-
Name
O,O,S-trimethyl phosphorothioate
- EINECS
- CAS No. 152-20-5
- Article Data36
- CAS DataBase
- Density 1.211g/cm3
- Solubility
- Melting Point
- Formula C3H9 O3 P S
- Boiling Point 169°Cat760mmHg
- Molecular Weight 156.142
- Flash Point 56°C
- Transport Information
- Appearance
- Safety Poison by ingestion, intravenous, and intraperitoneal routes. An experimental teratogen. Other experimental reproductive effects. When heated to decomposition it emits toxic fumes of POx and SOx.
- Risk Codes
-
Molecular Structure
- Hazard Symbols
- Synonyms HC 7901;Methyl phosphorothioate ((MeO)2(MeS)PO); O,O,S-Trimethyl phosphorothioate;O,O,S-Trimethyl phosphorothiolate; O,O,S-Trimethyl thiophosphate; O,O-DimethylS-methyl phosphorothioate; O,O-Dimethyl S-methyl thiophosphate
- PSA 70.64000
- LogP 1.75030
Synthetic route
-
-
67-56-1
methanol

-
-
71216-76-7
Benzyl-thiophosphoramidic acid O,S-dimethyl ester

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With boron trifluoride methanol complex 1) 0 deg C, 1h, 2) RT, overnight; | 86% |
-
-
67-56-1
methanol

-
-
65960-94-3, 87000-69-9, 25218-42-2
N,N-dimethyl O,S-dimethyl phosphoramidothioate

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With boron trifluoride methanol complex 1) 0 deg C, 1h, 2) RT, overnight; | 80% |

| Conditions | Yield |
|---|---|
| With boron trifluoride methanol complex 1) 0 deg C, 1h, 2) RT, overnight; | 74% |
-
-
96-36-6, 868-85-9
methyl phosphite

-
-
67-68-5
dimethyl sulfoxide

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With 1,3,5-trichloro-2,4,6-triazine In acetonitrile at 80℃; for 1h; | 71% |
-
-
133984-31-3
2,6-lutidinium O,O-dimethyl phosphorothioate

-
-
74-88-4
methyl iodide

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| In dichloromethane for 0.5h; | 63% |

| Conditions | Yield |
|---|---|
| With monoperoxyphthalic acid; magnesium salt In dichloromethane for 12h; Heating; | 51% |
-
-
1112-38-5
O,O-dimethyl S-hydrogen phosphorothioate

-
-
538-75-0
dicyclohexyl-carbodiimide

-
A
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
B
-
152-18-1
O,O,O-trimethylthiophosphate

-
C
-
1212-29-9
1,3-dicyclohexylthiourea

| Conditions | Yield |
|---|---|
| In diethyl ether; Petroleum ether Mechanism; Product distribution; Ambient temperature; solvents: diethylether, accetonitrile; | A n/a B n/a C 35% |

-
-
186581-53-3, 908094-01-9
diazomethane

-
-
42576-53-4
O,S-dimethyl phosphorothiolate

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With 1,4-dioxane; diethyl ether | |
| Methylation; |
-
-
556-64-9
methyl thiocyanate

-
-
96-36-6, 868-85-9
methyl phosphite

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With sodium; benzene |

| Conditions | Yield |
|---|---|
| at 100℃; | |
| With water | |
| With methanol |
-
-
556-64-9
methyl thiocyanate

-
-
2303-76-6, 118080-94-7
sodium diethyl phosphite

-
-
108-88-3
toluene

-
A
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
B
-
143-33-9
sodium cyanide

-
-
67-56-1
methanol

-
-
23754-87-2
Natriumsalz des Thiophosphorsaeure-O,O-dimethylesters

-
-
77-78-1
dimethyl sulfate

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
-
152-18-1
O,O,O-trimethylthiophosphate

