-
Name
Diphenylphosphinyl chloride
- EINECS 216-107-2
- CAS No. 1499-21-4
- Article Data72
- CAS DataBase
- Density 1.25 g/cm3
- Solubility Decomposes in water
- Melting Point 20 °C
- Formula C12H10ClOP
- Boiling Point 369.2 °C at 760 mmHg
- Molecular Weight 236.638
- Flash Point 177.1 °C
- Transport Information UN 2920 8/PG 2
- Appearance clear colorless to yellow liquid
- Safety 16-26-36/37/39-45
- Risk Codes 10-34
-
Molecular Structure
-
Hazard Symbols
 C,
C, F
F
- Synonyms Phosphinicchloride, diphenyl- (6CI,7CI,8CI,9CI);Chlorodiphenylphosphine oxide;Diphenylphosphorus oxychloride;NSC175848;
- PSA 26.88000
- LogP 3.15430
Synthetic route

| Conditions | Yield |
|---|---|
| With thionyl chloride In toluene for 2h; Heating; | 100% |
| With phosphorus pentachloride | |
| With thionyl chloride for 2h; Heating; |

| Conditions | Yield |
|---|---|
| With ozone In dichloromethane at -80℃; | 99% |
| With sulfur dioxide; chlorine | 88% |
| 80.3% |

| Conditions | Yield |
|---|---|
| With oxygen In toluene for 4h; | 93% |
-
-
1041751-59-0
methyl (2R)-2-[(diphenylphosphoryl)(methyl)amino]-2-phenylethanoate

-
B
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| Stage #1: methyl (2R)-2-[(diphenylphosphoryl)(methyl)amino]-2-phenylethanoate With hydrogenchloride; water In methanol for 0.5h; Stage #2: With sodium hydrogencarbonate In methanol; water | A 90% B n/a |

| Conditions | Yield |
|---|---|
| With dibutyl hydrogen phosphite | 65% |

| Conditions | Yield |
|---|---|
| for 50h; Ambient temperature; | 42% |
-
-
14278-72-9
diphenylthiophosphinic acid

-
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| With chlorine |
-
-
644-97-3
Dichlorophenylphosphine

-
-
587-85-9
diphenylmercury(II)

-
A
-
1079-66-9
chloro-diphenylphosphine

-
B
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| at 230 - 240℃; | |
| at 230 - 240℃; |


| Conditions | Yield |
|---|---|
| With thionyl chloride | |
| With tetrachloromethane; triethylamine In chloroform at 5℃; | |
| Multi-step reaction with 2 steps 1: dihydrogen peroxide; sodium hydroxide / water / 1 h / 90 - 100 °C 2: thionyl chloride / toluene / 3 h / 80 °C View Scheme |
-
-
56372-47-5
N,N-diethyl P,P-diphenylphosphinamide

-
A
-
1079-66-9
chloro-diphenylphosphine

-
B
-
1101-41-3
Tetraphenyldiphosphin

-
C
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| With pyridine; trichlorosilane In benzene Title compound not separated from byproducts; |

-
-
108-94-1
cyclohexanone

-
-
1079-66-9
chloro-diphenylphosphine

-
A
-
1707-03-5
diphenyl-phosphinic acid

-
B
-
38868-16-5
cyclohex-1-en-1-yl(diphenyl)phosphine oxide

-
C
-
20187-71-7
(1-hydroxycyclohexyl)diphenylphosphine oxide

-
F
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| at 30 - 40℃; for 6h; Kinetics; Mechanism; Product distribution; various time; |

-
-
1079-66-9
chloro-diphenylphosphine

-
-
76-03-9
trichloroacetic acid

-
A
-
79-36-7
dichloroacethyl chloride

-
B
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| Ambient temperature; |


| Conditions | Yield |
|---|---|
| With phosphorous acid trimethyl ester; triethyl phosphite | |
| With amino acid at 20 - 25℃; |

-
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| In chloroform for 0.25h; Heating; |
-
-
20180-11-4
O-methyl-P,P-diphenylselenophosphinate

