-
7087-68-5
N-ethyl-N,N-diisopropylamine

-
-
623-91-6
diethyl Fumarate

-
A
-
1187-74-2
diethyl 2-acetylmethyl-1,4-butanedioate

-
C
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With sulfur dioxide In N,N-dimethyl acetamide at 80℃; for 3.5h; | A 16% B 14% C 56% |


| Conditions | Yield |
|---|---|
| With sulfur dioxide; N-ethyl-N,N-diisopropylamine In N,N-dimethyl acetamide at 80℃; for 2.5h; | A 10% B 55% |
-
-
109-94-4
formic acid ethyl ester

-
-
123-25-1
succinic acid diethyl ester

-
-
5472-38-8
diethyl 2-formylbutanedioate

| Conditions | Yield |
|---|---|
| With sodium In diethyl ether at 40℃; for 5h; | 100% |
| With sodium In diethyl ether at 40℃; for 5h; | 100% |
| With sodium hydride In toluene at 20℃; for 16h; Reagent/catalyst; | 95.1% |

| Conditions | Yield |
|---|---|
| With C56H70Cl3N10Ru2(1+)*F6P(1-); potassium tert-butylate; hydrogen In tetrahydrofuran; dodecane at 70℃; under 37503.8 Torr; for 16h; Inert atmosphere; Glovebox; Autoclave; | 100% |
| With C30H34Cl2N2P2Ru; potassium methanolate; hydrogen In tetrahydrofuran at 100℃; under 38002.6 - 76005.1 Torr; for 15h; Glovebox; Autoclave; | 91% |
| With ethanol; sodium |

| Conditions | Yield |
|---|---|
| Stage #1: thiophene-2-carbaldehyde; succinic acid diethyl ester With potassium tert-butylate In tert-butyl alcohol for 1.5h; Heating / reflux; Stage #2: With hydrogenchloride; water In tert-butyl alcohol pH=2; | 100% |
-
-
112758-40-4
3-methyl-1H-pyrazole-4-carbaldehyde

-
-
123-25-1
succinic acid diethyl ester

-
-
200264-80-8
3-(ethoxycarbonyl)-4-(3-methyl-1H-pyrazol-4-yl)but-3-enoic acid

| Conditions | Yield |
|---|---|
| Stage #1: 3-methyl-1H-pyrazole-4-carbaldehyde; succinic acid diethyl ester With potassium tert-butylate; tert-butyl alcohol at 80℃; for 4h; Stage #2: With hydrogenchloride In water pH=~ 2; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: 3-ethyl-1H-pyrazole-4-carbaldehyde; succinic acid diethyl ester With potassium tert-butylate; tert-butyl alcohol at 80℃; for 3h; Stage #2: With hydrogenchloride In water pH=~ 2; | 100% |

-
-
123-25-1
succinic acid diethyl ester

-
-
1259401-36-9
C24H26O6

| Conditions | Yield |
|---|---|
| Stage #1: 4-Benzoyl-7-methoxy-2,2-dimethyl-2,3-dihydrobenzofuran; succinic acid diethyl ester With potassium tert-butylate In tetrahydrofuran at 55℃; for 1h; Stobbe Condensation; Stage #2: With hydrogenchloride In tetrahydrofuran; water | 100% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at 55℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: C15H14OS2; succinic acid diethyl ester With potassium tert-butylate In tetrahydrofuran at 55℃; Stage #2: With hydrogenchloride In tetrahydrofuran; water | 100% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at 55℃; for 1h; Stobbe Condensation; | 100% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at 55℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at 55℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at 55℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| Stage #1: C22H20O2S; succinic acid diethyl ester With potassium tert-butylate In tetrahydrofuran for 1h; Stage #2: With hydrogenchloride In water | 100% |
-
-
611-97-2
bis(p-methylphenyl)-methanone

-
-
123-25-1
succinic acid diethyl ester

-
-
95698-26-3
2-Di-p-tolylmethylene-succinic acid 1-ethyl ester

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tetrahydrofuran at 55℃; for 1h; | 100% |

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tert-butyl alcohol at 110℃; for 3h; Inert atmosphere; | 99% |
| With potassium tert-butylate In tert-butyl alcohol for 1.5h; Stobbe condensation; Heating; |

| Conditions | Yield |
|---|---|
| With hydrazine hydrate In methanol for 8h; Reflux; | 98% |
| With hydrazine hydrate In ethanol for 12h; Reflux; | 65% |
| With ethanol; hydrazine hydrate |
-
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran at -78℃; for 0.25h; | 98% |
-
-
120-14-9
3,4-dimethoxy-benzaldehyde

-
-
123-25-1
succinic acid diethyl ester

-
-
126274-95-1
2-(3,4-dimethoxybenzylidene)butanedioic acid monoethyl ester

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tert-butyl alcohol for 3h; Heating; | 98% |
| With potassium tert-butylate In tert-butyl alcohol at 50℃; for 5h; |
-
-
120-14-9
3,4-dimethoxy-benzaldehyde

-
-
123-25-1
succinic acid diethyl ester

-
-
182572-57-2
(E)-4-(3,4-dimethoxyphenyl)-3-(ethoxycarbonyl)but-3-enoic acid

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol for 4h; Stobbe Condensation; Inert atmosphere; Reflux; | 98% |
| With sodium ethanolate Stobbe Condensation; Reflux; | 96% |
| With sodium t-butanolate In tert-butyl alcohol Reflux; | 56% |
| With potassium tert-butylate In butan-1-ol at 20℃; for 3h; Stobbe condensation; |

| Conditions | Yield |
|---|---|
| Stage #1: 3,4-dichlorobenzophenone; succinic acid diethyl ester In tert-butyl alcohol at 25 - 30℃; Stage #2: With sodium hydride In tert-butyl alcohol at 85 - 90℃; for 4h; Stage #3: With hydrogenchloride; water In hexane; ethyl acetate | 98% |