-
-
74-88-4
methyl iodide

-
A
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
B
-
2181-42-2
trimethylsulphonium iodide

| Conditions | Yield |
|---|---|
| at 100℃; |

-
-
67-56-1
methanol

-
-
71216-76-7
Benzyl-thiophosphoramidic acid O,S-dimethyl ester

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With boron trifluoride methanol complex 1) 0 deg C, 1h, 2) RT, overnight; | 86% |
-
-
67-56-1
methanol

-
-
65960-94-3, 87000-69-9, 25218-42-2
N,N-dimethyl O,S-dimethyl phosphoramidothioate

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With boron trifluoride methanol complex 1) 0 deg C, 1h, 2) RT, overnight; | 80% |

| Conditions | Yield |
|---|---|
| With boron trifluoride methanol complex 1) 0 deg C, 1h, 2) RT, overnight; | 74% |
-
-
96-36-6, 868-85-9
methyl phosphite

-
-
67-68-5
dimethyl sulfoxide

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With 1,3,5-trichloro-2,4,6-triazine In acetonitrile at 80℃; for 1h; | 71% |
-
-
133984-31-3
2,6-lutidinium O,O-dimethyl phosphorothioate

-
-
74-88-4
methyl iodide

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| In dichloromethane for 0.5h; | 63% |

| Conditions | Yield |
|---|---|
| With monoperoxyphthalic acid; magnesium salt In dichloromethane for 12h; Heating; | 51% |
-
-
1112-38-5
O,O-dimethyl S-hydrogen phosphorothioate

-
-
538-75-0
dicyclohexyl-carbodiimide

-
A
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
B
-
152-18-1
O,O,O-trimethylthiophosphate

-
C
-
1212-29-9
1,3-dicyclohexylthiourea

| Conditions | Yield |
|---|---|
| In diethyl ether; Petroleum ether Mechanism; Product distribution; Ambient temperature; solvents: diethylether, accetonitrile; | A n/a B n/a C 35% |

-
-
186581-53-3, 908094-01-9
diazomethane

-
-
42576-53-4
O,S-dimethyl phosphorothiolate

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With 1,4-dioxane; diethyl ether | |
| Methylation; |
-
-
556-64-9
methyl thiocyanate

-
-
96-36-6, 868-85-9
methyl phosphite

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With sodium; benzene |

| Conditions | Yield |
|---|---|
| at 100℃; | |
| With water | |
| With methanol |
-
-
556-64-9
methyl thiocyanate

-
-
2303-76-6, 118080-94-7
sodium diethyl phosphite

-
-
108-88-3
toluene

-
A
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
B
-
143-33-9
sodium cyanide

-
-
67-56-1
methanol

-
-
23754-87-2
Natriumsalz des Thiophosphorsaeure-O,O-dimethylesters

-
-
77-78-1
dimethyl sulfate

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
-
152-18-1
O,O,O-trimethylthiophosphate

-
-
74-88-4
methyl iodide

-
A
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
B
-
2181-42-2
trimethylsulphonium iodide

| Conditions | Yield |
|---|---|
| at 100℃; |

-
-
186581-53-3, 908094-01-9
diazomethane

-
-
1112-38-5
O,O-dimethyl S-hydrogen phosphorothioate

-
A
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
B
-
152-18-1
O,O,O-trimethylthiophosphate

-
-
12127-74-1
tetramethylammonium O,O-dimethyl phosphorothioate

-
-
152-20-5
O,O,S-trimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With triethylmethylammonium; trifluoroacetic acid In dichloromethane at 24.9℃; Rate constant; Mechanism; |
-
-
81623-63-4
(2S,4R,5S)-2-ethoxy-4-methyl-5-phenyl-1,3,2-oxazaphospholidine-2-thione

-
A
-
420087-33-8
(2R,3R)-2-methyl-3-phenylaziridine

-
B
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
C
-
57557-26-3
R-(+)-ethyl OS-dimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| With sodium methylate; methyl iodide Multistep reaction; |

-
-
124-41-4
sodium methylate

-
-
81623-63-4
(2S,4R,5S)-2-ethoxy-4-methyl-5-phenyl-1,3,2-oxazaphospholidine-2-thione