-
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| With sulfuryl dichloride at -20℃; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: 30percent H2O2 2: SOCl2 View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: Diethyl phosphonate; magnesium; iodine / tetrahydrofuran / 1 h / Cooling with ice; Reflux 2: dihydrogen peroxide; sodium hydroxide / water / 1 h / 90 - 100 °C 3: thionyl chloride / toluene / 3 h / 80 °C View Scheme | |
| Multi-step reaction with 3 steps 1.1: magnesium; 1,2-dibromomethane / tetrahydrofuran / 1 h / Reflux; Inert atmosphere 1.2: 0 °C / Reflux 2.1: dihydrogen peroxide; sodium hydroxide / 1 h / 100 °C / Inert atmosphere 3.1: thionyl chloride / toluene / 3 h / 80 °C / Inert atmosphere View Scheme |

| Conditions | Yield |
|---|---|
| With trichlorophosphate In diethyl ether at 20℃; for 21.5h; Inert atmosphere; | |
| Multi-step reaction with 3 steps 1: phosphonic acid diethyl ester / tetrahydrofuran / 1 h / Reflux 2: dihydrogen peroxide; sodium hydroxide / water / 1 h / 95 - 100 °C 3: thionyl chloride / toluene / 3 h / 80 °C View Scheme |

| Conditions | Yield |
|---|---|
| Stage #1: phosphonic acid diethyl ester; halo(phenyl)magnesium Stage #2: Atherton-Todd Synthesis; |
-
-
824-72-6
P,P-dichlorophenylphosphine oxide

-
-
108-90-7
chlorobenzene

-
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| With phosphorus at 340 - 360℃; for 4h; Reagent/catalyst; Temperature; Inert atmosphere; Large scale; | 233 kg |
-
-
4559-70-0
Diphenylphosphine oxide

-
A
-
5849-36-5
diphenylphosphinic anhydride

-
B
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| With tetrachloromethane; ((2S,3R)-2-((R)-2-((tert-butoxycarbonyl)amino)-propanamido)-3-((tert-butyldiphenylsilyl)oxy)butyl)(methyl)diphenylphosphonium iodide; caesium carbonate In diethyl ether at 20℃; for 0.833333h; |

| Conditions | Yield |
|---|---|
| With sodium azide In acetonitrile at 23℃; for 12h; | 100% |
| With sodium azide In acetonitrile for 16h; Ambient temperature; | 90% |
| With sodium azide In acetone for 18h; Ambient temperature; | 66% |
| With triethylammonium azide In chloroform | |
| With trimethylsilylazide |
-
-
18156-74-6
1-(Trimethylsilyl)imidazole

-
-
1499-21-4
Diphenylphosphinic chloride

-
A
-
75-77-4
chloro-trimethyl-silane

-
B
-
71638-08-9
1-(diphenylphosphinoyl)imidazole

| Conditions | Yield |
|---|---|
| In dichloromethane for 1h; Ambient temperature; | A n/a B 100% |

-
-
771-61-9
2,3,4,5,6-pentafluorophenol

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
138687-69-1
pentafluorophenyl diphenyl-phosphinate

| Conditions | Yield |
|---|---|
| With 1H-imidazole In dichloromethane for 1h; Ambient temperature; | 100% |
| With 1H-imidazole In dichloromethane at 25℃; for 3h; | 97% |
| With 1H-imidazole In dichloromethane Ambient temperature; | 92% |
-
-
2028-63-9
but-3-yn-2-ol

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
23834-42-6
O-1-methyl-2-propynyl phenylphenylphosphinate

| Conditions | Yield |
|---|---|
| With triethylamine; trifluoroacetic acid In dichloromethane at 0 - 20℃; Inert atmosphere; | 100% |
-
-
112780-08-2, 129364-62-1, 109-50-2
hex-3-yn-2-ol

-
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| With triethylamine; trifluoroacetic acid In dichloromethane at 0 - 20℃; Inert atmosphere; | 100% |
-
-
13139-86-1
4-methoxyphenyl magnesium bromide