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol for 4h; Stobbe Condensation; Inert atmosphere; Reflux; | 98% |
| With sodium ethanolate for 4h; Stobbe condensation; Reflux; |

| Conditions | Yield |
|---|---|
| With sodium ethanolate In ethanol for 4h; Stobbe Condensation; Inert atmosphere; Reflux; | 98% |
-
-
123-25-1
succinic acid diethyl ester

| Conditions | Yield |
|---|---|
| With potassium tert-butylate In tert-butyl alcohol for 4h; Stobbe condensation; Heating; | 97% |
-
-
90009-92-0
ethyl [3H]formate

-
-
123-25-1
succinic acid diethyl ester

-
-
1035845-94-3
diethyl ([3H]formyl)succinate

| Conditions | Yield |
|---|---|
| With sodium ethanolate In toluene at 20℃; | 97% |
-
-
7311-34-4
3,5-dimethoxybenzaldehdye

-
-
123-25-1
succinic acid diethyl ester

-
-
607356-91-2
(E)-4-(3,5-dimethoxyphenyl)-3-(ethoxycarbonyl)but-3-enoic acid

| Conditions | Yield |
|---|---|
| With ethanol; sodium hydride In toluene; mineral oil at 0 - 20℃; Inert atmosphere; | 97% |
Diethyl succinate Chemical Properties
IUPAC Name: Diethyl butanedioate
Synonyms of Diethyl succinate (CAS NO.123-25-1): Butanedioic acid, diethyl ester ; Diethyl ethanedicarboxylate ; Diethylester kyseliny jantarove ; Ethyl succinate
CAS NO: 123-25-1
Molecular Formula: C8H14O4
Molecular Weight: 174.20
Molecular Structure: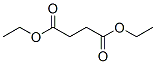
EINECS: 204-612-0
H bond acceptors: 4
H bond donors: 0
Freely Rotating Bonds: 7
Polar Surface Area: 52.6 Å2
Melting point: -20°C
Boiling Point: 218.4 °C at 760 mmHg
Flash Point: 99.5 °C
Index of Refraction: 1.422
Molar Refractivity: 42.66 cm3
Molar Volume: 167.5 cm3
Surface Tension: 32.4 dyne/cm
Density: 1.039 g/cm3
Enthalpy of Vaporization: 45.48 kJ/mol
Vapour Pressure: 0.126 mmHg at 25°C
Appearance: clear liquid
Product Categories of Diethyl succinate (CAS NO.123-25-1): Pharmaceutical Intermediates;Organics;SuccinicSeries;Fatty Acid Esters (Plasticizer);Functional Materials;Plasticizer;ester Flavor;Alpha Sort;D;Volatiles/ Semivolatiles;Plasticizers;Polymer Additives;Polymer Science;Alphabetical Listings;C-D;Flavors and Fragrances
Diethyl succinate Uses
Diethyl succinate (CAS NO.123-25-1) is used as a food flavoring agent, solvent, organic synthesis intermediates, gas chromatography stationary phase,and it is also used in the plastics industry.
Diethyl succinate Production
Diethyl succinate (CAS NO.123-25-1) is derived from the esterification of Succinic acid with Ethanol.
Diethyl succinate Toxicity Data With Reference
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| rat | LD50 | oral | 8530mg/kg (8530mg/kg) | AMA Archives of Industrial Hygiene and Occupational Medicine. Vol. 4, Pg. 119, 1951. |
Diethyl succinate Consensus Reports
Reported in EPA TSCA Inventory.
Diethyl succinate Safety Profile
Safety Information about Diethyl succinate (CAS NO.123-25-1):
Hazard Codes:  Xi
Xi
Risk Statements: 36/37/38
R36/37/38: Irritating to eyes, respiratory system and skin.
Safety Statements: 26-36-24/25-22
S22: Do not breathe dust.
S24/25: Avoid contact with skin and eyes.
S26: In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36: Wear suitable protective clothing.
WGK Germany: 2
RTECS: WM7400000
HS Code: 29171990
Mildly toxic by ingestion. A skin and eye irritant. Combustible liquid. Reaction with ethyl trifluoroacetate + sodium hydride may cause a fire or explosion. When heated to decomposition it emits acrid smoke and irritating fumes. See also ESTERS.
Diethyl succinate Specification
General Information: As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. Will burn if involved in a fire. Combustible liquid.
Extinguishing Media: Use agent most appropriate to extinguish fire.
Handling: Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Storage: Keep away from sources of ignition. Store in a cool, dry place. Store in a tightly closed container.
Related Products
- Diethyl (1-phenylpropyl)malonate
- Diethyl (2-(cyclohexylamino)vinyl)phosphonate
- Diethyl (2-(diethoxymethylsilyl)ethyl)phosphonate
- Diethyl (2-(triethoxysilyl)ethyl)phosphonate
- Diethyl (2,4,6-trifluorophenyl)malonate
- Diethyl (2-oxopropyl)phosphonate
- Diethyl (2-thienylmethyl)phosphonate
- Diethyl (4-cyanobenzyl)phosphonate
- Diethyl (4-nitrobenzyl)phosphonate
- Diethyl (beta,gamma-epoxypropyl)phosphonate
- 123252-96-0
- 123253-05-4
- 12325-31-4
- 12325-59-6
- 12325-81-4
- 123259-33-6
- 123-26-2
- 123266-59-1
- 123266-63-7
- 123273-02-9
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View