-
-
74-88-4
methyl iodide

-
A
-
420087-33-8
(2R,3R)-2-methyl-3-phenylaziridine

-
B
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
C
-
57557-26-3
R-(+)-ethyl OS-dimethyl phosphorothioate

| Conditions | Yield |
|---|---|
| 1.) several hours; Multistep reaction; | |
| Multistep reaction; |

-
-
152-18-1
O,O,O-trimethylthiophosphate

-
-
6832-16-2
methyl diazoacetate

-
A
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
B
-
57212-78-9
O,O-Dimethyl-S-(methoxycarbonylmethyl)-thiophosphorsaeure

| Conditions | Yield |
|---|---|
| With boron trifluoride diethyl etherate In dichloromethane |


| Conditions | Yield |
|---|---|
| With methanol |

| Conditions | Yield |
|---|---|
| With diethyl ether at 100℃; |

| Conditions | Yield |
|---|---|
| With methanol |

| Conditions | Yield |
|---|---|
| Stage #1: O,O-dimethyl phosphorodithioic acid With chlorine In toluene at 5 - 10℃; for 1.5h; Heating / reflux; Stage #2: With potassium hydroxide In water; toluene pH=> 7.0; Stage #3: methyl iodide In acetonitrile at 20℃; for 10h; |
-
-
1113-02-6
omethoate

-
A
-
512-56-1
trimethyl phosphite

-
B
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
C
-
20938-74-3
mercapto-acetic acid methylamide

-
D
-
868-85-9
Dimethyl phosphite

| Conditions | Yield |
|---|---|
| With oxygen In acetone for 0.0333333h; Plasma treatment; |

-
-
298-00-0
parathion-methyl

-
A
-
100-02-7
4-nitro-phenol

-
B
-
58175-57-8
2-propyl-1-pentanol

-
C
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
D
-
79-41-4
poly(methacrylic acid)

-
E
-
950-35-6
methyl paraoxon

-
F
-
84-66-2
Diethyl phthalate

-
G
-
123-31-9
hydroquinone

-
H
-
507-09-5
thioacetic acid

| Conditions | Yield |
|---|---|
| With oxygen at 22.2 - 25.3℃; Kinetics; Quantum yield; Wavelength; UV-irradiation; Neat (no solvent); |

-
-
298-00-0
parathion-methyl

-
A
-
100-02-7
4-nitro-phenol

-
B
-
58175-57-8
2-propyl-1-pentanol

-
C
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
D
-
79-41-4
poly(methacrylic acid)

-
E
-
950-35-6
methyl paraoxon

-
F
-
802294-64-0
propionic acid

-
G
-
123-31-9
hydroquinone

-
H
-
507-09-5
thioacetic acid

| Conditions | Yield |
|---|---|
| With water at 22.2 - 25.3℃; Kinetics; Quantum yield; Wavelength; UV-irradiation; Neat (no solvent); Inert atmosphere; |


| Conditions | Yield |
|---|---|
| In further solvent(s) to (MeO)2(MeS)PO Me3SnI added, stirred at 85°C; volatiles removed, residue crystalized; elem. anal.; | 95% |
-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
A
-
3711-50-0
phosphorochloridothioic acid,O,S-dimethyl ester

-
B
-
677-24-7
methyl dichlorophosphoridate

| Conditions | Yield |
|---|---|
| With trichlorophosphate at 75℃; for 2.5h; Chlorination; | A 72% B n/a |
-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
-
17356-08-0
thiourea

-
-
121255-86-5
S-methyl-isothiourea; S-methyl-isothiuronium-(O,S-dimethyl thiophosphate )

| Conditions | Yield |
|---|---|
| With ethanol |
-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
-
75-50-3
trimethylamine

-
-
14495-24-0
tetramethyl-ammonium; (O,S-dimethyl thiophosphate )

| Conditions | Yield |
|---|---|
| With diethyl ether |
-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
-
74-88-4
methyl iodide