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
795-44-8
4-(diphenylphosphoryl)benzonitrile

| Conditions | Yield |
|---|---|
| In tetrahydrofuran at 0 - 20℃; for 2h; Inert atmosphere; | 100% |

| Conditions | Yield |
|---|---|
| With n-butyllithium In tetrahydrofuran; hexane cooling; | 99% |
| Stage #1: (R)-(+)-N-ethyl-1,1'-binaphthyl-2,2'-diamine With n-butyllithium In tetrahydrofuran; hexane at -40℃; for 0.5h; Stage #2: Diphenylphosphinic chloride In tetrahydrofuran; hexane at -40 - 20℃; | 99% |
| Stage #1: (R)-(+)-N-ethyl-1,1'-binaphthyl-2,2'-diamine With n-butyllithium In tetrahydrofuran; hexane at -40℃; Stage #2: Diphenylphosphinic chloride In tetrahydrofuran; hexane at -40 - 20℃; Further stages.; |

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
872423-09-1
(1R,2R)-2-((R)-(diphenylphosphinylamino)phenyl-methyl)cyclopropane carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: (1R,2R)-2-((R)-Amino-phenyl-methyl)-cyclopropanecarboxylic acid methyl ester; hydrochloride; Diphenylphosphinic chloride With triethylamine In dichloromethane at 100℃; for 0.166667h; microwave irradiation; Stage #2: With MP-isocyanate; MP-trisamine at 100℃; for 0.0833333h; microwave irradiation; | 99% |
-
-
18531-94-7
(R)-1,1'-Bi-2-naphthol

-
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| With sodium hydride In tetrahydrofuran at 20℃; for 3h; | 99% |
| Stage #1: (R)-1,1'-Bi-2-naphthol With sodium hydride In tetrahydrofuran at 0℃; for 0.25h; Inert atmosphere; Stage #2: Diphenylphosphinic chloride In N,N-dimethyl-formamide at 0 - 20℃; for 3.25h; Inert atmosphere; | 96% |
| Stage #1: (R)-1,1'-Bi-2-naphthol With sodium hydride In tetrahydrofuran at 0℃; for 0.25h; Stage #2: Diphenylphosphinic chloride In tetrahydrofuran at 0 - 20℃; | |
| With sodium hydride In tetrahydrofuran at 0 - 20℃; |

| Conditions | Yield |
|---|---|
| Stage #1: 3-Bromophenol With dmap; triethylamine In dichloromethane at 0℃; Stage #2: Diphenylphosphinic chloride In dichloromethane at 20℃; | 99% |
| With dmap; triethylamine In dichloromethane at 0 - 20℃; for 12h; Inert atmosphere; | 96% |
| With dmap; N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; for 16h; Inert atmosphere; Schlenk technique; | 96% |
-
-
2133-40-6
L-proline methyl ester monohydrochloride

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
608533-84-2
(S)-1-(Diphenyl-phosphinoyl)-pyrrolidine-2-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: L-proline methyl ester monohydrochloride With N-ethyl-N,N-diisopropylamine In tetrahydrofuran for 0.5h; Inert atmosphere; Stage #2: Diphenylphosphinic chloride With N-ethyl-N,N-diisopropylamine In tetrahydrofuran for 36h; Inert atmosphere; | 99% |
-
-
16652-71-4
benzyl (2S)-2-pyrrolidinecarboxylate hydrochloride

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
62056-97-7
P-(S-2-benzyloxycarbonylpyrrolidin-1-yl),P,P-diphenylphosphine P-oxide

| Conditions | Yield |
|---|---|
| Stage #1: benzyl (2S)-2-pyrrolidinecarboxylate hydrochloride With N-ethyl-N,N-diisopropylamine In tetrahydrofuran for 0.5h; Inert atmosphere; Stage #2: Diphenylphosphinic chloride With N-ethyl-N,N-diisopropylamine In tetrahydrofuran for 72h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In toluene at 25℃; for 24h; | 99% |

| Conditions | Yield |
|---|---|
| Stage #1: 4,4'-Dihydroxybenzophenone With triethylamine In tetrahydrofuran at 0℃; for 0.333333h; Stage #2: Diphenylphosphinic chloride In tetrahydrofuran at 0 - 20℃; for 2h; | 99% |
-
-
148-24-3
8-quinolinol