-
-
2181-42-2
trimethylsulphonium iodide

| Conditions | Yield |
|---|---|
| at 100℃; |
-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
-
2857-97-8
trimethylsilyl bromide

-
-
65315-92-6
S-methyl O,O-bis(trimethylsilyl)phosphorothioate

| Conditions | Yield |
|---|---|
| In dichloromethane |
-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
-
10369-92-3
methyl-carbamic acid-(2-diethylamino-ethyl ester)

| Conditions | Yield |
|---|---|
| In benzene |
-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
-
18515-57-6
ethyl-carbamic acid-(2-diethylamino-ethyl ester)

| Conditions | Yield |
|---|---|
| In benzene |
-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
-
18515-53-2
N-Methyl-carbaminsaeure-(2-diisopropylamino-ethylester)

| Conditions | Yield |
|---|---|
| In benzene |
-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
-
18515-58-7
N-Ethyl-carbaminsaeure-(2-diisopropylamino-ethylester)

| Conditions | Yield |
|---|---|
| In benzene |
-
-
152-20-5
O,O,S-trimethyl phosphorothioate

-
-
18515-54-3
N-Methyl-carbaminsaeure-(2-dibutylamino-ethylester)

| Conditions | Yield |
|---|---|
| In benzene |
Dimethylthiomethylphosphate Chemical Properties
Chemical Name: O,O,S-Trimethyl phosphorothioate
IUPAC NAME: [Methoxy(methylsulfanyl)phosphoryl]oxymethane
CAS No.: 152-20-5
Molecular Formula: C3H9O3PS
Molecular Weight: 156.14 g/mol
Density: 1.211 g/cm3
Flash Point: 56 °C
Boiling Point: 169 °C at 760 mmHg
Following is the structure of Dimethylthiomethylphosphate (152-20-5):
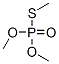
The chemical synonymous of Dimethylthiomethylphosphate (152-20-5) are O,O,S-Trimethylphosphorothiolate ; Trimethylphosphorothioate ; Phosphorothioic acid, O,O,S-trimethyl ester ; HC-7901 ; Thiophosphoric acid O,O,S-trimethyl ; Thiophosphoric acid O,O,S-trimethyl ester
Dimethylthiomethylphosphate Toxicity Data With Reference
| 1. | orl-rat TDLo:40 mg/kg (female 20D post):REP | ARTODN Archives of Toxicology. 61 (1988),378. | ||
| 2. | orl-rat TDLo:40 mg/kg (female 8-10D post):TER | TOLED5 Toxicology Letters. 32 (1986),185. | ||
| 3. | orl-rat LD50:15 mg/kg | JAFCAU Journal of Agricultural and Food Chemistry. 27 (1979),463. | ||
| 4. | ipr-rat LD50:51 mg/kg | ARTODN Archives of Toxicology. 51 (1982),221. | ||
| 5. | ivn-rat LD50:45 mg/kg | DTESD7 Developments in Toxicology and Environmental Science. 8 (1980),631. | ||
| 6. | orl-mus LD50:38 mg/kg | PCBPBS Pesticide Biochemistry and Physiology. 24 (1985),251. | ||
| 7. | ipr-mus LD50:5 mg/kg | ACPMAP Actualites Pharmacologiques. 16 (1963),7. | ||
| 8. | ivn-mus LD50:123 mg/kg | DTESD7 Developments in Toxicology and Environmental Science. 8 (1980),631. |
Dimethylthiomethylphosphate Safety Profile
Poison by ingestion, intravenous, and intraperitoneal routes. An experimental teratogen. Other experimental reproductive effects. When heated to decomposition it emits toxic fumes of POx and SOx.
Related Products
- Dimethylthiomethylphosphate
- 15220-85-6
- 152211-15-9
- 1522-13-0
- 152213-40-6
- 15221-84-8
- 152218-75-2
- 1522-22-1
- 152231-44-2
- 1522-34-5
- 15224-07-4
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View