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
1394229-21-0
diphenyl (8-quinolinyloxy)phosphinate

| Conditions | Yield |
|---|---|
| With diisopropylamine In tetrahydrofuran at 20℃; | 98% |
| With sodium hydride In tetrahydrofuran at 0℃; Inert atmosphere; Schlenk technique; |

| Conditions | Yield |
|---|---|
| Stage #1: 4-Hydroxybenzophenone With triethylamine In tetrahydrofuran at 0℃; for 0.333333h; Stage #2: Diphenylphosphinic chloride In tetrahydrofuran at 0 - 20℃; for 2h; | 98% |
| With triethylamine In tetrahydrofuran at 20℃; for 2h; Cooling with ice; | 93% |
-
-
407-25-0
trifluoroacetic anhydride

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
78811-86-6
Diphenyl(trifluoracetoxy)phosphanoxid

| Conditions | Yield |
|---|---|
| With 1-methyl-1H-imidazole In benzene | 97% |
| With 1-methyl-1H-imidazole Ambient temperature; |
-
-
367277-98-3
α-methyl(3-bromo-2-naphthalene)methanol

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
367277-97-2
1-(3-bromo-2-naphthalenyl)ethyl diphenylphosphinate

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane for 1h; | 97% |

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
872423-08-0
(1S,2S)-2-((S)-(diphenylphosphinylamino)phenyl-methyl)cyclopropane carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| Stage #1: (1S,2S)-2-((S)-Amino-phenyl-methyl)-cyclopropanecarboxylic acid methyl ester; hydrochloride; Diphenylphosphinic chloride With triethylamine In dichloromethane at 100℃; for 0.166667h; microwave irradiation; Stage #2: With MP-isocyanate; MP-trisamine at 100℃; for 0.0833333h; microwave irradiation; | 97% |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In toluene at 25℃; for 24h; | 97% |

| Conditions | Yield |
|---|---|
| With aluminum (III) chloride In toluene at 25℃; for 24h; | 97% |

| Conditions | Yield |
|---|---|
| With ammonia In dichloromethane at -78 - 20℃; | 96% |
| With ammonia In methanol; dichloromethane | 80% |
| With ammonia In tetrahydrofuran; water Inert atmosphere; | 76% |
-
-
2216-51-5
(-)-menthol

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
16933-43-0
(1R,2S,5R)-2-isopropyl-5-methylcyclohexyl diphenylphosphinate

| Conditions | Yield |
|---|---|
| With dmap; triethylamine In dichloromethane at 20℃; for 0.5h; Inert atmosphere; | 96% |
| With pyridine for 24h; Heating; | 32% |
| With sodium In toluene | |
| With pyridine | |
| With 1H-imidazole In diethyl ether; dichloromethane at 0 - 20℃; Inert atmosphere; |
-
-
2577-48-2
methyl (2S)-pyrrolidine carboxylate

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
608533-84-2
(S)-1-(Diphenyl-phosphinoyl)-pyrrolidine-2-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran | 96% |
-
-
7328-91-8
2,2-Dimethyl-1,3-diaminopropane

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
596106-64-8
N,N'-bis(P,P-diphenylphosphinoyl)-2,2-dimethyl-1,3-propanediamine

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine In dichloromethane at 0 - 20℃; | 96% |
| With triethylamine at 0℃; for 6h; |
-
-
79815-19-3
methyl (3S)-1,2,3,4-tetrahydro-3-isoquinolinecarboxylate

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
608533-83-1
(S)-2-(Diphenyl-phosphinoyl)-1,2,3,4-tetrahydro-isoquinoline-3-carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| With triethylamine In tetrahydrofuran | 96% |
-
-
850089-11-1
(1S,2S)-2-((S)-Amino-phenyl-methyl)-cyclopropanecarboxylic acid methyl ester

-
-
1499-21-4
Diphenylphosphinic chloride

-
-
872423-08-0
(1S,2S)-2-((S)-(diphenylphosphinylamino)phenyl-methyl)cyclopropane carboxylic acid methyl ester

| Conditions | Yield |
|---|---|
| In dichloromethane at 20℃; for 10h; | 96% |

-
-
1499-21-4
Diphenylphosphinic chloride

| Conditions | Yield |
|---|---|
| With 1H-imidazole In tetrahydrofuran; 1,2-dichloro-ethane at 60℃; for 16h; | 96% |
Diphenylphosphinyl chloride Specification
The IUPAC name of Diphenylphosphinic chloride is [chloro(phenyl)phosphoryl]benzene. With the CAS registry number 1499-21-4 and EINECS 216-107-2, it is also named as Phosphinic chloride, P,P-diphenyl-. The product's categories are Pharmaceutical Intermediates; Phosphorus Compounds; Biochemistry; Coupling Reactions (Peptide Synthesis); Peptide Synthesis; Protection & Derivatization Reagents (for Synthesis); Reagents for Oligosaccharide Synthesis; Synthetic Organic Chemistry. It is clear colorless to yellow liquid which is used in pesticide synthesis and as fire retardant. Additionally, this chemical should be stored at the temperature of 2-8 °C.
The other characteristics of this product can be summarized as: (1)ACD/LogP: 2.75; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.75; (4)ACD/LogD (pH 7.4): 2.75; (5)ACD/BCF (pH 5.5): 72.67; (6)ACD/BCF (pH 7.4): 72.67; (7)ACD/KOC (pH 5.5): 748.09; (8)ACD/KOC (pH 7.4): 748.09; (9)#H bond acceptors: 1; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 26.88 Å2; (13)Index of Refraction: 1.585; (14)Molar Refractivity: 63.03 cm3; (15)Molar Volume: 187.9 cm3; (16)Polarizability: 24.98×10-24 cm3; (17)Surface Tension: 45.2 dyne/cm; (18)Density: 1.25 g/cm3; (19)Flash Point: 177.1 °C; (20)Enthalpy of Vaporization: 59.17 kJ/mol; (21)Boiling Point: 369.2 °C at 760 mmHg; (22)Vapour Pressure: 2.57E-05 mmHg at 25°C.
Preparation of Diphenylphosphinic chloride: It can be obtained by diphenylphosphinous acid chloride and propan-2-one. This reaction reacts at ambient temperature for 50 hours. The yield is 42%.
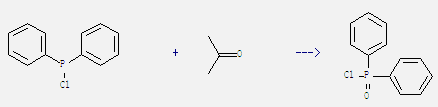
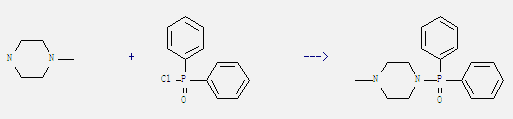
Uses of Diphenylphosphinic chloride: It can react with 1-methyl-piperazine to get N-(diphenylphosphinyl)-N'-methylpiperazine. This reaction needs solvent CH2Cl2 at temperature of 0 °C. The reaction time is 10 min. The yield is 95%.
When you are using this chemical, please be cautious about it as the following:
It is flammable and can cause burns, so people should keep away from sources of ignition. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. If you want to contact this product, you must wear suitable protective clothing, gloves and eye/face protection. In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.)
People can use the following data to convert to the molecule structure.
1. SMILES:ClP(=O)(c1ccccc1)c2ccccc2
2. InChI:InChI=1/C12H10ClOP/c13-15(14,11-7-3-1-4-8-11)12-9-5-2-6-10-12/h1-10H
3. InChIKey:QPQGTZMAQRXCJW-UHFFFAOYAH
Related Products
- Diphenylphosphinyl chloride
- 14992-59-7
- 14992-62-2
- 149926-91-0
- 149934-21-4
- 14994-25-3
- 1499-46-3
- 149947-15-9
- 149947-19-3
- 149949-86-0
- 149950-60-7
Hot Products
About|Contact|Cas|Product Name|Molecular|Country|Encyclopedia
Message|New Cas|MSDS|Service|Advertisement|CAS DataBase|Article Data|Manufacturers | Chemical Catalog
©2008 LookChem.com,License: ICP
NO.:Zhejiang16009103
complaints:service@lookchem.com Desktop View